The Zoraptera (“zorapterans,” “angel insects,” or less appropriately “soil lice”) are one of the smallest insect orders with only JL 35 living species and 10 more known from fossils. Zorapterans are rarely observed; indeed many entomologists have never seen them alive. There is a single family (Zorotypidae) and only two genera, Zorotypus and the fossil genus Xenozorotypus1 from the middle Cretaceous. Some authors have at times attempted to divide living species of Zorotypus into multiple genera or subgenera, but none have successfully characterized natural (i.e., monophyletic) groups. Owing to the paucity of species and the great morphological homogeneity of the order, the recognition of numerous living genera is unwarranted.
Zorapterans superficially resemble termites (Isoptera) and bark lice (Psocoptera) (Fig. 1A, 1B, and 1D) and they have at times been classified near or within these groups. Individuals are typically less than 4 mm in body length (excluding their antennae) and live in
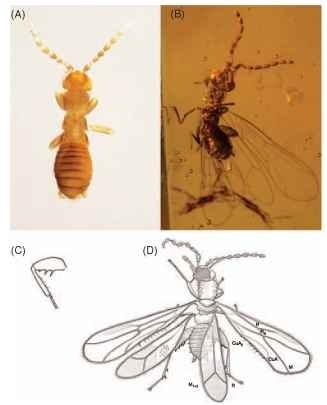
FIGURE 1 (A) Adult female of Zorotypus huxleyi (most legs curled under body), blind-apterous morph. (B) Alate female of Z. goeleti1 in Miocene amber from the Dominican Republic. (C) Hind leg of Z. huxleyi depicting femoral and tibial spines. (D) Alate of Z. nascimbenei1 in Cretaceous amber from Myanmar, with wing veins labeled.
small colonies. The most readily noticeable traits of the order are the expanded hind femora that bear stout spines on their ventral surface (Fig. 1C), the presence of only two tarsomeres (the basal most of which is greatly reduced in size; Fig. 1C) , and paddle-shaped wings with reduced venation (Fig. 1B and 1D) that are shed by an ill-defined basal fracture.
ORIGIN AND EVOLUTION
Although zorapterans are relatively minute both in size and in species diversity, they have weighed heavily on the minds of evolutionary biologists. The order has been at one time or another classified in a bewildering array of phylogenetic positions, at times being considered relatives of the termites, roaches (Blattaria), mantises (Mantodea), webspinners (Embiidina), or bark lice; sister to Paraneoptera; or even allied to the Holometabola. The most well supported hypothesis is that they are polyneopterans and the living sister to the webspinners, both orders sharing a unique development of the musculature in the hind legs, among several other important traits. The Zoraptera-Embiidina split likely occurred during the middle Mesozoic, perhaps as long ago as the early-middle Jurassic, while the ancestor of both groups diverged from crown-group stoneflies (Plecoptera) perhaps sometime in the Permian or Triassic. The Zoraptera are clearly ancient as evidenced by the presence of extinct species of the living genus Zorotypus in middle Cretaceous amber (e.g., Fig. 1D). These extinct Mesozoic species already show the development of the dual-morphs, with blind-apterous and eyed-winged individuals. Only the extinct genus Xenozorotypus1 shows a departure in morphology (albeit not nearly as dramatically as the anatomical diversity found in other orders) from all other zorapterans by the primitive retention of additional wing veins and the structure of spines on the hind tibia.
DISTRIBUTION
Species principally occur in tropical habitats throughout the world although four species occur north of the Tropic of Cancer (i.e., north of 23.5°N): two in North America and two in Tibet. Similarly, the known fossil zorapterans have all been discovered in ambers that were formed in warm-tropical paleoclimates. The northernmost areas of distribution for Z. hubbardi in North America are likely a result of human activity. Most northerly colonies are formed in sawdust piles (rather than natural logs) where warmth from the decaying material can sustain a colony for a few seasons. Thus, once a pile is no longer suitable, dispersal to new sawdust piles or into natural logs is required or, conversely, those northernmost populations repeatedly become extinct and are maintained over the long term by perpetual reintroductions from more southern populations. Once believed to be highly endemic as a result of an inability to disperse, several Zorotypus species are increasingly being recognized over larger geographic ranges (e.g., Z. hubbardi in south-central and southeastern United States), evidence of an ability to disperse to some degree so as to maintain specific integrity.
GENERAL BIOLOGY
The biology of zorapterans is poorly documented. Only three species have been studied and only one, Z. hubbardi, generated a sizeable literature. Species are gregarious with colonies consisting of 15-120 individuals in relatively ephemeral, subcortical habitats. Colonies occur in logs (or man-made sawdust piles) but only after wood decay is well progressed. Zorapterans feed principally on fungal hyphae and spores, although they can also be predatory, victimizing nematodes, mites, and other minute arthropods such as springtails (Collembola). The number of nymphal instars is estimated to be either four or five. Captive individuals live up to 110 days.
Individuals occur in two morphs. Blind, wingless individuals predominate during the general life of a colony, whereas dispersive individuals (with eyes and wings) are generally rare and are produced as resources become depleted and the colony becomes crowded. Winged females are more common than winged males, suggesting that females perhaps mate prior to dispersal. After arriving at a new log, individuals shed their wings and dealated individuals are often found in young colonies. Wide distributions for some species as well as the presence of species endemic to distantly isolated islands such as Christmas Island, Fiji, and Hawaii are evidence for their dispersal abilities.
BEHAVIOR
Although Zorotypus species are gregarious and live in small colonies, they evidently do not distinguish between individuals from the same colony and introduced vagrants. Isolated individuals do not survive.
Zorapterans spend much of their time grooming either themselves or other individuals. Grooming may be a way of removing fungal pathogens and this may be important for their colonial lifestyle. Most of the movements in the grooming repertoire are found in other insect orders but some are unique to zorapterans. Most notable are the movements associated with the cleaning of the posterior sterna, the cerci, and the genital-anal area using their mouthparts. This complex action involves the raising of the body on a four-point stance, with both anterior and posterior ends of the body bent downward (perpendicular with the substrate) to meet under the insect and between the middle and the hind legs.
Females mate multiple times (with up to eight mates) and with multiple males (up to three multiple matings). Prior to copulation, zorapteran species have complex courtship behaviors which appear to be species specific. The process is begun by the male who strokes the female with his antennae. If the female reciprocates with similar anten-nal signals, the male initiates courtship displays during which his head and a portion of his neck are extended. If the female approaches, the male secretes fluids from a cephalic gland, which she ingests while the male and female bend their abdomens toward one another. Copulation is initiated by the linking of the male to the female via a hook located medially on his 10th abdominal tergum (females have a small groove into which the hook is placed on their eighth sternum). Once the hook is coupled with the female, the male inverts himself, assuming an upside-down position and facing away from the female.
ECONOMIC IMPORTANCE
Although zorapteran colonies at times can be found in wood or sawdust piles at lumber yards, owing to their relatively minute colony sizes, general scarcity, and apparent minimal ecological impact, it is unlikely that they will ever be considered of any economic importance. If the fragmentary reports of wood ingestion by zorapterans are confirmed as well as studies of their gut fauna (bacteria, protozoa, etc.) undertaken, then the economic role of Zorotypus might be reevaluated.
