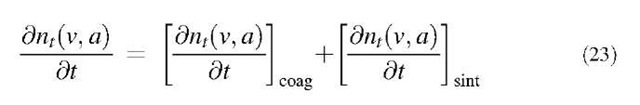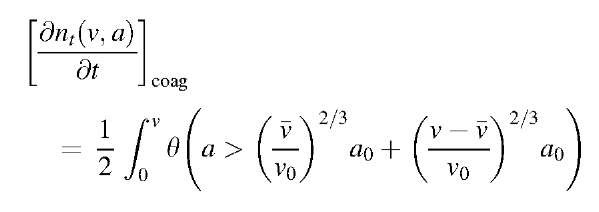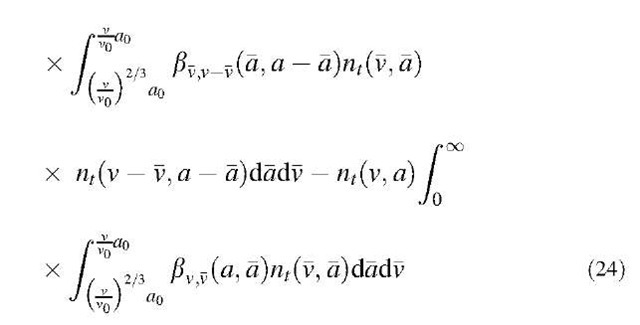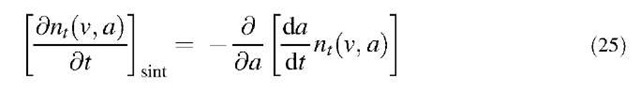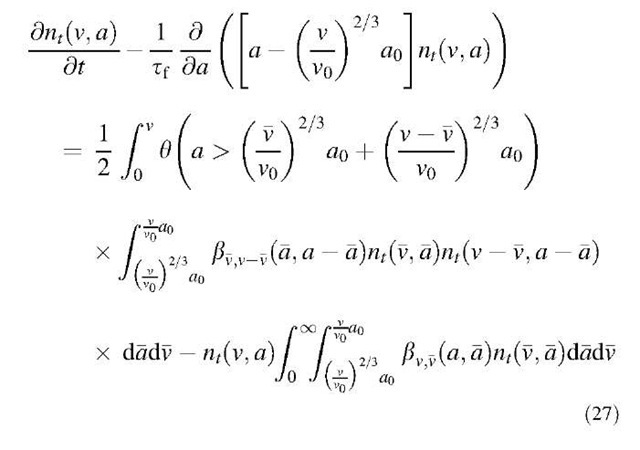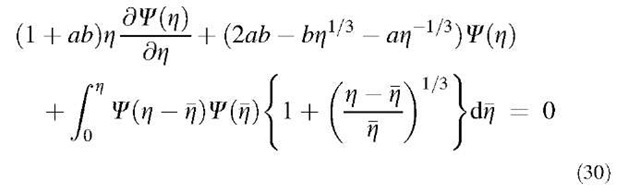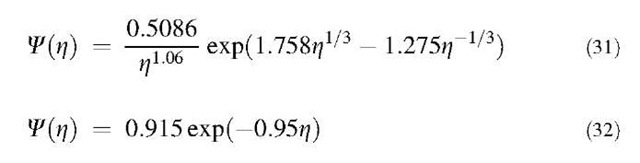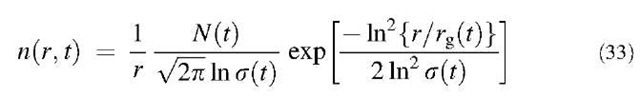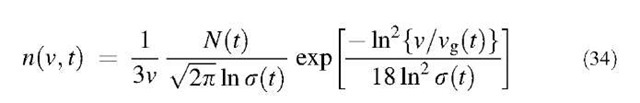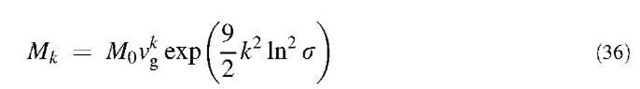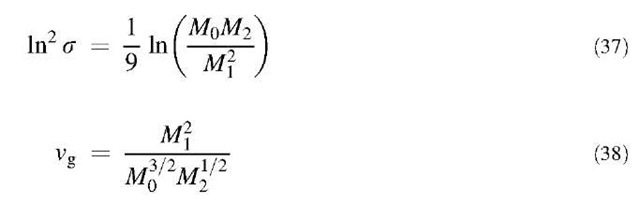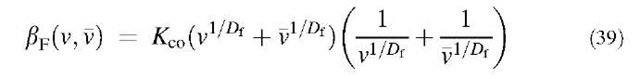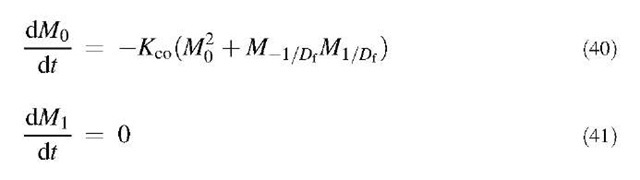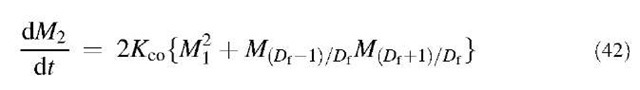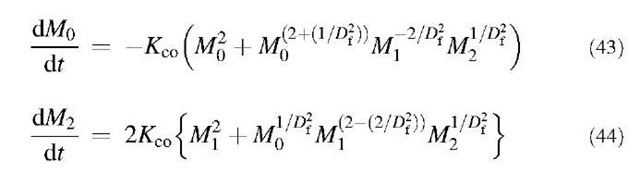SOLUTIONS TO THE COAGULATION EQUATION
Methods of solving a coagulation equation were summarized by Williams and Loyalka.[17] These methods range from the discrete (computationally intensive) and sectional models in which Eq. 5 is transformed into a number of differential equations, to the less accurate mo-nodisperse models. Approximate solutions can be found using the method of moments. Exact solutions for asymptotic limiting cases can be obtained with the self-preserving theory.
Sectional Method
Because solving the coagulation equation with a direct numerical method is impractical owing to its time-consuming property, several approximate methods have been developed. The sectional method developed by Gelbard et al.[18] is known as a very accurate but comparatively time-efficient tool. Their model solved a one-dimensional form of the aerosol general dynamic equation by dividing the particle size domain into a finite number of sections by particle volume, and by calculating the addition and subtraction of particle mass to each section. The model assumed that particles kept their spherical shape during growth; therefore the volume sections corresponded to sections of particle size. However, as irregular particles have become omnipresent in nanoparticle production (e.g., generation of titanic or silica particles by gas-phase reaction), substantial progress has been made in developing models that account for irregular particle shape through fractal dimensions. The volume and surface area of irregularly shaped particles were chosen as the two particle size dimensions because these are the most commonly employed powder properties in engineering applications. Then the two-dimensional aerosol dynamic equations were solved by extending a one-dimensional sectional technique to the two-dimensional space.[19] A two-dimensional particle size distribution function is defined as nt(v,a), where nt(v,a)dadv is the number density of particles having a volume between v and v+dv and a surface area between a and a+da at time t. For an aerosol that is formed by gas-phase reaction at high temperatures, the rate of change in nt(v,a) is given by the rate of simultaneous coagulation and coalescence (by sintering) among aerosol particles:[19,20]
The coagulation term in Eq. 23 can be obtained by extending the classical collision theory to the two-dimensional space v,a as:
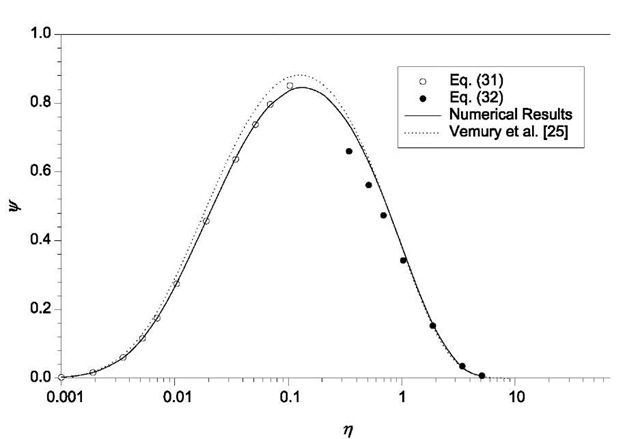
Fig. 3 Self-preserving size distribution for Brownian coagulation.
where v0 and a0 are the volume and surface area, respectively, of the primary particle, which is the smallest possible particle (e.g., a molecule or a monomer).
The sintering contribution in Eq. 23 is related to the particle sintering rate through the continuity:[20]
The sintering of solid particles or highly viscous fluids can be the result of various sintering mechanisms such as solid-state surface or volume diffusion and viscous flow. For the mechanism of viscous flow coalescence beyond a small initial time scale, Hiram and Nir[21] found that the neck size approaches the radius of the resulting sphere exponentially. For longer times, this leads to a similar behavior for the particle surface:[20]
where afinal is the surface area of the completely fused sphere of volume v, and tf is the characteristic coalescence time. The characteristic time for coalescence or sintering tf is the time needed to reduce by 63% the excess agglomerate surface area over that of a spherical particle with the same mass.
Therefore by combining Eqs. 23 and 24 and Eqs. 25 and 26, the overall aerosol population balance equation can be written as:
Eq. 27 is a two-dimensional partial integrodifferential equation that needs to be solved numerically. In solving the conventional one-dimensional coagulation equation, a sectional model has been developed. The sectional method proves to be both computationally efficient and numerically robust, especially in dealing with aerosols having an extremely large size spectrum. Xiong and Pratsinis[19] presented a set of M ordinary differential equations (ODEs) using an efficient ODEs solver.
Self-Preserving Solution
One of the interesting features of coagulation known to date is that the shape of the size distribution of suspended particles undergoing coagulation often does not change after a long time and the distribution becomes self-preserving.
Conventionally, in the self-preserving formulation, the dimensionless particle volume is defined as:
and the dimensionless size distribution density function is defined as:
Friedlander and Wang[24] obtained the following equation for the Brownian coagulation in the continuum regime:
Friedlander and Wang derived analytical solutions of Eq. 30, shown in Eqs. 31 and 32, for small z regime and for large z regime, respectively:
Eq. 30 can be solved numerically for the entire z range. The results obtained by Friedlander and Wang are shown in Fig. 3 together with the results of numerical simulations by Vemury et al.,[25] who solved the coagulation equation (Eq. 5) using the sectional method of Landgrebe and Pratsinis.[26] Lai et al.[27] used the above technique to derive the self-preserving size distribution for the free molecule regime.
Wang and Friedlander[28] applied the same method to the Brownian coagulation either with slip correction or with simultaneous shear flow. However, in their work, they had to introduce an unrealistic assumption that some parameters are special functions of time. Pich et al.[29] obtained the self-preserving particle size distribution for the problem of simultaneous coagulation and condensation, but the similar constraint confined the application of the solution obtained.
Moment Method
Although the self-preserving size distribution theory played a very important role for researchers in understanding the coagulation mechanism, one shortcoming of the theory is its inability to resolve the size distribution for the time period before an aerosol attains the self-preserving size distribution. Therefore it was still necessary to resort to numerical calculations. However, during coagulation, the size distribution of particles changes by the interaction of particles in so many size classes at each time step that the computing time becomes excessive for the calculation of the change of size distribution of particles. To overcome this problem and to accelerate the computations, much effort has been made. Cohen and Vaughan[30] succeeded in reducing the coagulation equation to a set of ODEs for the moments of size distribution. By choosing a specific functional form for the distribution of the particle size, they were able to calculate the parameters of the size distribution as a function of the three leading moments. This method, known as the moment method, has the advantage of simplicity while providing important information on the change of the size distribution of aerosol.[31] Therefore the moment method has been widely applied in the simulation of aerosol coagula-tion.[32,33] Here, the application of the moment method to Brownian coagulation of fractal agglomerates,[34] which are often involved in the synthesis of nanoparticles, is addressed as an example for the continuum regime where the slip correction factor is equal to 1.
In the moment method, to represent a polydisperse aerosol size distribution, the log-normal function (one of the most commonly used mathematical forms for the study of the dynamics of particles) is used. The size distribution density function for particles whose radius is r for the log-normal distribution is written as:
where N(t) is the total number concentration of particles, s(t) is the geometric standard deviation based on particle radius, and rg(t) is the geometric number mean particle radius. For studying the coagulation problem in which two particles collide to become a particle whose volume is the sum of the two volumes, it is convenient to rewrite Eq. 33 in terms of particle volume:
where vg is the geometric number mean particle volume. If one obtains the time evolution of the three parameters N(t), s(t), and vg(t), the particle size distribution of the coagulating aerosol of interest for any time t can be constructed using Eq. 34.
The kth moment of the particle size distribution is written as:
where k is an arbitrary real number. According to the properties of a log-normal function, any moment can be written in terms of M0, vg, and s as follows:
If Eq. 36 is written for k=0, 1, and 2, and subsequently solved for vg and s in terms of M0, M1, and M2, we have:
The collision kernel of fractal agglomerates bF(v,v) covering the continuum regimes is represented by the following expression:[35]
where Kco is defined by Eq. 3, and Df is the mass fractal dimension, which varies between 1 and 3.[36] Substituting Eq. 39 into Eq. 5 and integrating from 0 to 1, one can obtain the following equations using Eq. 35:
Eq. 41 merely indicates that Mj=const. Substituting Eq. 36 for the moments appearing in the right-hand side of Eqs. 40 and 42, and substituting Eqs. 37 and 38 thereafter, we have the following two equations for k=0 and 2, respectively:
The governing equation has just been converted into a set of three first-order ODEs. Eqs. 41, 43, and 44 can be solved using any standard numerical package for solving first-order ODEs. After M0, Mi, and M2 are solved from Eqs. 43, 41, and 44, respectively, s and vg can be computed using Eqs. 37 and 38. Subsequently, the size distribution of the fractal agglomerates for any time can be constructed using Eq. 34.
Coagulation in Nanoparticle Synthesis
Among the most recent developments in the theoretical description of gas-to-particle conversion process is the simultaneous modeling of precursor decomposition, coagulation, and sintering processes in the aerosol, originally reported by Koch and Friedlander[20] and Kobata et al.,[37] and refined further by Xiong and Pratsinis,[19] Xiong et al.,[38] and Tsantilis and Pratsinis[39] using elaborate two-dimensional sectional models of the population balance equation. In chemical vapor synthesis (CVS), particles pass through the complete temperature profile of a hot wall reactor together with the gas stream. The CVS technique is based on the process of chemical vapor deposition (CVD), which is widely employed to prepare coatings at a high level of control of the growth parameter and, consequently, of the microstructure. As the process is very important for basic research and industrial production, the conditions for CVD growth have been determined for many material systems. The formation process of nanoparticles in the gas phase has been studied intensively both experimentally and theoretically. Many kinetic models exist to describe the dependence of the particle size on various synthesis parameters such as partial and total pressure, temperature, etc. It is of particular interest to study the particle formation on an atomic scale to understand the diffusional processes that lead to coagulation and particle rearrangement on the short time scale of their formation. Molecular dynamics (MD) simulation is used to study the particle formation and the coagulation/sintering processes.
CONCLUSION
Therefore the coagulation of particles suspended in a gas or a liquid strongly influences the particle size distribution and is of fundamental interest in a wide range of applications in science and engineering. In this chapter, some theories of coagulation are reviewed.
The coagulation of a monodisperse aerosol was studied for Brownian coagulation by Smoluchowski[1] and the change of particle number concentration with respect to time was provided. The general coagulation equation of polydisperse aerosol particles was introduced and the collision kernels for Brownian, gravitational, laminar shear, turbulent, and electrostatic coagulations were presented. In case of Brownian coagulation, the kinetic theory and the continuum diffusion theory were used to derive the collision kernel for the free molecule regime and the continuum regime, respectively, and the two theories were brought together by flux matching to produce the kernel for transition regime. At the end of this chapter, various solution techniques were introduced including the sectional method, the self-preserving solution, and the moment method. The sectional method provides the most accurate prediction on the size distribution changes. The self-preserving size distribution theory presents the asymptotic size distribution. The moment method has the advantage of simplicity while providing important information on the change of the size distribution of aerosol.