PET/CT
FDG-PET/CT studies in oncology utilize labeled glucose with the tracer fluorine-18 (F-18) fluorodeoxyglucose (FDG), in combination with MDCT, to provide the benefits of metabolic and anatomic imaging in a single study. FDG is a glucose analog that is taken up by glucose-using cells and phosphorylated by hexokinase in the mitochondria. Increased tumor [18F]-FDG uptake is depicted by positron emission tomography (PET), although proliferative activity is also broadly related to the number of viable tumor cells. When glucose is metabolized, the tissue incorporates the radioactive isomer, increasing local particle emission and producing a high intensity signal on PET/CT images (Young, et al., 1999).
PET-CT is a sensitive method for assessment of bone and bone marrow metastases, as well as vertebral and extravertebral skeletal masses. It is sensitive for osteolytic and osteoblastic metastasis and depicts early malignant bone-marrow infiltration (Kruger, et al., 2009). It provides precise localization and is sensitive to accompanying soft tissue metastases in the lung, liver, lymphatic system, and elsewhere (Metser, et al., 2004; Nguyen, et al., 2007). Low resolution PET scans are combined with the high resolution of thin slice CT, for exquisite depiction of even small metastases in early stages. PET/CT has higher sensitivity and specificity compared to bone scintigraphy or CT in the detection of skeletal metastases (Kruger, et al., 2009), however the rate of false-positive findings that require follow-up imaging with other modalities is also higher (Kuo & Cheng, 2005).
Cord compression
Discussions of metastatic epidural cord compression usually focus on the thoracic spine. Although the spinal cord usually ends in the lumbar spine between L1 and L2 in adults, up to 20% of metastatic cord compression occurs at these levels; thus lumbar cord compression must also be discussed.
Metastatic spinal cord compression (SCC) occurs in 5-10% of patients with cancer (Bach, et al., 1990). It is a true emergency, because delay in diagnosis and treatment may result in permanent neurological impairment. SCC is caused by direct compression of metastasis or primary tumors invading the vertebral bodies, breaking through the cortex, and compressing the vertebral canal and nerve roots (Bilsky, et al., 2000). The most commonly affected site is the thoracic spine, which is also the area in which the canal is the tightest, leaving little space for movement of the cord. Breast cancer is the most frequent primary malignancy associated with SCC, followed by lung, prostate, and renal cancers (Byrne, 1992). However, SCC is also common in patients with hematological tumors such as lymphoma and myeloma. SCC is the initial manifestation of a metastatic spread of a tumor in approximately 20% of cancer patients; in patients with lung cancer the rate of SCC at first presentation climbs to 30% (Chahal, et al., 2003). In most of the cases of SCC, back pain has been present for several months before detection, but was ignored by the patient and caretakers (Byrne, 1992).
The most important factor in determining the post-treatment outcome is pretreatment ambulatory function of the patient. Late presentation with neurological deficits, including bowel or bladder dysfunction, is often associated with irreversible paraplegia. In recent series, 74-100% of patients who were ambulatory before surgery retained the ability to walk after decompression, and 57-82% of nonambulatory patients regained ambulation (Bilsky, et al., 2009). However ambulation at surgery remains a key determinant of outcome, therefore, urgent investigations must be performed to facilitate treatment before function is lost.
Treatment
Medical management of pain
Basic pain control is achieved by the use of narcotics and non-narcotic analgesics. In patients with back pain due to metastases, pain control is one of the most important goals in management, as the life expectancy of the patients is limited (Padalkar & Tow, 2011). Sadly, despite this concept, most patients are undertreated for pain, which causes a significant reduction in quality of life (Cleeland, 2006). Two types of pain needed to be addressed when dealing with medical management of the metastatic patients, the constant pain of the metastasis and an acute episode of sharp pain, known as "breakthrough pain" (Lipton, 2011).
The first line of treatment in patients with mild low back pain is the use of nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen. These agents may provide adequate pain relief. There have been nine trials indicating that the use of NSAIDS provides pain relief in bone metastasis, including one showing that NSAIDS (specifically COX-2 inhibitors) assist in tumor control as well (Smith, 2011). Acetaminophen is preferred in patients with thrombocytopenia, renal dysfunction, those receiving nephrotoxic agents, and those at risk for gastrointestinal bleeding. In patients with liver dysfunction, NSAIDs are preferred for mild pain (Hitron & Adams, 2009)
In patients who need the next level of treatment for their low back pain, opioid therapy should be added. Common protocols begin with low doses of immediate-release short-acting agents (i.e. morphine), with reassessment for the level of effect every 1 to 2 hours. After 24 hours of pain control on a short-acting regimen, patients should be converted to a long-acting agent such as sustained-release morphine, oxycodone, fentanyl, or methadone for basal control. Patients who are opioid tolerant should begin with higher doses of short-acting agents, with the higher dose compensating for shorter duration of effect. If a patient is already using a long-acting product, this should be continued. A bowel regimen with a stimulant plus stool softener should be initiated to prevent constipation from opioid use. Care must be taken to avoid misuse and abuse of opioids, even in cancer patients (Manchikanti, et al., 2010).
Adjunct agents such as anticonvulsants (e.g., gabapentin, lamotrigine, topiramate), tricyclic antidepressants (e.g., amitriptyline, imipramine, desipramine, nortriptyline), venlafaxine, duloxetine, or topical analgesics (e.g., lidocaine or capsaicin) may also help to reduce neuropathic pain caused by nerve compression (Hitron & Adams, 2009).
In cases of breakthough pain, oral transmucosal fentanyl and transdermal patches have been shown to be an effective treatment for pain including sharp pain episodes, yielding a more rapid onset of relief. In cases where patients developed tolerance towards opioid narcotics, the use of anesthetics, such as ketamine can provide adequate pain relief (Chazan, et al., 2008).
The use of cannabinoids as an alternative or to augment narcotics has become popular, primarily for light-to-intermediate pain. Three cannabinoids, all a combination of the delta-9-tetra-hydrocannabinol and cannabidiol cannabinoids from the Cannabis sativa plant, are available on the market. Sativex, a cannabinoid medication, has recently received approval as an adjuvant medication for the treatment of cancer pain in North America. It is a sublingual (mouth) spray that can be titrated up to the most effective dose (Bonneau, 2008). Other forms of cannabinoids treatment include smoking marijuana, or ingestion of oils or even cookies with insertion of active cannabinoid components. In our experience, "cannabis cookies" have provided relief or reduction in chronic pain levels for some cancer patients. The use of cannabinoids is indicated in cancer pain with a neuropathic component that is not adequately controlled with opioids. Cannabinoids are not suitable as a single medication in spine metastasis.
Steroids
Steroids are commonly prescribed in patients with metastatic bone disease. Steroids reduce edema and have been shown to reduce the size of metastases from tumors of hematological origin, and occasionally breast cancer (Cole & Patchell, 2008). They have also been shown to have a rapid analgesic effect (Bonneau, 2008). However, they are associated with side effects, such as wound dehiscence, gastric ulcers, rectal bleeding, psychosis, and diabetes mellitus, and may increase susceptibility to infections (Shih & Jackson, 2007).
An early study (Greenberg, et al., 1980) demonstrated quick and significant pain reduction in patients treated with steroids. A subsequent randomized trial (Vecht, et al., 1989) compared the effect of 10 mg IV dexamethasone versus 100 mg IV followed by 16 mg daily orally and found no differences between the conventional and high-dose group on pain, ambulation, or bladder function. Both the conventional and high-dose regimens provided significant pain relief.
A randomized, controlled trial (Sorensen, et al., 1994) studied the administration of 96 mg of IV dexamethasone followed by 96 mg orally for 3 days and then tapered in 10 days, with subsequent radiotherapy, versus radiotherapy as a single modality in the treatment of SCC. Steroid treatment provided a statistically significant improvement in ambulation at 3 to 6 months, albeit with increased side effects, including psychoses and gastric ulcers requiring surgery. The effect of dexamethasone on pain reduction was not addressed.
Currently there are no absolute guidelines for steroid treatment in patients with back pain due to spinal metastases. The decision of whether to treat, as well as steroid dose are determined by the treating physician. In cases of SCC, steroid treatment is given as an adjuvant to surgery or to chemo- or radiotherapy, and not as sole treatment modality.
Bisphosphonates
Bisphosphonates are pyrophosphate analogues. The bisphosphonates strongly bind hydroxyapatite crystals in bone, preventing the creation of the ruffle border of the osteoclasts that prevent bone resorption. In addition, bisphosphonates induce osteoclast apoptosis (Li, et al., 2011).
The rationale for using drugs such as bisphosphonates in patients with metastatic cancer is that osteoclasts, as the mediators of bone absorption, are often activated by the tumors and thus allow metastatic invasion into the medulla of the vertebrae. Preventing such activity may reduce metastatic invasion and spread (Orita, et al., 2011). In addition, by inhibiting bone resorption, a secondary correction of hypercalcaemia and hypercalciuria will be achieved (Woodward & Coleman, 2010).
Most importantly, the use of bisphosphonates reduces pain and the occurrence of fracture, as well as the development of new osteolytic lesions and, as a consequence, improves patients’ quality of life (Fleisch, 1991). Compounds that are commercially available for use in tumor-induced bone disease are, in order of increasing potency, etidronate, clodronate, pamidronate, and alendronate.
Van Holten-Verzantvoort et al found a significant reduction in morbidity from bone metastases in pamidronate-treated breast cancer patients, with a 30-50% reduction of pain and lower rate of new pathological fractures (van Holten-Verzantvoort, et al., 1991). Thurlimann et al showed that pain relief was achieved in about 30% of patients who received pamidronate every 4 weeks (Thurlimann, et al., 1994).
Side effects from bisphosphonates, including transient low grade fever, nausea, myalgia, gastrointestinal side-effects, bone pain, and mild infusion-site reactions, are usually minimal. More rarely, osteonecrosis of the jaw and long bone "frozen bone" fractures due to long term use have been reported (Kim, et al., 2011; Mercadante, 1997).
A meta-analysis (Fulfaro, et al., 1998) concluded that bisphosphonates, and in particular IV pamidronate, are an important therapeutic tool in association with other therapeutic modalities for the treatment of metastatic bone disease with marked osteolysis, such as multiple myeloma and breast cancer. The authors did not evaluate the impact on back pain, but looked at the general effect of bisphosphonates on bone metastases.
Chemotherapy
As is the case with metastatic disease in other locations, the long-term control of spine metastases entails systemic chemotherapy. The type of treatment and its duration largely depend on the tumor histology and specific tumor receptors. The full treatment plan should be determined by a team of oncologists, spine surgeons, and radiotherapists, balancing the different modalities of treatment with the side effects and complications.
Hoy et al showed that many patients experience pain relief with chemotherapy, even in cases where there is no objective tumor response (Hoy, 1989). Although palliative, the exact benefit of chemotherapy is difficult to measure. Potential indications for palliative chemotherapy include symptoms due to metastatic disease, hypercalcemia, bone marrow infiltration, and clinical conditions known to be associated with bone or spine metastases. As patients are mainly treated with a combination of therapies, the exact effect of the response in the bone is more difficult to assess (Lote, et al., 1986).
In tumors that react quickly to chemotherapy, as in testicular cancers, lymphoma, or myeloma, administration of chemotherapy can have a pronounced effect in reducing back pain (Samoladas, et al., 2008). In tumors where the response to chemotherapy is slower, as in pancreatic cancer, pain reduction parallels the decrease in pressure exerted on spinal structures as the tumor slowly diminishes in size (Takuma, et al., 2006). However, in very advanced cancer patients there is little justification for using chemotherapy because of its toxicity (Bruera, 1993).
Hormonal therapy, based on the principle of depriving tumor cells of the growth stimulus induced by hormones that change the hormonal environment, may be beneficial in breast, prostate, and endometrial cancers (Wood, 1993). This treatment has a lower toxicity profile compared to chemotherapy, and should be the first therapeutic modality if possible. Dearnaley showed that hormonal therapy provides sustained symptom relief in patients with widespread painful bone metastases from prostatic cancer (Dearnaley, 1994). Cresenda et al showed that the combination of hormonal therapy with radiation for spinal metastasis reduces pain in 77% of patients (Cereceda, et al., 2003). There are no data regarding the impact on pain from the isolated use of hormonal therapy in patients with spinal metastasis.
Radiation-based treatment
Radiotherapy has served as a cornerstone for the treatment of bone metastases since it was introduced in the 1950s. Its benefits include reduction of pain and neurological complications arising from spinal cord compression. Radiation-based treatment is undergoing a revolution, with new stereotactic techniques for single-shot or fractionated treatment showing good results. However the mainstay of radiation-based treatment is external beam radiation therapy (EBRT) (Cole & Patchell, 2008; Harel & Angelov, 2010).
External beam radiation therapy
The exact pathophysiology of tumor response to radiotherapy is not fully understood, however EBRT can lead to rapid reduction in the tumor size, sometimes within 24 hours, decreasing local periosteal nerve stimulation. EBRT is typically given from a posterior field to the affected vertebra(e), with radiation delivered to a treatment area including one vertebral body above and one below to compensate for daily variations in patient setup.
A meta-analysis (Agarawal, et al., 2006), reported that over 40% of patients with bone metastases treated with radiation achieved at least a 50% reduction in pain, however fewer than 30% experienced complete pain relief at 1 month.
In cases of SCC, when surgery is not performed, the use of radiation combined with steroids has been shown to provide improved pain management and improved outcome when compared to radiation alone (Loblaw, et al., 2005). In cases were surgery is planned, the use of preoperative EBRT is associated with an increase in local wound-related complications (Bilsky, et al., 2000; Itshayek, et al., 2010; Itshayek, et al., In press).
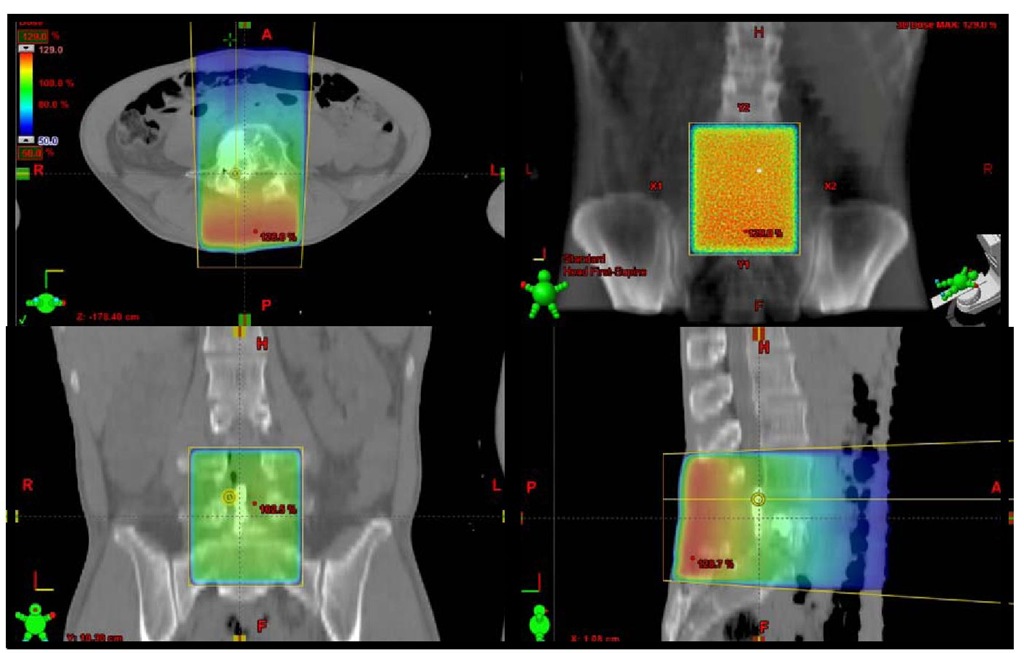
Numerous EBRT protocols may be employed in the management of painful bone metastasis, including fractionated and single-fraction regimens. The dose ranges from 8 Gy delivered in a single fraction, up to a cumulative dose of 30-40 Gy administered over 3-4 weeks (Gerrard & Franks, 2004; Harel & Angelov, 2010). In a retrospective evaluation of treatment, comparing a single 8 Gy dose with several hypofractionated regimens in 1304 patients with spine metastases, there was no statistical difference for the various protocols in terms of patient survival, pain control, or side effects, however there were higher recurrence rates in patients treated with a lower cumulative dose (Rades, et al., 2005).
Fig. 3. Treatment plan showing a map of the radiation fields for external beam radiation therapy. A 42-year-old male presented with low back pain and cauda equina syndrome due to metastatic synovial sarcoma to L4 (Fig. 2). Following laminectomy and resection, he suffered persistent axial back pain and proceeded to vertebral augmentation (Fig. 5). After augmentation ERBT was administered for tumor control.
Stereotactic radiosurgery
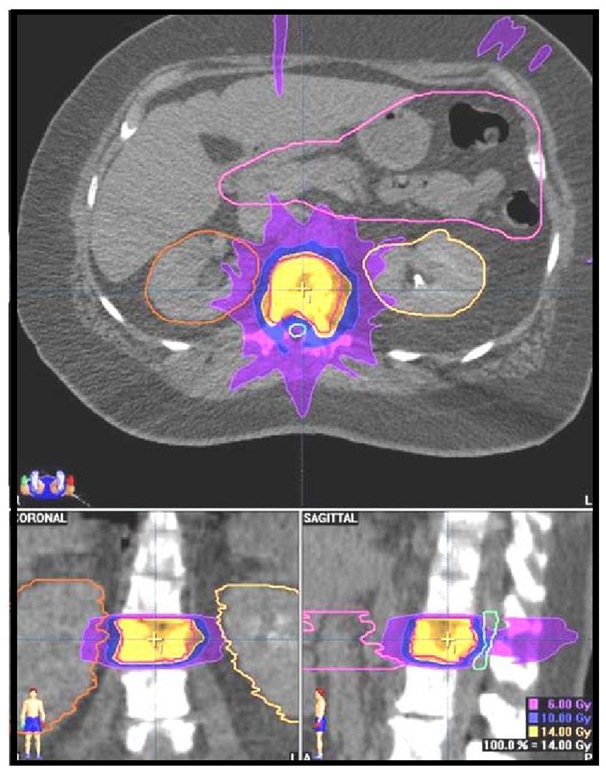
Stereotactic radiosurgery (SRS) is a new modality that is used in patients with spinal metastases in some centers. The use of stereotaxy enables deployment of high doses of radiation to inactivate or eradicate defined targets in the head or spine, even tumor found in close proximity to the spinal cord or other critical structures. SRS uses sophisticated computer algorithms to divide the lethal doses of radiation into multiple projections, focusing a very high dose in the target zone with rapid dose fall off, and thus minimal collateral damage to adjacent tissues (Moulding, et al., 2010). These systems are extremely accurate, with less than 1 mm deviation from the target.
The optimal SRS protocol is highly individual, and should take into account the tumor’s size, its pathological composure, and distance from the cord, as well as other radiosensitive organs. Data accumulating from an increasing number of studies suggests that SRS is a relatively safe, quick method to control spine metastases (Bilsky, et al., 2009). It provides excellent pain relief in most patients. In one study, significant pain relief was reported 6 months after treatment in 91% of patients treated with the Cyberknife (Accuray, Sunnyvale CA, USA) (Chang, et al., 2009). Another study reported improved pain control and improved quality of life 1 year after treatment (Degen, et al., 2005).
A possible side affect of spinal SRS is compression fracture, which may be attributed to rapid tumor lysis after radiation. These fractures can be managed conservatively or with pre-radiation vertebral augmentation (Gerszten, et al., 2005).
Fig. 4. Treatment plan showing radiation fields for a stereotactic radiosurgery procedure.
A 65-year-old male with renal cell carcinoma that was metastatic to L2 that was treated with single-shot SRS.
Surgery
Tumor decompression and instrumental stabilization
The surgical treatment of spinal metastases has evolved significantly over the last 20 years, with mounting evidence of the value of surgery in the treatment of metastasis (Bilsky, et al., 2009; Cole & Patchell, 2008; Harel & Angelov, 2010; Tomita, et al., 1997). Surgery was once limited to posterior laminectomy for the management of neurological decompression in SCC {Roy-Camille, 1990 #219}. With this philosophy, outcomes were mixed since most metastases are anterior to the cord and thus not directly accessible with posterior surgical approaches. With advances in technique, new surgical approaches, and spinal instrumentation, patient outcomes have significantly improved, due to a combination of anterior tumor decompression and instrumental stabilization. The surgical approach is tailored, depending on the anatomic locations of metastases, aiming if possible to reach en block total resection of the tumor, the best possible outcome for the patient {Tomita, 2006 #220}. Tumors anterior to the spinal cord are usually approached with a purely anterior approach, especially in the cervical spine, or through a posterolateral or lateral approach in the thoracic and lumbar spine. These approaches allow for anterior decompression with anterior column support, as well as posterior instrumented fusion for improved stability. Although laminectomy can help to increase the diameter of the spinal canal at the affected levels, it does not achieve immediate cord decompression and can lead to instability {Black, 1996 #213; Camins, 2004 #221}.
As in most procedures for the management of metastatic disease, the aim is not to provide a cure but rather to relieve pain, stabilize the spine, prevent neurological deterioration, and at times provide a pathological diagnosis (Choi, et al., 2010). In select cases, complete resection of a spinal metastasis can be performed, which can provide the best opportunity for long-term local control and palliation in patients with isolated metastases or radioresistant tumors (Gallo & Donington, 2007). In cases where total resection cannot be achieved, stabilization and decompression provide a window of opportunity for other treatment modalities to take their course without jeopardizing the patients’ health. In two large cohorts of patients, surgery reduced pain, and improved physical function and quality of life in patients with symptomatic vertebral metastases at 12 month follow up and beyond, with an acceptable complication rate (Falicov, et al., 2006; Quan, et al., 2011).
In cases of SCC, treatment should include corticosteroids, surgery, and radiotherapy along with aggressive systemic chemotherapy. Surgical decompression followed by radiotherapy was shown to be superior to radiotherapy alone in a study that was halted early after interim analysis demonstrated superior results in the surgery and radiotherapy group compared to the radiotherapy alone group (Patchell, et al., 2005). Patients treated with surgery plus radiotherapy had a median survival of 126 days with ambulation for 122 days, compared to a median 100 days survival and only 13 days ambulation in patients treated with EBRT alone. These findings were confirmed in a retrospective study conducted at our center (unpublished results). Patients who were operated had longer periods of ambulation relative to patients treated solely by radiotherapy. They also required lower doses and lighter pain medications.
Vertebral augmentation using vertbroplasty and kyphoplasty
Over the past two decades, percutaneous cement augmentation techniques have been developed for the treatment of spinal metastases and fractures. The two most common techniques are vertebroplasty and kyphoplasty. Vertebroplasty is a technique in which cement is injected into the vertebral body; kyphoplasty uses a balloon to create a void in the vertebrae into which the cement is injected. In a recent review comparing these techniques, pain scores for both the vertebroplasty and the kyphoplasty groups decreased significantly from preoperative values at 6-month follow-up (Liu, et al., 2010). The National Institute for Health and Clinical Excellence (NICE) guidelines (N.H.S., 2003; 2006) state that vertebroplasty or kyphoplasty should be considered for patients with vertebral metastases when their pain is refractory to analgesia, or when there is evidence of vertebral body collapse, in cases where is no evidence of cord compression or spinal instability.
Fig. 5. Vertebral augmentation. A 47-year-old male presented with persistent axial low back pain that was refractory to medical management following laminectomy and resection of an epidural mass from an L4 metastasis of a synovial sarcoma (see Figs. 2, 3). The patient underwent vertebral augmentation. (A) An OsseoFix implant (Alphatech Spine, Carsbad CA, USA) is inserted in an unexpanded configuration into the L4 vertebral body. (B) Once proper placement is confirmed on fluoroscopy, the implant is expanded. (C) The implant is filled with PMMA bone cement. Following augmentation the patient is pain free.
Conclusions
Back pain is one of the most prevalent medical problems, and is usually benign. However, in patients with a history of cancer, one must take extreme precautions to make sure that a metastatic spread of the tumor is not missed. Metastases may present as mechanical pain, radicular pain or weakness, or with myelopathy. Pain is usually unremitting and progressive.
MRI is the gold standard for image-based diagnosis, with excellent depiction of soft tissue, including the spinal cord and nerve roots; CT provides optimal visualization of bony structures.
Pain is the most common and earliest complaint, and sadly, it is undertreated in most patients. Most studies are not focused on pain control, but rather on the neurological status of the patient, while pain control is a secondary objective.
Management of back pain in metastatic patients may involve a combination of pain medications, chemotherapy, radiation-based treatment, and surgery. Patient management thus requires the combined efforts of a multidisciplinary team, including oncologists, radiation oncologists, and spine surgeons. Treatment should be tailored to the needs of specific patients, based on their prognosis, neurological status, age, and primary pathology.