Role of the liver
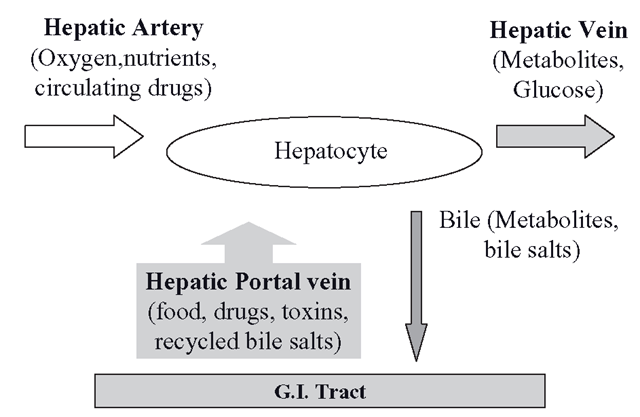
Drugs, toxins and all other chemicals can enter the body through a variety of routes. The major route is through the digestive system, but chemicals can by-pass the gut via the lungs and skin. Although the gut metabolizes many drugs, the liver is the main biotransforming organ and the CYPs and other metabolizing enzymes reside in the hepatocytes. These cells must perform two essential tasks at the same time. They must metabolize all substances absorbed by the gut whilst also processing all agents already present (from whatever source) in the peripheral circulation. This would not be possible through the conventional way that organs are usually supplied with blood from a single arterial route carrying oxygen and nutrients, leading to a capillary bed that becomes a venous outflow back to the heart and lungs. The circulation of the liver and the gut have evolved anatomically to solve this problem by receiving a conventional arterial supply and a venous supply from the gut simultaneously (Figure 2.3); all the blood eventually leaves the organ through the hepatic vein towards the inferior vena cava.
The hepatic arterial blood originates from the aorta and the venous arrangement is known as the hepatic portal system, which subsequently miniaturizes inside the liver into sinusoids, which are tiny capillary blood-filled spaces. This capillary network effectively routes everything absorbed from the gut direct to the hepatocytes, which are bathed at the same time in oxygenated arterial blood. Metabolic products can leave the hepatocytes through the hepatic vein or by a separate system of canalicali, which ultimately form the bile duct, which leads to the gut. So, essentially, there are two blood routes into the hepatocytes and one out, which ensures that no matter how a xenobiotic enters the body, it will be presented to the hepatocytes for biotransformation.
Figure 2.3 The hepatocytes can simultaneously metabolize xenobiotics in the circulation and those absorbed from the gut through their dual circulation of venous and arterial blood. Metabolites escape in the hepatic vein for eventual renal excretion, whilst biliary metabolites reach the gut
Drug and xenobiotic uptake: transporter systems
Although an agent might be presented to the vicinity of a hepatocyte, there is no guarantee it will enter the cell. This depends on the lipophilicity, size, charge and other physiochemi-cal properties of the agent. If an agent is too lipophilic, as described in section 2.3, it may enter a cell and become trapped in the membrane. Alternatively, if a drug is very water soluble, it would not be capable of crossing the lipid membrane bilayer of the cell. Until the last decade or so, it was often assumed that drug absorption would usually be simply through passive diffusion from high to low concentration. It is now apparent that many drugs and toxins which are charged or amphipathic diffuse rather poorly across lipid membranes and their successful cellular and systemic absorption is in a large part due to their exploitation of the complex membrane transport systems which are found not only in the gut, but also on the sinusoidal (sometimes called the basolateral) membranes of hepatocytes, which are bathed in blood from the portal circulation direct from the gut, as well as arterial blood. These membrane transporters regulate cellular entry of amino acids, sugars, steroids, lipids and hormones which are vital for homeostasis. We know this because if the hepatocyte transporters are inhibited, the bioavailability of several drugs increases because they escape hepatic clearance by the CYPs and other systems. Transporter proteins are found in all tissues and can be broadly categorized into two ‘superfamilies’; those that assist the entry of drugs, toxins and nutrients into cells (uptake, or influx transporters) and those that actively pump them out using ATP in the process, usually against concentration gradients.
Hepatic and gut uptake (influx) transporter systems
These transporters, usually known as the solute carriers (SLCs), are found in the liver, gut, brain, kidney and the placenta. These systems operate without using ATP and transport everything from small peptides to anions like bilirubin-related metabolites. The main hepatic uptake transporters are known as organic anion transporting peptides, or OATPs. These transporters originate from a gene known as SLCO1B1 which is found on chromosome 12. OATPs are sodium independent and they effectively operate a process of facilitated diffusion, known as electroneutral exchange. For every amphipathic molecule they pump in, they expel a neutralizing anion, like glutathione (GSH), bicarbonate or even a drug metabolite. The system is rather like a revolving door and many drugs enter gut epithelial cells and hepatocytes this way, particularly the more hydrophilic statins. The best documented OATPs are OATP1A2, OATP1B1 and OATP1B3. These transporters are vital to the uptake of several classes of drugs and OATP1B1 can be inhibited by gemfibrozil, rifampicin, cyclosporine and by the antiiHIV protease inhibitors such as ritonavir.
Regarding other hepatic transporters, NTCP (sodium taurocholate cotransporting polypeptide) transports bile salts, but also can handle rosuvastatin and NTCP has also been used to selectively target liver tumours by linking cytotoxic agents to bile salts. There are several other uptake transporters which are of most relevance in tissues other than the liver, such as the kidneys and the gut. The OATs pump small anions mainly in the kidney, but OAT2 and OAT5 are hepatic. OATs can be inhibited by the cephalosporin antibiotics, which may be linked with their renal toxicity.
Aims of biotransformation
Once drugs or toxins enter the hepatocytes, they are usually vulnerable to some form of biotransformation. Although you can see some of the many functions of CYPs and other biotransformational enzymes (Figure 2.2), it is essential to be clear on what they have to achieve with a given molecule. Looking at many endogenous substances like steroids or xenobiotic agents, such as drugs, all these compounds are mainly lipophilic. Drugs often parallel endogenous molecules in their oil solubility, although many are considerably more lipophilic than these molecules. Generally, drugs, and xenobiotic compounds, have to be fairly oil soluble or they would not be absorbed from the GI tract. Once absorbed these molecules could change both the structure and function of living systems and their oil solubility makes these molecules rather ‘ elusive , , in the sense that they can enter and leave cells according to their concentration and are temporarily beyond the control of the living system. This problem is compounded by the difficulty encountered by living systems in the removal of lipophilic molecules. As previously mentioned,even after the kidney removes them from blood by filtering them, the lipophilicity of drugs, toxins and endogenous steroids means that as soon as they enter the collecting tubules, they can immediately return to the tissue of the tubules, as this is more oil-rich than the aqueous urine. So the majority of lipophilic molecules can be filtered dozens of times and only low levels are actually excreted. In addition, very high lipophilicity molecules like some insecticides and fire retardants might never leave adipose tissue at all (unless moved by dieting or breast feeding, which mobilizes fats). Potentially these molecules could stay in our bodies for years. This means that for lipophilic agents:
• the more lipophilic they are, the more these agents are trapped in membranes, affecting fluidity and causing disruption at high levels;
• if they are hormones, they can exert an irreversible effect on tissues that is outside normal physiological control;
• if they are toxic, they can potentially damage endogenous structures;
• if they are drugs, they are also free to cause any pharmacological effect for a considerable period of time.
The aims of a biotransformational system include assembly of endogenous molecules, as well as clearance of these and related chemicals from the organism. These aims relate to control for endogenous steroid hormones (assembly and elimination), as well as protection- in the case of highly lipophilic threats, like drugs, toxins and hormone ‘mimics ’ (endocrine disruptors). Metabolizing systems have developed mechanisms to control balances between hormone synthesis and clearance so the organism can finely tune the effects of potent hormones such as sex-steroids. These systems also actually detect the presence of drugs and act to eliminate them.
Task of biotransformation
Essentially, the primary function of biotransforming enzymes such as CYPs is to ‘move’ a drug, toxin or hormone from the left-hand side of Figure 2.1 to the right-hand side. This means making very oil-soluble molecules highly water-soluble. This sounds impossible at first and anyone who has tried to wash their dishes without using washing up liquid will testify to this problem. However, if the lipophilic agents can be structurally altered, so changing their physicochemical properties, they can be made to dissolve in water. Once they are water-soluble, they can easily be cleared by the kidneys into urine and they will finally be eliminated.
Phase’s I-III of biotransformation
Most lipophilic agents that invade living systems, such as aromatic hydrocarbons, hormones, drugs and various toxins, vary in their chemical stability, but many are relatively stable in physiological environments for quite long periods of time. This is particularly true of polycyclic aromatics. This means that a considerable amount of energy must be put into any process that alters their structures. This energy expenditure will be carried out pragmatically. This means that some molecules may require several changes to attain water solubility, such as polycyclics, whilst others such as lorazepam and AZT, only one. The stages of biotransformation are often described as ’ Phases ’ I, II and III. Phase I metabolism mainly describes oxidative CYP reactions, but non-CYP oxidations such as reductions and hydrolyses are also sometimes included in the broad term ‘Phase I’. This has been highlighted as rather arbitrary and inconsistent and it is recommended that it is more accurate to refer to a particular process specifically, rather than using the loose term ‘Phase I’.
The term , Phase II , describes generally conjugative processes, where water, soluble endogenous sugars, salts or amino acids are attached to xenobiotics or endogenous chemicals. The very term ‘Phase II’ suggests that ‘Phase I’ processes must necessarily occur prior to conjugative reactions with a molecule. Although this does often happen, conjugation also occurs directly without prior ‘preparation’ by oxidative processes. The products of ‘Phase II’ tend also to be strongly associated with detoxification and high water solubility. This is not always the case either and it is important to realize that some conjugative ‘Phase II’ processes can form either toxic species, or metabolites even less water-soluble than the parent drug. The more recent term Thase III , describes the system of efflux pumps that excludes water-soluble products of metabolism from the cell to the interstitial fluid, blood and finally the kidneys. The efflux pumps can also exclude drugs as soon as they are absorbed from the gut, as well as metabolites. Although the Phase I-III terminology remains popular and thus is sometimes used in this topic, it is important to recognize the limitations of these terms in the description of many processes of biotransformation.
Biotransformation has a secondary effect, in that there is so much structural change in these molecules that pharmacological action is often removed or greatly diminished. Even if the metabolite retained some potential pharmacodynamic effects, its increased polarity compared with the parent drug means that the Phase III systems are likely to remove it relatively quickly, so diminishing any effects it might have exerted on the target tissue.
The use of therapeutic drugs is a constant battle to pharmacologically influence a system that is actively undermining the drugs’ effects by removing them as fast as possible. The processes of oxidative and conjugative metabolism, in concert with efflux pump systems, act to clear a variety of chemicals from the body into the urine or faeces, in the most rapid and efficient manner. The systems that manage these processes also sense and detect increases in certain lipophilic substances and this boosts the metabolic capability to respond to the increased load. The next topic will outline how mainly CYP-mediated oxidative systems achieve their aim of converting stable lipophilic agents to water-soluble products.