IMPACT OF CHEMICAL HETEROGENEITY
Design of surfaces using SAMs provides a hitherto unexplored approach to experimentally understand the effects of chemical heterogeneity of surfaces on polymer adsorption.[27,28] It is well known that millimeter- or micrometer-sized patterns in self-assembled monolayers can be created using techniques such as UV photooxida-tion, microcontact printing, and micromachining.[29] However, these patterns are large compared to the typical dimensions of a polymer chain and not predicted to dramatically alter the adsorption behavior. Patterning on length scales smaller than 200 nm using nano-lithography is possible but impractical for areas as large as 1 cm2 that are necessary for noninvasive optical measurements of adsorption. In contrast, mixed SAMs formed by coadsorp-tion from a mixture of long and short chains [e.g., CH3(CH2)9SH (DT) and HOOC(CH2)3SH (MPA)] can produce quenched heterogeneous surfaces with a statistical distribution of nanometer-sized domains when the relative proportion of the two species and the time of formation is carefully controlled.1-30-32-1
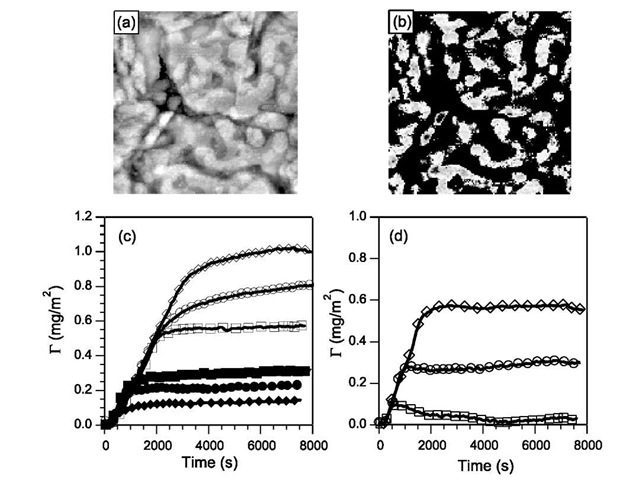
Fig. 5a shows a low-current STM image of the typical mixed monolayer used in the polymer adsorption. Bright domains correspond to areas dominated by a longer methyl-terminated alkanethiol (DT) while the rest of the gold surface corresponds to regions that are areas rich in the carboxylic acid terminated MPA. The STM image shows that the surface consists of isolated DT areas that are as small as a few nanometer in dimension and also regions where the DT domains cluster into larger patches. It is evident that the domains on the surface fall predominantly between 10 and 100 nm2 in area and that the heterogeneous surface is well suited to study polymer adsorption. Because SAMs of MPA are known[33] to exhibit a pKa greater than 5, variation of the solution pH can also be used to control the state of ionization of the acid groups on the surface and create a patchy distribution of charges on the surface. Using the heterogeneous SAMs we have studied the influence of chemical heterogeneity of the surface on adsorption of two broad classes of polymers: flexible, linear polymers and globular proteins.
Fig. 5 A mixed SAM showing patchy domains of DT in the STM image before (a) and after image processing (b). The size of the image is 200 x 200 nm. Adsorption of (c) P2VP and (d) PEO on homogeneous surfaces of DT (diamonds) and MPA (squares) compared to adsorption on a patchy surface (circles). Open symbols correspond to pH=4.5 and filled symbols to pH = 2.
Adsorption of Flexible Polymers on Chemically Heterogeneous Surfaces
A number of polymer-surface combinations are realizable and therefore experiments have focused on a subset using nonionic or ionic polymers. The experiments have explored the adsorption of an uncharged polymer [e.g., nonionic PEO or neutral poly(2-vinylpyridine) (P2VP) or neutral poly(acrylic acid)(PAA)] or a charged polymer (e.g., positively charged P2VP) on a largely neutral heterogeneous surfaces and a neutral polymer (e.g., PEO) on a surface with heterogeneous distribution of charge.[27,28] Physically, these combinations represent simplified cases where no electrostatic contributions to the surface-segment interaction exist.
Fig. 5c and d shows the typical G(t) for P2VP and PEO on the chemically heterogeneous surface containing both DT and MPA sites. A comparison with G(t) on homogeneous surfaces of either MPA or DT is also shown. The kinetic curves show a characteristic shape of a constant initial rate of adsorption followed by a slow approach to a plateau. For P2VP adsorption on each type of homogeneous surface (MPA or DT) the value of Gmax is almost four times larger at pH 4.5 0.6-1.2 mg/m2) when compared to the lower pH of 2.0 0.15-0.35 mg/m2). P2VP becomes ionized at the low pH and electrostatic repulsion between neighboring segments of a charged polymer chain leads to lower G as accumulation of charge within the surface layer is not favorable. In contrast, no effect of pH is observed on the adsorption of PEO and the G(t) shown in Fig. 5d remains unchanged.
At low pH, the largely hydrophobic backbone of P2VP is shielded by the hydrophilic (protonated) pyridine group, which decreases the adsorption affinity to the hydropho-bic methyl DT surface and results in a Gmax that is lower than the value observed on MPA. Thus at pH 2.0 the domains of DT in the heterogeneous surfaces correspond to sites that are less attractive to P2VP relative to the surrounding regions of MPA on the surface. In contrast, at pH 4.5 the DT sites are more attractive relative to the MPA sites.
The adsorption curves clearly show the effect of chemical heterogeneity. For both P2VP and PEO the initial rate of adsorption on the heterogeneous surface is the same as that observed on the homogeneous surfaces. Adsorption of either a charged polymer (P2VP at pH2) or an uncharged polymer (P2VP at pH4.5 and PEO) on a neutral surface shows that the adsorbed amount Gmax lies in between the value observed on the two types of homogeneous surfaces. Thus although the initial kinetics are not altered dramatically by the presence of heterogeneity, the surface heterogeneity does impact later stages of adsorption. Physically, the intermediate value of Gmax indicates that the effective surface adsorption energy of the polymer chain represents some average of the interactions with the two different sites. The experiments also suggest that in these simplified physical situations it does not matter if the minority domains are more or less attractive than the surrounding areas. Other recent experiments by Chun and coworkers[28] have shown that beyond a threshold density of DT sites on the surface, the adsorbed amount of the polymer depends on the fraction of heterogeneous sites on the surface.
These first experimental results on compositionally heterogeneous surface verify theoretical predictions regarding polymer adsorption on chemically heterogeneous surfaces. Van der Linden and coworkers used a self-consistent lattice model to study the influence of the degree of clustering of attractive and repulsive sites. On a surface with a patchwise distribution of chemical sites, which is similar to the experiments reported here, the lattice model predicted that the homopolymer adsorption was proportional to the fraction of adsorbing sites. Fig. 5 demonstrates that the adsorption behavior in a real system closely matches the lattice model. Zajac and Chakrabarti used a Monte Carlo simulation to explore the adsorption of a homopolymer on chemically heterogeneous surfaces containing 30-50% of sites that were either repulsive to the polymer or were simply inactive. They predicted that during the early stages the presence of surface impurities did not alter the growth kinetics of the adsorbed layer. However, as chains spread on the surface and competed for sites, there was a net decrease in the number of chains that were adsorbed and that the decrease was proportional to the fraction of inactive or repulsive surface sites. The kinetic curves of G(t) measured in Fig. 5 corroborate the predictions from the simulations.
Adsorption of Globular Proteins on Chemically Heterogeneous Surfaces
Protein adsorption at a surface is ubiquitous in biology and a critical component of synthetic implants and biocompatibility. Because of the intrinsic chemical heterogeneity of a globular protein and its nanometer size, the role of surface heterogeneity in protein adsorption is significant. Therefore we have focused on the adsorption behavior and morphology of a common protein-bovine serum albumin (BSA). To better understand the impact of surface heterogeneity, we also review the adsorption of BSA on homogeneous surfaces and illustrate the changes in adsorption as chemical heterogeneity is introduced into the surface.
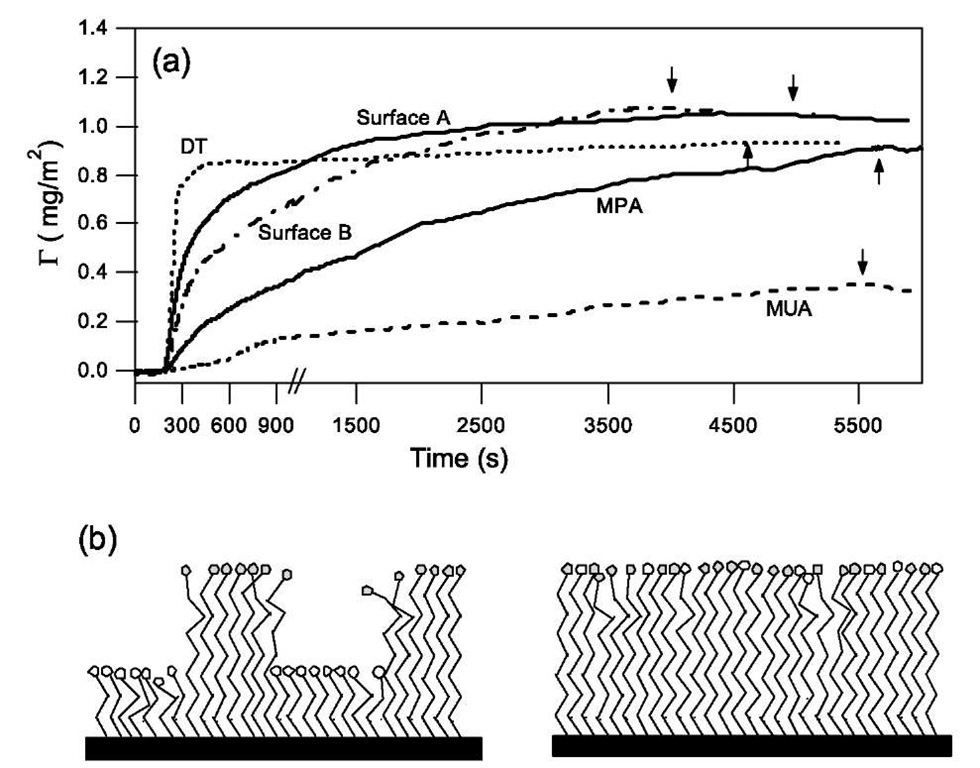
Fig. 6a shows adsorption kinetics on chemically homogeneous surfaces formed using single-component SAMs of DT, MPA, and MUA (HOOC(CH2)10SH). G(t) shows that the protein is irreversibly adsorbed on those model surfaces and that the surface chemical properties clearly affect the adsorption amount. The hydrophobicity of each homogeneous surface in Fig. 6a was probed using contact angle measurement with water, which gave 0advancing as 107°, 35°, and 25° for SAMs of DT, MPA, and MUA, respectively. The plateau values (Gmax) therefore follow a trend and increase with the hydrophobic nature of the surface, which is consistent with previous studies that have reported that the adsorption amount of BSA is dependent on the hydrophobicity of surfaces.[34,35] The initial short span of time magnified in Fig. 6a shows that the chemical properties of the surface influence not only the adsorption amount but also the adsorption kinetics, which contrasts with the adsorption behavior of linear flexible polymers. Fig. 6a also shows that despite the identical carboxylic acid (COOH) terminal group of MUA and MPA, the rates of adsorption BSA are vastly different. These differences in G(t) result from the combined influence of electrostatic forces and hydropho-bic interactions that are an intimate part of protein adsorption. Bovine serum albumin has an isoelectric point of pH ~5 and at the experimental pH of 7.5 the protein is negatively charged because of the presence of three domains that have a net charge of —10, — 8, and 0, respectively.[36- The SAMs of MPA and MUA ionize at pH>5 and can be considered to be largely deprotonated at a pH of 7.5.[37- In contrast, DT remains neutral in charge. The slow adsorption rate on deprotonated MUA SAMs is therefore attributable to the electrostatic repulsion between the surface and the charged domains of BSA molecules. Because of the shorter chain length of MPA, the self-assembled monolayer is more disordered than the monolayer formed from the longer MUA, where inter-molecular interactions between the alkanethiol chain lead to more conformational order. The disorder in the SAMs formed from MPA causes the exposure of methylene groups at the interface and this is manifested in the higher advancing contact angles. We believe that the gradual increase in the adsorption amount over time to Gmax that is close to the hydrophobic DT surface results from the exposure of the methylene groups, which allows hydro-phobic interaction between the hydrophobic residues on BSA and the SAMs even with the presence of electrostatic repulsion. The adsorption of BSA occurs when the molecule arrives in a preferred orientation, which overcomes the electrostatic repulsion from the surfaces and the adsorbed neighboring molecules but results in a slower rate of adsorption.
Aside from single-component SAMs as model homogeneous surfaces, several recent studies have used binary SAMs to investigate the specific and nonspecific binding of proteins and DNA.[38-40] In these studies, the surfaces were heterogeneous on a ”molecular level” with each component being uniformly dispersed in the ”well-mixed” binary SAMs. Fig. 6b illustrates schematically the differences between the well-mixed SAMs and the phase-separated SAMs that contain nanometer-sized heterogeneous sites distributed in a patchwise manner. The adsorption of BSA was studied on both types of heterogeneous surfaces to establish the role of the length-scale of surface heterogeneity. For comparison with the patchy heterogeneous surfaces (surface A), well-mixed SAMs (surface B) were prepared from DT and MUA, which are similar in chain length and lead to uniformly dispersed monolayers.[41,42] The advancing contact angles are 87° and 85° for surfaces A and B, respectively, which lie in between the values measured on the corresponding single-component surfaces.
Fig. 6 (a) Comparison of the kinetics of BSA adsorption on homogeneous surfaces of DT, MPA, and MUA with the adsorption on heterogeneous surfaces A and B. Arrow indicates time when washing with pure buffer solution was begun. The time scale at the beginning of adsorption has been magnified in the plot. (b) Schematic illustration of heterogeneous surfaces with a well-mixed and a nanometer scale, patch-wise distribution of heterogeneity.
Fig. 6a shows the kinetics of BSA adsorption on surfaces A and B. Surprisingly, the plateau value (Gmax) exceeds that on DT suggesting that adsorption can be enhanced by the presence of two well-mixed functional groups of CH3 and COO~ on the surface. The initial rate of adsorption on the well-mixed heterogeneous surfaces falls in between the rates on chemically homogeneous surfaces and once again illustrates the difference in adsorption behavior of a globular protein from that of a flexible polymer.
The morphology of the protein (Fig. 7) is one of many interesting aspects in the study of adsorption. Tapping mode AFM (TM-AFM) is a common scanning probe microscopy technique for visualization of biomaterials, cells, and proteins. Use of TM-AFM minimizes the force exerted on the surface during imaging, which avoids possible damage and distortion of the proteins. Fig. 7a shows that the adsorbed BSA on a DT surface has a dendrite-like structure of approximately 5 nm in height and 20 nm in width. A similar morphology was also observed by Ge and coworkers.[43] In past studies, it was found that a hydrophobic surface generally causes protein to unfold or denature upon adsorption and the unfolding can expose internal hydrophobic sites of the BSA. The preadsorbed BSA, in turn, provides preferential sites for adsorption of BSA from bulk solutions. Our experiments show that the dendrite-like structures form in an extremely short time (within seconds), which supports the hypothesis that proteins rapidly unfold upon adsorption. Kim and coworkers have proposed a different mechanism for a protein such as lysozyme, where clusters of proteins on the surface are believed to form by diffusion of adsorbed proteins on the surface.[44] Clearly, more experiments are needed to reach a definitive conclusion.
Fig. 7 Tapping mode AFM images of adsorbed BSA on SAMs of homogeneous surfaces of DT (a) and MPA (b) compared with the adsorbed BSA on well-mixed (c) and patch-wise (d) heterogeneous surfaces.
The adsorption morphology on surfaces formed by MPA and MUA (Fig. 7b) shows fluffy and relatively featureless morphology. Bovine serum albumin is a globular protein with approximate dimensions of 4 x 4 x 14 nm, which indicates that Gmax should be 2.5 mg/m2 for a closely packed protein monolayer in a side-on orientation and 8.8 mg/m2 in an end-on orientation. The adsorption amounts on MPA and MUA range from 0.3 to 0.9 mg/m2 (Fig. 6), which are much lower than these estimates of Gmax. The adsorbed BSA layers are therefore not continuous when one considers the rigidity of the BSA molecule resisting total unfolding on hydrophilic sur-faces.[45,46] However, the individual BSA molecules or clusters of BSA cannot be clearly imaged due to the effects of tip-broadening and the chemical interplay between the SAMs and the BSA layers in tip-surface forces.[47] In these AFM images, nevertheless, the deep boundaries of gold grains/terraces can be roughly mapped out, which suggests that the BSA is likely adsorbed in a distributed manner on the hydrophilic surfaces.
The AFM images of adsorbed BSA on each type of heterogeneous surfaces are shown in Fig. 7c and d. While the adsorption amounts on the well-mixed and patchy heterogeneous surfaces do not differ significantly, the morphology of adsorbed BSA layer is strikingly different although the SAMs possess similar surface energy (reflected in the comparable values of 8advancing). Comparison between Fig. 7a and Fig. 7c shows that when the surface changes from a hydrophobic (0a ~ 107), methyl-terminated interface to an interface where CH3 and COO— are well mixed (0a ~ 86), the morphology of adsorbed BSA changes from a dendrite-like structure to a networklike structure. Surprisingly, the TM-AFM image of the adsorbed BSA on patchy surfaces indicates breakdown of both dendrite and network structure. Instead, the layer of BSA shows few distinct features but appears as a continuous layer (Fig. 7d). When compared to the BSA layer on a MPA surface (Fig. 7b) where the morphology was featureless, the image in Fig. 7d shows the presence of a few holes/depression and occasional high spots. Results in Fig. 7 underscore the importance of the nanometer scale of the heterogeneity in surfaces. Recent studies [43,48,49- have reported adsorption of BSA on phase-separated Langmuir-Blodgett films where micrometer circular domains of a methyl o-functionalized organosilane were formed in a matrix of perfluryl o-functionalized organo-silanes. The morphology of the adsorbed BSA on each domain was found to be more or less similar to the corresponding single component LB film. Fig. 7c and d demonstrates that the adsorption characteristics of the BSA are strongly affected by the patchwise distribution of hydrophobic domains only when the surface heterogeneity is commensurate in size with the nanometer-sized, globular protein.
CONCLUSION
The experiments reported here and the agreement with theoretical studies demonstrate that, in addition to fundamental insights into adsorption on heterogeneous surfaces, we can now pursue experimental systems that exploit the interactions between lateral nanoscale morphology of a surface and an adsorbing polymer chain. For example, recent simulations have shown that a pattern of distributed charges on a surface can be recognized during the adsorption of an oppositely charged polyelectrolyte chain. Similarly others have predicted that the nanoscale morphology of a surface and the heterogeneous chemical interactions with a polymer chain can be used to direct segregation or patterning within adsorbed layers. The present strategy of using molecularly designed self-assembled surfaces for polymer adsorption enables the exploration of physical phenomena such as biomimetic recognition and segregation in polymer adsorption on heterogeneous surfaces and promises both scientific and technological advances.