Introduction
Low back pain is the second most common reason to seek a physician in the United States, third most common reason for a surgical procedure, and fifth most common cause for hospitalization (Andersson, 1997). The lifetime prevalence of low back pain is predicted to range from 60 to 80 percent (Hart, 1995; Van Tulder, 2002). The annual prevalence is estimated to be between 15 to 45 percent, with a point prevalence of 30 percent. Low back pain is the most common and most expensive cause of work-related disability in the United States (Atlas, 2000). Between 2002 and 2004, the estimated annual medical costs for all spine related conditions were approximately 193 billion dollars, with about 14 billion dollars in lost wages due to spine disorders (Bone and Joint Decade, 2005). It is a burden to both the individual and society, in terms psychosomatic impairment and socioeconomic impact. In fact, the presence of comorbidity adds to the burden and negatively impacts the patient’s functional status (Fanuele et al, 2000).
Fortunately, the majority of these patients recover within 3 months. With conservative care, it has been estimated that about 60 percent recover in 6 weeks, and 80 to 90 percent recover within 3 months (Andersson, 1999). Therefore, only a minority warrants further workup and care that can potentially include surgery. Non-surgical treatment consists of medications, cognitive training, physical therapy, and local injections. Some studies have shown that intensive, structural cognitive behavior therapy including encouragement and daily physical therapy can produce equivalent results compared to fusion in non-specific chronic low back pain (Brox et al, 2006; Fairbank et al, 2005).
In contrast, Fritzell et al (2001) showed that non-intensive, non-structural therapy yielded less optimal results than surgery. What comprises a structural or non-structural non-operative therapy regimen still remains unanswered. Additionally, only a few non-surgical interventional therapies have been shown to be effective, while prolotherapy, facet joint injection, intradiscal steroid injection, and percutaneous intradiscal radiofrequency thermocoagulation have been proven to be ineffective (Chou et al, 2009). The relative merits of non-surgical treatment for these conditions are beyond the scope of this topic.
The literature on surgical management for low back pain similarly elicits uncertainties due to non-specific diagnosis, but Glassman et al (2009) showed that with diagnostic specificity and stratification, the outcome of surgery depends on the underlying diagnosis. Functional improvement after surgery is not equal among diagnostic subgroups. Since surgery is a highly technical treatment modality, it is imperative to clearly define the pathological condition causing the symptoms rather than relying on simply a ‘diagnosis’ of low back pain, being a symptom rather than a clinical diagnosis or disease. Identifying a pathological condition allows surgeons to determine whether a surgical intervention can correct the problem and, in turn, improve the symptoms.
The aim of the topic is to discuss current options of surgical treatment of degenerative spinal disorders presenting with predominantly axial low back pain. Although the perception of the benefits of surgery for axial spine pain stemming from degenerative changes remains controversial, our aim is to discuss the current literature on the relative merits of surgery for selected patient groups.
Differential diagnosis
There are many causes of axial low back pain. Generally, the history and physical can play a paramount role in illuminating the etiology. Patients exhibiting constitutional signs such as fevers and chills can insinuate infectious etiologies, whereas weight loss, night sweats, and personal or family history of cancer can imply malignancy. Obviously, any recent trauma warrants imaging to rule out fracture.
Other causes of axial low back pain are divided into non-structural and structural entities relating to the vertebral column. Non-structural causes, sometimes referred to as nonspecific low back pain, are due to strain or sprain around the vertebral column, whereas structural reasons involve abnormalities within the vertebral column identified on imaging and can be considered as stable or unstable conditions. For this topic, we will focus on structural degenerative causes of axial low back pain. Stable conditions include degenerative disc disease (DDD) and facet arthropathy, while degenerative and isthmic spondylolistheses and degenerative scoliosis are more unstable conditions. Before delving into specific causes, general surgical outcomes for low back pain will be discussed.
Surgical outcomes for low back pain
However, there is a minority with persistent or increased pain, needing further workup and possibly surgery. There is a wealth of studies gauging the efficacy of surgical treatment for non-neurogenic axial low back pain. Systematic reviews of randomized controlled trials provide a strong level of evidence by setting inclusion and exclusion criteria when looking at the study methodologies, participants, interventions, and outcome measures. Yet, when it comes to comparing operative and non-operative results for axial low back pain, there can be conflicting results due to the lack of specificity in describing the cause of back pain since the outcomes from surgery differ between diagnostic subgroups. The shortcoming of these systematic reviews is their broad categorization of causes of back pain by combining the aforementioned causes as just degenerative disease. Also, differences in patient inclusion criteria, fusion technique, non-surgical treatment, and outcome measures make it hard to draw conclusions.
The general view of surgery for axial low back pain is met with skepticism. Mirza and Deyo (2007) systematically reviewed surgical compared to non-surgical treatment of discogenic back pain and concluded that surgery may not be more efficacious than structured cognitive behavior therapy. However, careful analysis of this study shows that the specific diagnostic indications for surgery are poorly defined. The population in this review was deemed to have low back pain for 12 months or longer without a specific diagnosis, and there was no established way of diagnosing discogenic pain. Furthermore, there was no uniform surgical technique, but rather an inclusion of a myriad of interventions, including one group that received flexible stabilization without fusion. Thus, due to its limitations, this study fails to accurately measure the effectiveness of surgery for axial low back pain.
Similarly, Chou et al (2009) systematically reviewed the benefits and harms of surgery for non-radicular back pain compared with non-operative measurements. They looked at different trials addressing fusion for chronic back pain mostly due to DDD, but not exclusively limited to DDD. Their conclusion is that fusion is no better than intensive rehabilitation with cognitive behavior emphasis, but slightly better than non-intensive non-surgical therapy. Again, a fault of the analysis is the inability to specify the specific causes of low back pain. It also combined various surgical techniques for undefined chronic back pain. This leads to the presumption that there is no role for surgery in axial low back pain when, in fact, the success of surgery depends on specific causes.
Glassman et al (2009) demonstrated that it is possible to stratify by specific diagnostic indication when looking at lumbar fusion for different diagnostic subgroups. In contrast to prior systematic reviews, they prospectively collected clinical outcome measures and reported on the impact of lumbar posterolateral fusion on different subgroups. Outcome measures such as the Oswestry Disability Index (ODI), Short Form-36 (SF-36), and numeric rating scales for back pain were used. Their findings showed that outcomes scores were not equal among diagnostic subgroups. In fact, the diagnostic subgroup that demonstrated the most significant improvement in ODI scores were the patients with spondylolisthesis, followed in decreasing order by scoliosis, disc pathology (i.e. DDD), postdiscectomy revisions, stenosis, and adjacent level degeneration. This was based on the percentage of patients in each subgroup to reach minimum clinically important difference, or an improvement of at least 10 points in ODI scores, during a 2-year follow-up.
Carreon et al (2008) provided another study looking at outcome measures while stratifying subgroups. They also used ODI and SF-36 to compare surgery with no surgery. The mean improvement in ODI in the surgical group was higher than the non-surgical group. Within this group, patients with spondylolisthesis had the greatest improvement, followed by those with DDD, then patients with chronic non-structural non-specific low back pain. This implies that non-specific etiologies, as displayed by the chronic low back pain group, can portend less success with surgery. On the other hand, more specific structural etiologies such as spondylolisthesis can benefit from surgical interventions.
Stable degenerative conditions
Degenerative disc disease
Degenerative disc disease (DDD) (Figure 1) stems from structural changes of the disc, which eventually leads to disc space narrowing, endplate osteophyte formation and sclerosis, and gas formation within the disc space. This is in contrast with internal disc disruption (IDD), which displays only abnormal discal properties without loss of disc height or endplate changes. The exact pathophysiology of DDD is not fully understood, and so its natural history is still unknown. Kirkaldy-Willis et al (1978) proposed a pathoetiology for this condition. They viewed each level of the lumbar vertebra as a three-joint complex consisting of a disc and two posterior joints. Stresses to one joint can affect the others. The process of degenerative disc disease starts with internal disruption, followed by resorption of the disc and endplate changes. With a degenerative disc and therefore more strain on the posterior facet joints, this eventually leads to advanced facet arthropathy and spondylosis. With minor repeated trauma, the degenerative interaction between the three-joint complex leads to more stresses on the adjacent levels, thus, potentially leading to a multilevel degenerative spine. In 10 to 39 percent of chronic low back pain cases, the intervertebral disc is suggested to be the source of pain (Schwarzer et al, 1995; Manchikanti et al, 2001). Despite this, it is still controversial as to how much DDD correlates to low back pain. As a result, there is a debate regarding its treatment. Clinical examination may show midline spinal tenderness and reduced range of motion, typically in flexion.
Fig. 1. Degenerative disc disease
Lateral radiograph of the lumbosacral spine depicting marked loss of disc spaces from L2 to S1. There are anterior osteophytes and endplate subchondral sclerosis.
Non-surgical management may include such modalities as physical therapy, medications, and interventional injection treatments. According to Cochrane reviews, long-term bedrest and back braces are not recommended (Hagen et al, 2004; van Duijvenbode et al, 2008). Interventional modalities such as epidural injections and intervertebral disc injections and manipulation have yet to be proven effective by randomized controlled trials. Acetaminophen and non-steroidal anti-inflammatory drugs are the most commonly used medications. Non-surgical options for this condition are discussed elsewhere in this topic.
Surgical options for DDD may include fusion or motion-preservation strategies, such as artificial disc replacement. There have been only a handful of high quality randomized controlled studies assessing the effectiveness of surgery for DDD. Because the diagnosis of DDD is still controversial, these studies are still non-specific in terms of diagnostic categorization. In a meta-analysis of randomized trials comparing fusion to conservative treatment for DDD, surgery led to improved functional scores compared to non-surgical treatment (Ibrahim et al, 2008). However, the difference in functional improvement was not statistically significant. Meanwhile, disc replacement can be an option for isolated disc pathology, without arthrosis of the facet joints or spinal instability. There has been little research comparing total disc replacement versus conservative care, however, some studies have shown non-inferiority of disc replacement to fusion (Blumenthal et al, 2005; Zigler et al, 2007). Surgical options for these conditions will be discussed in more detail later in the topic.
Facet arthropathy
Along the spectrum of degenerative changes, facet arthrosis results from increased load to the posterior elements due to abnormal load sharing from disc derangement and repetitive minor trauma over time as discussed by Kirkaldy-Willis. Similar to other synovial joints, like the hip, knee, and shoulder, degenerative arthropathy of the facet joints can lead to joint space narrowing, osteophyte and cyst formation, joint effusions, and mechanical pain. Facetogenic pain is typically worse with extension and may be relieved with rest. Patients often get relief of their back pain with leaning on a walker or shopping cart. It is the same degenerative process that may be implicated in spinal stenosis whereby the osteophyte formation, cyst formation, disc bulging, and redundancy of the ligamentum flavum from disc height loss all cause encroachment on the neural elements.
Conservative management, in addition to physical therapy and medications, consists of intra-articular facet injection and medial branch block. Medial branches of the dorsal rami are usually blocked at the junction between the superior articular facet and transverse process. In patients who respond to medial branch blocks, medial branch neurotomy via radiofrequency ablation presents as an effective non-surgical treatment for facet arthropathy (Dreyfuss et al, 2000). Once the diagnosis is confirmed with positive blocks, but the pain still recurs, then surgical options include posterior fusion and facet replacement (discussed later in the topic).
Unstable degenerative conditions
Degenerative scoliosis
Introduction
Scoliosis is defined as an abnormal curvature of the spine of more than 10 degrees (Figure 2). In adults, scoliosis can be a result of untreated scoliosis that existed before skeletal maturity or can develop after skeletal maturity, otherwise known as de novo scoliosis. An example of de novo scoliosis is degenerative scoliosis, which is caused by a continuum of degenerative changes as described in DDD and facet arthropathy, leading to central canal and foraminal narrowing. Adult scoliosis has detrimental effects on the health status of the affected person. Berven et al (2003) illustrated that compared with control subjects, adults with scoliosis have more pain, lower self-image, less functional capacity, and lower mental health scores. They also concluded that radiographic parameters do not necessarily correlate well with the patient’s self-assessment of health status.
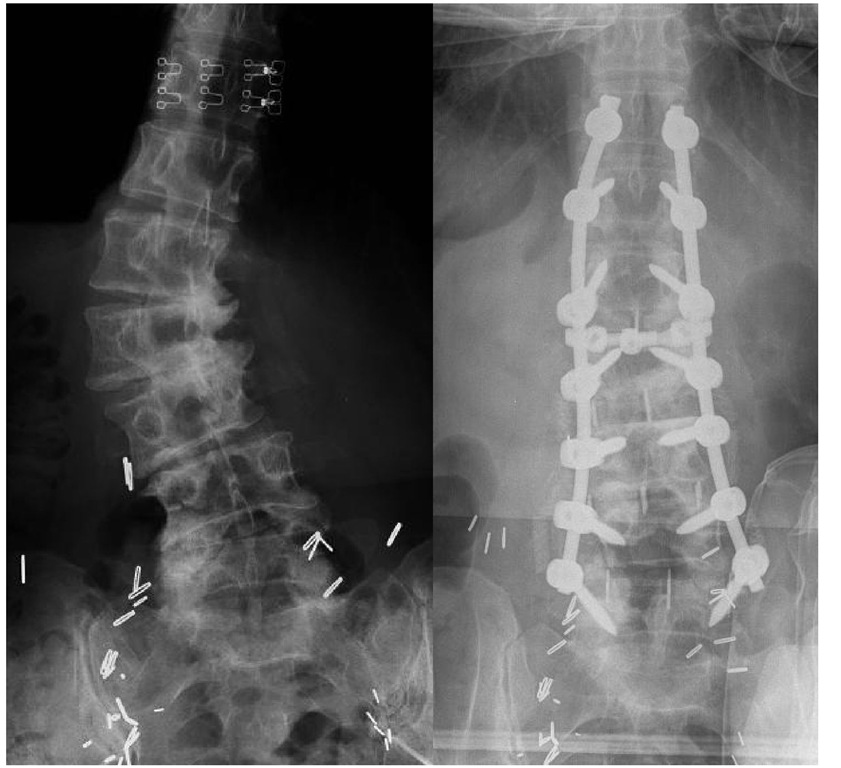
Fig. 2. Degenerative Scoliosis
Figure 2a shows an anteroposterior (AP) radiograph of the lumbosacral spine showing degenerative lumbar scoliosis with the apex at the L2-3 level. Notice the degenerative disc disease from L1-L5 and the rotational deformities of the vertebral bodies based on the asymmetric pedicles. Figure 2b shows postoperative AP radiograph with posterior instrumentation and lateral interbody fusions for deformity correction. Note that the lumbar spine now sits in line with the mid-sacrum.
Natural history
Progression of the curve is common, but the extent of it is unknown so continual observation is important. Chin et al (2009) demonstrated that women older than 69 years of age with levoscoliosis and lateral listhesis of more than 5mm might progress rapidly. Deviren et al (2002) showed that increasing age and curve magnitude correlate to decreased curve flexibility. Also, the degree of axial back pain associates with increasing age. Pritchett and Bortel (1993) studied 200 patients older than 50 years of age with adult scoliosis and found that certain factors might predict curve progression. In general, those with significant curves with rotation can progress rapidly.
Diagnostic imaging
Full-length standing radiographs are required to fully assess the overall spinal balance. Cobb angles are determined for the structural curve, which is usually the largest curve, and any compensatory curve. Coronal balance is determined with a plumb line from the middle of C7 vertebral body on posteroanterior (PA) radiograph. This should intersect the midsacrum. Sagittal balance is measured using a plumb line from the center of C7 on the lateral radiograph. This line should typically fall within 2 to 4 centimeters from the the posterior margin of the lumbosacral disc. If it falls anterior to the posterior margin of the disc then positive sagittal imbalance is present, while negative sagittal imbalance is when the line falls posterior to the disc. Global sagittal imbalance has the most significant impact on pain and function compared to other radiographic parameters (Glassman et al, 2005). The evaluation of the flexibility of the main structural curve and its compensatory curve is done through various specialized radiographs. Obliquities such as pelvic tilt and shoulder asymmetry should also be noted. Flexibility and obliquity assessment dictate which level to fuse and instrument when performing surgery. Any radicular or neurogenic pain should merit obtaining magnetic resonance imaging (MRI).