The digestive system consists of the alimentary canal (gut) and salivary glands, and is responsible for all steps in food processing: digestion, absorption, and feces delivery and elimination. These steps occur along the gut. The anterior (foregut) and posterior (hindgut) parts of the gut have cells covered by a cuticle whereas, in the midgut, cells are separated from the food by a filmlike anatomical structure referred to as the peritrophic membrane. Salivary glands are associated with the foregut and may be important in food intake but usually not in digestion. Remarkable adaptations are found in taxa with very specialized diets, such as cicadas (plant sap), dung beetles (feces), and termites (wood), and in insects with short life spans, as exemplified by flies and moths. Digestion is carried out by insect digestive enzymes, apparently without participation of symbiotic microorganisms.
GUT MORPHOLOGY AND FUNCTION
Figure 1 is a generalized diagram of the insect gut. The foregut begins at the mouth, includes the cibarium, the pharynx, the esophagus, and the crop (a dilated portion, as in Fig. 2A, or a diverticulum, such as Fig. 2K). The crop is a storage organ in many insects and also serves as a site for digestion in others. The foregut in some insects is reduced to a straight tube (Fig. 2F). The proventriculus is a triturating (grinding into fine particles) organ in some insects, and in most it provides a valve controlling the entry of food into the midgut, which is the main site of digestion and absorption of nutrients.
The midgut includes a simple tube (ventriculus) from which blind sacs (gastric or midgut ceca) may branch, usually from its anterior end (Figs. 1 and 2A). Midgut ceca may also occur along the midgut in rings (Fig. 2F) or not (Fig. 2H) or in the posterior midgut (Fig. 2Q). In most insects, the midgut is lined with a filmlike anatomical structure (peritrophic membrane) that separates the luminal contents into two compartments: the endoperitrophic space and the ectoperi-trophic space. Some insects have a stomach, which is an enlargement of the midgut to store food (Fig. 2R). In the region of the sphincter (pylorus) separating the midgut from the hindgut, Malpighian tubules branch off the gut. Malpighian tubules are excretory organs that may be joined to form a ureter (Fig. 2B); in some species, however, they are absent (Fig. 2O).
The hindgut includes the ileum, colon, and rectum and terminates with the anus (Fig. 1). In some insects it is reduced to a straight tube (Fig. 2G), in others it is modified in a fermentation chamber (Fig. 2F) or paunch (Fig. 2D), with both structures storing ingested food and harboring microorganisms that have a controversial role in assisting cellulose digestion.
The gut epithelium is always simple and rests on a basal lamina that is surrounded by conspicuous circular and a few longitudinal muscles. Wavelike contractions of the circular muscles cause peristalsis, propelling the food bolus along the gut.
Salivary glands are labial or mandibular glands opening in the cibarium. They are usually absent in Coleoptera. The saliva lubricates the mouthparts, may contain an array of compounds associated with blood intake, or may be used as a fixative of the stylets of sap-sucking
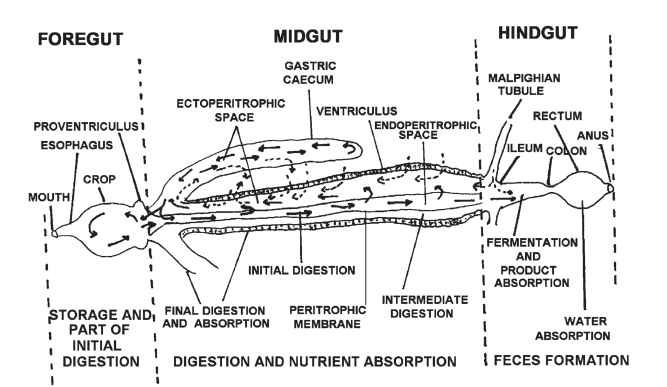
FIGURE 1 Generalized diagram of the insect gut.
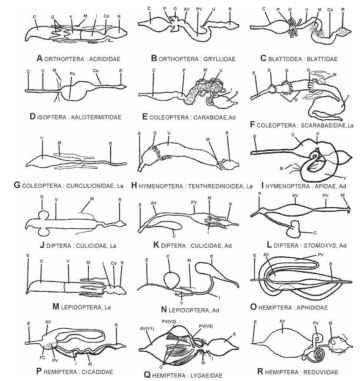
FIGURE 2 Major insect gut types: Ad, adult; AV, anterior ventricu-lus (midgut); C, crop; Co, colon; E, esophagus; F, fermentation chamber; FC, filter chamber; G, midgut (gastric) ceca; I, ileum; La, larva; M, Malpighian tubules; P, proventriculus; Pa, paunch; PV, posterior ventricu-lus (midgut); R, rectum; V, ventriculus. Not drawn to scale.
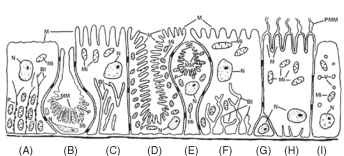
FIGURE 3 Diagrammatic representation of typical insect midgut cells: (A) columnar cell with plasma membrane infoldings arranged in long and narrow channels, usually occurring in fluid-absorbing tissues; (B) lepidopteran long-necked goblet cell; (C) columnar cell with highly developed basal plasma membrane infoldings displaying few openings into the underlying space, usually occurring in fluid-absorbing tissue; (D) cyclorrhaphan dipteran oxyntic (cuprophilic) cell; (E) lepidopteran stalked goblet cell; (F) columnar cell with highly developed plasma membrane infoldings with numerous openings into the underlying space, frequently present in fluid-secreting tissue; (G) regenerative cell; (H) hemipteran midgut cell; (I) endocrine cell. Note particles (portasomes) studding the cytoplasmic side of the apical membranes in B, D, and E and of the basal plasma membranes in A. Abbreviations: Bl, basal plasma membrane infoldings; M, micro-villi; Mi, mitochondria; MM, modified microvilli; N, nucleus; P, porta-somes; PMM, perimicrovillar membranes; V, vesicles.
bugs. Saliva usually contains only amylase and maltase or no enzymes at all, although in a few hemipteran predators it may have the whole complement of proteolytic enzymes.
The epithelium of the midgut is composed of a major type of cell usually named columnar cell (Fig. 3A, 3C, and 3F), although it may

FIGURE 4 Midgut cell apexes: (A) Electron micrograph of M. domestica posterior midgut cell. L, lumen; Mv, microvilli; PM, peritrophic membrane. Magnification: 7500 X. (B) Electron micrograph of columnar cell of Erinnyis ello anterior midgut. Detail of microvilli showing glycocalyx (arrows). Magnification: 52,000 X. (Reproduced with permission from Santos, C. D., Ribeiro, A. F., Ferreira, C., Terra, W. R. (1984). The larval midgut of the cassava hornworm (Erinnyis ello). Ultrastructure, fluid fluxes and the secretory activity in relation to the organization of digestion. Cell Tiss. Res. 237, 565-574). (C) Diagrammatic representation of the distribution of enzymes on the midgut cell surface. Glycocalyx: the carbohydrate moiety of intrinsic proteins and glycolipids occurring in the luminal face of microvillar membranes.
have other forms; it also contains regenerative cells (Fig. 3G) that are often collected together in nests at the base of the epithelium, cells (Fig. 3I) believed to have an endocrine function, and also specialized cells (goblet cells, Fig. 3B and 3E; oxyntic cells, Fig. 3D; hemipteran midgut cell, Fig. 3H).
The peritrophic membrane (Fig. 4A) is made up of proteins (per-itrophins) and chitin to which other components (e.g., enzymes and food molecules) may associate. This anatomical structure is sometimes called the peritrophic matrix, but this term is better avoided because it does not convey the idea of a film and suggests that it is the fundamental substance of some structure. The argument that “membrane” means a lipid bilayer does not hold here because the peritrophic membrane is an anatomical structure, not a cell part. Peritrophins have domains similar to mucins (gastrointestinal mucus proteins) and other domains able to bind chitin. This suggests that the peritrophic membrane may have derived from an ancestral mucus. According to this hypothesis, the peritrophins evolved from mucins by acquiring chitin-binding domains. The parallel evolution of chitin secretion by midgut cells led to the formation of the chitin-protein network characteristic of the peritrophic membrane.
The formation of the peritrophic membrane may occur in part of the midgut or in the entire organ (type I), or only at the entrance of the midgut (cardia) (type II). Type I peritrophic membrane occurs in most insects, whereas type II is restricted to larval and adult (except hematophagous) mosquitoes and flies (Diptera) and a few adult Lepidoptera. Although a peritrophic membrane is found in most
insects, it does not occur in Hemiptera and Thysanoptera, which have perimicrovillar membranes in their cells (Fig. 3H). The other insects that do not seem to have a peritrophic membrane are adult Lepidoptera, Phthiraptera, Psocoptera, Zoraptera, Strepsiptera, Raphidioptera, Megaloptera, and Siphonaptera as well as bruchid beetles and some adult ants (Hymenoptera). Most of the pores of the peritrophic membrane are in the range of 7-9 nm, although some may be as large as 36 nm. Thus, the peritrophic membrane hinders the free movement of molecules, dividing the midgut lumen into two compartments (Fig. 1) with different molecules. The functions of this structure include those of the ancestral mucus (protection against food abrasion and microorganism invasion) and several roles associated with the compartmentalization of the midgut. These roles result in improvements in digestive efficiency and assist in decreasing digestive enzyme excretion, and in restricting the production of the final products of digestion close to their transporters, thus facilitating absorption.
DIGESTIVE PHYSIOLOGY
Overview
The study of digestive physiology involves the spatial organization of digestive events in the insect gut. Digestive enzymes that participate in primary digestion (cleavage of polymers such as protein and starch), secondary digestion (action on oligomers exemplified by polypeptides and dextrans), and final digestion (hydrolysis of dimers such as dipeptides and disaccharides) are assayed in different gut compartments. Samples of the ectoperitrophic space contents (Fig. 1) are collected by puncturing the midgut ceca with a capillary or by washing the luminal face of midgut tissue. Midgut tissue enzymes are intracellular, glycocalyx-associated, or microvillar membrane-bound. Their location is determined by cell fractionationation. Midgut cell microvilli have, in addition to be the most frequent site of final digestion, a role in midgut protection in lepidopterans. This role includes protection against oxidative stress, detoxification of H2O2, and aldehydes and against the action of the insect’s own lumi-nal serine proteinases. In addition to the distribution of digestive enzymes, the spatial organization of digestion depends on midgut fluxes. Fluxes are inferred with the use of dyes. Secretory regions deposit injected dye onto the midgut hemal side, whereas absorbing regions accumulate orally fed dyes on the midgut luminal surface.
Upon studying the spatial organization of the digestive events in insects of different taxa and diets, it was realized that the insects may be grouped relative to their digestive physiology, assuming they have common ancestors. Those putative ancestors correspond to basic gut plans from which groups of insects may have evolved by adapting to different diets.
Neopteran insects evolved along three lines: the Polyneoptera (which include Blattodea and Mantodea, Isoptera, and Orthoptera), the Paraneoptera (which include Hemiptera), and the Holometabola (which include Coleoptera, Hymenoptera, Diptera, and Lepidoptera). Polyneoptera and Paraneoptera evolved as external feeders occupying the ground surface, on vegetation, or in litter, and developed distinct feeding habits. Some of these habits are very specialized (e.g., feeding wood and sucking plant sap), implying adaptative changes of the digestive system. Major trends in the evolution of Holometabola were the divergence in food habits between larvae and adults and the exploitation of new food sources, exemplified by endoparasitism and by boring or mining living or dead wood, foliage, fruits, or seeds. This biological variation was accompanied by modifications in the digestive system. Among the hymenopteran and panorpoid (an assemblage that includes Diptera and Lepidoptera) Holometabola, new selective pressures resulted from the occupation of more exposed or ephemeral ecological niches. Following this trend, those pressures led to shortening life spans, so that the insects may have more generations per year, thus ensuring species survival even if large mortality occurs at each generation. Associated with this trend, the digestive system evolved to become more efficient to support faster life cycles.
The basic plan of digestive physiology for most winged insects (Neoptera ancestors) is summarized in Fig. 5A. In these ancestors, the major part of digestion is carried out in the crop by digestive enzymes propelled by antiperistalsis forward from the midgut. Saliva plays a minor role or no role at all in digestion. After a while, following ingestion, the crop contracts, transferring digestive enzymes and partly digested food into the ventriculus. The anterior ventricu-lus is acidic and has high carbohydrase activity, whereas the posterior ventriculus is alkaline and has high proteinase activity. The food bolus moves backward in the midgut of the insect by peristalsis. As soon as the polymeric food molecules have been digested to become small enough to pass through the peritrophic membrane, they diffuse with the digestive enzymes into the ectoperitrophic space (Fig. 1) . The enzymes and nutrients are then displaced toward the ceca with a countercurrent flux caused by secretion of fluid at the Malpighian tubules and its absorption back by cells (similar to Fig. 3A and 3C) at the ceca (Fig. 5A), where final digestion is completed and nutrient absorption occurs. When the insect starts a new meal, the ceca contents are moved into the crop. As a consequence of the countercurrent flux,digestive enzymes occur as a decreasing gradient in the midgut, and lower amounts are excreted.
The Neoptera basic plan is the source of that of the Polyneoptera orders and evolved to the basic plans of Paraneoptera and Holometabola. Lack of data limits the proposition of a basic plan to a single Paraneoptera order, Hemiptera. Symbiont microorganisms may occur in large numbers in insect gut. For example, the bacteria Nocardia rhodnii may represent up to 5% of the Rhodnius prolixus midgut dry weight. The symbionts are believed to provide nutrient factors (such as essential amino acids, B vitamins, or fermentation end products) to the host. Microorganism symbionts have rarely been associated with digestion, and the few that are known are implicated in rupturing plant cell walls, mainly by degrading lignin. They are important, however, in overcoming barriers to herbivory by detoxifying noxious glucosides, flavonoids, and alkaloids.
Polyneoptera
BLATTODEA AND MANTODEA
Cockroaches, which are among the first neopteran insects to appear in the fossil record, are extremely generalized in most morphological features. They are usually omnivorous. Digestion in cockroaches occurs as described for the Neoptera ancestor (Fig. 5A), except that part of the final digestion of proteins occurs on the surface of midgut cells. The differentiation of pH along the midgut is not conserved among cockroaches like Periplaneta americana, but is maintained in others exemplified by the blaberid Nauphoeta cinerea. Another difference observed is the enlargement of hindgut structures (Fig. 2C) , noted mainly in wood-feeding cockroaches. These hindgut structures harbor bacteria producing acetate and butyrate from ingested wood or other cellulose-containing materials. Acetate and butyrate are absorbed by the hindgut of all cockroaches. Cellulose digestion may be accomplished by bacteria, but there is evidence that wood roaches have their own cellulases. Mantids have a capacious crop, and a short midgut and hindgut. It is probable that the major part of digestion takes place in their crops.
ISOPTERA
Termites are derived from and are more adapted than wood roaches in dealing with refractory materials such as wood and humus. Associated with this specialization, they lost the crop and midgut ceca and enlarged their hindgut structures (Fig. 2D) . Termites digest cellulose with their own cellulase, and the products pass from the midgut into the hindgut, where they are converted into acetate and butyrate by hindgut bacteria as in wood roaches. Symbiotic bacteria are also responsible for nitrogen fixation in hind-gut, resulting in bacterial protein. This is incorporated into the termite body mass after being expelled in feces by one individual and being ingested and digested by another. This explains the ability of termites to develop successfully in diets very poor in protein.
ORTHOPTERA
Grasshoppers feed mainly on grasses, and their digestive physiology clearly evolved from the neopteran ancestor. Carbohydrate digestion occurs mainly in the crop, under the action of midgut enzymes, whereas protein digestion and final carbohydrate digestion take place at the anterior midgut ceca. The abundant saliva (devoid of significant enzymes) produced by grasshoppers saturate the absorbing sites in the midgut ceca, thus hindering the countercurrent flux of fluid. Starving grasshoppers present midgut countercurrent fluxes. Cellulase found in some grasshoppers is believed to facilitate the access of digestive enzymes to the plant cells ingested by the insects by degrading the cellulose framework of cell walls. Crickets are omnivorous or predatory insects with most starch and protein digestion occurring in their capacious crop (Fig. 2B).
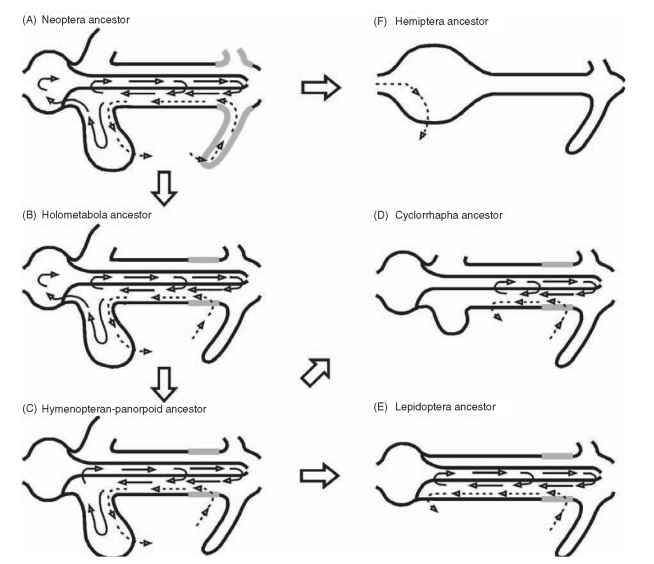
FIGURE 5 Diagrammatic representation of water fluxes (dashed arrows) and of the circulation of digestive enzymes (solid arrows) in putative insect ancestors that correspond to the major basic gut plans. In Neoptera ancestors (A), midgut digestive enzymes pass into the crop. Countercurrent fluxes depend on the secretion of fluid by the Malpighian tubules and its absorption by the ceca. Enzymes involved in initial, intermediate, and final digestion circulate freely among gut compartments. Holometabola ancestors (B) are similar except that secretion of fluid occurs in posterior ventriculus. Hymenopteran-Panorpoid (Lepidoptera and Diptera assemblage) ancestors (C) display countercurrent fluxes like Holometabola ancestors, midgut enzymes are not found in crop, and only the enzymes involved in initial digestion pass through the peritrophic membrane. Enzymes involved in intermediate digestion are restricted to the ectoperitrophic space and those responsible for terminal digestion are immobilized at the surface of midgut cells. Cyclorrhapha ancestors (D) have a reduction in ceca, absorption of fluid in middle midgut, and anterior midgut playing a storage role. Lepidoptera ancestors (E) are similar to panorpoid ancestors, except that anterior midgut replaced the ceca in fluid absorption. Hemiptera ancestors (F) lost crop, ceca, and fluid-secreting regions. Fluid is absorbed in anterior midgut.
Paraneoptera
HEMIPTERA
The characteristics of the Paraneoptera ancestors cannot be inferred because midgut function data are available only for Hemiptera. The Hemiptera comprise insects of several suborders (e.g., cicadas, leafhoppers, aphids, and fulgorids) that feed almost exclusively on plant sap, and insects of the taxon Heteroptera (e.g., assassin bugs, plant bugs, stink bugs, and lygaeid bugs) that are adapted to different diets. The ancestor of the entire order is supposed to be a sapsucker similar to present-day cicadas and fulgorids.
The hemipteran ancestor (Fig. 5F) differs remarkably from the neopteran ancestor, as a consequence of adaptations to feeding on plant sap. These differences consist of the lack of crop and anterior midgut ceca, loss of the enzymes involved in initial and intermediate digestion and loss of the peritrophic membrane associated with the lack of luminal digestion, and, finally, the presence of hemipteran midgut cells (Figs. 3H and 6). These cells have their microvilli ensheathed by an outer (perimi-crovillar) membrane that extends toward the luminal compartment with a dead end (Fig. 6). Aphids have modified perimicrovillar membranes.

FIGURE 6 The microvillar border of midgut cells from Hemiptera. Electron micrograph of R prolixus posterior midgut cell. Detail of microvilli showing the extension of the perimicrovillar membrane (PMM) in to the midgut lumen (arrowheads). Scale bar: 1 |im. (Reproduced with permission from: Ferreira, C., Ribeiro, A. F., Garcia E. S., and Terra, W. R.. (1988). Digestive enzymes trapped between and associated with the double plasma membranes of Rhodnius prolixus posterior midgut cells. Insect Biochem. 18, 521-530.)
Sap-sucking Hemiptera may suck phloem or xylem sap. Phloem sap is rich in sucrose (0.15-0.73 M) and relatively poor in free amino acids (15-65 mM) and minerals. Some rare phloems have considerable amounts of protein. Xylem fluid is poor in amino acids (3-10 mM) and contains monosaccharides (about 1.5 mM), organic acids, potassium ions (about 6 mM), and other minerals. Thus, as a rule, no food digestion is necessary in sapsuckers except for dimer (sucrose) hydrolysis. The major problem facing a sap-sucking insect is to absorb nutrients, such as essential amino acids, that are present in very low concentrations in sap. Amino acids may be absorbed according to a hypothesized mechanism that depends on perimicrovillar membranes.
Organic compounds in xylem sap need to be concentrated before they can be absorbed by the perimicrovillar system. This occurs in the filter chamber (Fig. 2P) of Cicadoidea and Cercopoidea, and Cicadelloidea. The filter chamber consists of a thin-walled, dilated anterior midgut in close contact with the posterior midgut and the proximal ends of the Malpighian tubules. This arrangement enables water to pass directly from the anterior midgut to the Malpighian tubules through specific channels made up of aquaporin molecules, thus concentrating food in midgut.
The evolution of Heteroptera was associated with regaining the ability to digest polymers. Because the appropriate digestive enzymes were lost, these insects instead used proteinases (cathep-sins) derived from lysosomes. Compartmentalization of digestion was maintained by the perimicrovillar membranes as a substitute for the lacking peritrophic membrane. Digestion in the two major Heteroptera taxa—Cimicomorpha, exemplified by the blood feeder R. prolixus, and Pentatomorpha, exemplified by the seed sucker Dysdercus peruvianus—is similar. The dilated anterior midgut stores food and absorbs water and, at least in D. peruvianus, also absorbs glucose. Digestion of proteins and absorption of amino acids occur in the posterior ventriculus. Most protein digestion occurs in lumen with the aid of a cysteine proteinase and ends in the perimicrovillar space under the action of aminopeptidases and dipeptidases. Many Heteroptera feed on parenchymal tissues of plants. In some of these insects, excess water passes from the expanded anterior midgut to the closely associated midgut ceca, which protrude from the posterior midgut (Fig. 2Q). These ceca may also contain symbiont bacteria.
Holometabola
The basic gut plan of the Holometabola (Fig. 5B) is similar to that of Neoptera except that fluid secretion occurs in the posterior ventriculus by cells similar to Fig. 3F, instead of by the Malpighian tubules. Because of this the posterior midgut fluid, unlike Malpighian tubular fluid, does not contain wastes, the accumulation of wastes in ceca is decreased. Digestive systems may change remarkably between larvae and adults of holometabolous insects. Despite these changes, adult digestive systems probably evolved in parallel to larval systems because, except for minor differences, the compartmentali-zation of digestion in larvae and adults seems to be similar.
The basic plan of Coleoptera did not evolve dramatically from the Holometabola ancestor, whereas the basic plan of Hymenoptera, Diptera, and Lepidoptera (hymenopteran-panorpoid ancestor, Fig. 5C) presents important differences. Thus, hymenopteran-panorpoid ancestors have countercurrent fluxes like Holometabola ancestors but differ from these in the lack of crop digestion, in midgut differentiation in luminal pH, and in which compartment is responsible for each phase of digestion. In Holometabola ancestors, all phases of digestion occur in the endoperitrophic space (Fig. 1 ) , whereas in hymenopteran-panorpoid ancestors only initial digestion occurs in that region. In the latter ancestors, intermediate digestion is carried out by free enzymes in the ectoperitrophic space and final digestion occurs at the midgut cell surface by immobilized enzymes. The free digestive enzymes do not pass through the peritrophic membrane because they are larger than the peritrophic membrane pores. Immobilized enzymes may be either soluble enzymes entrapped in the cell glycocalyx or membrane-bound enzymes (Fig. 4C). As a consequence of the compartmentalization of digestive events in panorpoid insects, there is an increase in the efficiency of digestion of polymeric food by allowing the removal of the oligomeric molecules from the endoperitrophic space, which in turn is powered by the recycling mechanism associated with the midgut fluxes. Because oligomers may be substrates or inhibitors for some polymer hydrolases, their presence should decrease the rate of polymer degradation. Another consequence of compartmentalization is an increase in the efficiency of oligomeric food hydrolysis by allowing the transference of oligomeric molecules to the ectoperitrophic space and by restricting oligomer hydrolases to this compartment. In these conditions, oligomer hydrolysis occurs in the absence of probable partial inhibition (because of nonproductive binding) by polymer food and presumed nonspecific binding by nondispersed undigested food.
Finally, the peritrophic membrane prevents nonspecific binding of undigested material onto midgut cell surface. All these functions were supported by data obtained with model systems.
COLEOPTERA
Larvae and adults of Coleoptera usually display the same feeding habit; that is, both are plant feeders (although adults may feed on the aerial parts, the larvae may feed on the roots of the same plant) or both are predatory. Coleoptera ancestors are like Holometabola ancestors except for the anterior midgut ceca, which were lost and replaced in function by the anterior midgut. Nevertheless, there are evolutionary trends leading to derived systems. Thus, in predatory Carabidae most of the digestive phases occur in the crop by means of midgut enzymes, whereas in predatory larvae of Elateridae initial digestion occurs extraorally by the action of enzymes regurgitated onto their prey. The preliquefied material is then ingested by the larvae, and its digestion is finished at the surface of midgut cells. In Tenebrionidae, the final digestion of proteins takes place at midgut cell surface; in Curculionidae and Cerambycidae, the final digestion of all nutrients is carried out at midgut cell surface. The distribution of enzymes in gut regions of adult Tenebrionidae is similar to that of their larvae. This suggests that the overall pattern of digestion in larvae and adults of Coleoptera is similar even though (in contrast to adults) beetle larvae usually lack a crop. Insects of the series Cucujiformia (which includes Tenebrionidae, Chrysomelidae, Bruchidae, and Curculionidae) have cysteine proteinases in addition to (or in place of) serine proteinases as digestive enzymes, suggesting that the ancestors of the whole taxon were insects adapted to feed on seeds rich in serine proteinase inhibitors.
Scarabaeidae evolved considerably from the Coleoptera ancestor. Scarabid larvae, exemplified by dung beetles, usually feed on cellulose materials undergoing degradation by a fungus-rich flora. Digestion occurs in the midgut, which has three rows of ceca (Fig. 2F), with a ventral groove between the middle and posterior row. The alkalinity of gut contents increase to almost pH 12 along the midgut ventral groove. This high pH probably enhances cellulose digestion, which occurs mainly in the hindgut fermentation chamber (Fig. 2F). The final product of cellulose degradation is mainly acetic acid, which is absorbed through the hindgut wall.
HYMENOPTERA
Hymenoptera comprise several primitive suborders (including sawflies and horntails) and Apocrita. Apocrita are divided into Parasitica, which are parasites of other insects, and the derived group Aculeata (bees, ants, wasps with thin waists), in which the piercing ovipositor of Parasitica evolved into a stinging organ. The first Apocrita were probably close to the ichneumon flies, whose larvae develop on the surface or inside the body of the host insect. Probably because of that, the larvae of Apocrita present a midgut that is closed at its rear end, and remains unconnected with the hindgut until the time of pupation. Hymenoptera ancestors are like panorpoid ancestor, but there are trends leading to the loss of anterior midgut ceca, and in compartmentalization of digestion. These trends appear to be associated with the development of parasitic habits and were maintained in Aculeata.
The sawfly Themos malaisei (Tenthredinoidea) larva has a midgut with a ring of anterior ceca that forms a U at the ventral side (Fig. 2H). Luminal pH is above 9.5 in the first two-thirds of the midgut. There is a recycling of enzymes involved in initial digestion and the final digestion occurs in the midgut cell surface. These characteristics (except the presence of ceca) are similar to those of lepidopteran larvae. Wood wasp larvae of the genus Sirex are believed to be able to digest and assimilate wood constituents by acquiring cellulase,xylanase, and possibly other enzymes from fungi present in wood on which they feed.
In larval bees, most digestion occurs in the endoperitrophic space. Countercurrent fluxes seem to occur, but the midgut luminal pH gradient hypothetically present in the Hymenoptera-Panorpoidea ancestor was lost. Adult bees ingest nectar and pollen. Sucrose from nectar is hydrolyzed in the crop (Fig. 2I) by the action of a sucrase from the hypopharyngeal glands. After ingestion, pollen grains extrude their protoplasm into the ventriculus, where digestion occurs. Workers of leaf-cutting ants feed on nectar, honeydew, plant sap, or partly digested food regurgitated by their larvae. These adults lack most digestive enzymes and appear to rely mainly on monosaccharides produced by fungal enzymes acting on plant polysaccharides. Other adult ants (e.g., Formicinae) have the whole complement of digestive enzymes in the endoperitrophic space.
DIPTERA
The Diptera evolved along two major lines: an assemblage of suborders corresponding to the mosquitoes, including the basal Diptera, and the suborder Brachycera, which includes the most evolved flies (Cyclorrhapha). The Diptera ancestor is similar to the hymenopteran-panorpoid ancestor (Fig. 5C) in having the enzymes involved in intermediate digestion free in the ectoperitrophic fluid (mainly in the large ceca), whereas the enzymes of terminal digestion are membrane bound at the midgut cell microvilli. Although these characteristics are observed in most nonbrachyceran larvae, the more evolved of these larvae may show reduction in size of midgut ceca (e.g., Culicidae, Fig. 2K). Nonhematophagous adults store (nectar or decay products) in their crops and carried out digestion and absorption at the anterior midgut. Blood, which is sucked only by females, passes to the posterior midgut, where it is digested and absorbed.
The Cyclorrhapha ancestor (Fig. 5D) evolved dramatically from the hymenopteran-panorpoid ancestor (Fig. 5C), apparently as a result of adaptations to a diet consisting mainly of bacteria. Digestive events in Cyclorrhapha larvae are exemplified by larvae of the house fly Musca domestica. These larvae ingest food rich in bacteria. In the anterior midgut, there is a decrease in the starch content of the food bolus, facilitating bacteria death. The bolus now passes into the middle midgut where bacteria are killed by the combined action of low pH, a special lysozyme, and an aspartic proteinase. Finally, the material released by bacteria is digested in the posterior midgut. Countercurrent fluxes occur in the posterior midgut powered by secretion of fluid in the distal part of the posterior midgut and its absorption back into the middle midgut. The middle midgut has specialized cells for buffering the luminal contents in the acidic zone (Fig. 3D), in addition to those functioning in fluid absorption (Fig. 3A). Except for a few bloodsuckers, cyclorrhaphan adults feed mainly on liquids associated with decaying material (rich in bacteria) in a way similar to house fly adults. The stable fly, Stomoxys calci-trans, stores and concentrates the blood meal in the anterior midgut and gradually passes it to the posterior midgut, where digestion takes place, resembling what occurs in larvae. These adults lack the characteristic cyclorrhaphan middle midgut and the associated low luminal pH. Stable flies occasionally take nectar.
LEPIDOPTERA
Lepidopteran ancestors ( Fig. 5E) differ from hymenopteran-panorpoid ancestors because they lack midgut ceca, have all their digestive enzymes (except those of initial digestion) immobilized at the midgut cell surface, and present long-necked goblet cells (Fig. 3B) and stalked goblet cells (Fig. 3E) in the anterior and posterior larval midgut regions, respectively. Goblet cells excrete K+ ions, which are absorbed from leaves ingested by larvae. Although most lepidopteran larvae have a common pattern of digestion, species that feed on unique diets generally display some adaptations. Tineola bisselliella (Tineidae) larvae feed on wool and display a highly reducing midgut for cleaving the disulfide bonds in keratin to facilitate proteolytic hydrolysis of this otherwise insoluble protein. Wax moths ( Galleria mellonella) infest beehives, and digest and absorb wax. The participation of symbiotic bacteria in this process is controversial. The occurrence of the whole complement of digestive enzymes in nectar-feeding moths may explain, at least on enzymological grounds, the adaptation of some adult Lepidoptera to new feeding habits such as blood and pollen.
ABSORPTION OF WATER AND NUTRIENTS
Overview and Absorption of Lipids
Absorption is the passage of molecules and ions from the gut lumen into the gut cells, thus traversing the cuticle (if present) and the cell plasma membrane. Absorption depends on the permeability of those barriers and on the concentration ratio of a compound in gut lumen and inside gut cells. The permeability of cuticles is variable, whereas that of the plasma membrane is greater for water and for hydrophobic compounds. Thus, absorption of hydrophilic compounds requires special devices (transporters) to help the molecules find their way through the cell membrane. These transporters are transmembrane proteins that bind the molecule to be transported in a membrane face and, after suitable conformational changes, deliver the molecule from the other face. A uniporter is a transporter that carries a single solute, whereas symporters and antiporters are transporters that carry two solutes into the same and opposite directions, respectively. To transport molecules against a concentration gradient, the process must be energized by coupling with ATP hydrolysis or with cotransport of another molecule down its concentration gradient. Transporter-mediated absorption may be inhibited by molecules resembling those of the transported solute, and its velocity attains a maximum (transporter becomes saturated) at a high solute concentration. This behavior is not observed in the case of simple diffusion, exemplified by lipid absorption.
Absorption sites in insect guts are identified by feeding groups of insects with known dye solutions and then dissecting insects at different periods of time. If the insect is large enough, absorption studies can go further, using gut sections mounted as a sac and measuring the rates at which compounds traverse it under different conditions.
Tracer studies showed that lipid is absorbed more heavily in the anterior than in the posterior midgut of insects, thus following the tissue distribution of fatty acid binding proteins. These proteins are thought to facilitate fatty acid uptake by cells, by decreasing their diffusion back from cells to the gut lumen and by targeting them to specific metabolic pathways. The fatty acids acetate and butyrate are absorbed in significant amounts by the hindgut of insects utilizing cellulose.
Water
Water absorption in the midgut occurs associated with midgut fluid fluxes, but in large amounts it is characteristic of insects feeding on dilute diets, of blood feeders, and also of insects in which salivation is important (e.g., grasshoppers and seed-sucker bugs). Water absorption in the hindgut is part of the water conservation mechanism that is important in all terrestrial insects. Frequently, special cell aggregates (rectal pads) are involved in this process. Water uptake is
thought to depend on salt being pumped into spaces enclosed by the basolateral infoldings (Fig. 3A and 3C) of the absorbing cell. This creates an osmotic pressure that moves water into these restricted spaces. The resulting hydrostatic pressure drives water into the hemolymph, with salts being absorbed from the water on its way out. Water is also cotransported with sugars.
Ions, Amino Acids, and Sugars
In insects, as is usual for all animals, most nutrient absorption occurs in the midgut through symporters, with ions being cotrans-ported down the concentration gradient. Favorable ion gradients are maintained by ion pumps. The most ubiquitous of these pumps is the ATP-driven Na+, K+-antiporter (Na+-K+-ATPase) localized in the midgut cell basal membrane. Another important ion pump is the H+, K+-ATPase found in the goblet cell (Fig. 3B and 3E) microvillar membranes of lepidopteran larvae. Those pumps maintain cell Na+ and cell K+ low in insects with Na+-rich and K+-rich diets, respectively. The midgut of lepidopteran larvae contains K+ -dependent symporters for amino acids, consistent with the K+ -rich plant diets of these larvae. There are usually symporters for neutral, acidic, and basic amino acids. Insects with high-Na+ diets seem to have midgut amino acid symporters dependent on Na+ , as shown in cockroaches. The absorption of glucose occurs with the aid of uniporters or ion-dependent symporters. Ongoing research suggests that both transporters carry also water molecules, even from hyperosmotic solutions.
The primary urine produced in Malpighian tubules contains salts and amino acids, and passes into the hindgut together with food remains. Salts are absorbed in the hindgut by means of special pumps, like those for chloride and calcium. Amino acids, at least in locusts, are absorbed in the hindgut through a Na+ -dependent amino acid symporter.
MIDGUT SECRETORY MECHANISMS
Insects are continuous (e.g., Lepidoptera and Diptera larvae) or discontinuous (e.g., predators and hematophagous insects) feeders. Synthesis and secretion of digestive enzymes in continuous feeders seem to be constitutive; that is, these functions occur continuously, whereas in discontinuous feeders they are regulated. It is widely believed (without clear evidence) that putative endocrine cells (Fig. 3I) play a role in regulating midgut events. The presence of food in the midgut is necessary to stimulate synthesis and secretion of digestive enzyme. This was clearly shown in mosquitoes.
Like all animal proteins, digestive enzymes are synthesized in the rough endoplasmic reticulum, processed in the Golgi complex, and packed into secretory vesicles (Fig. 7). There are several mechanisms by which the contents of the secretory vesicles are freed in the mid-gut lumen. During exocytic secretion, secretory vesicles fuse with the midgut cell apical membrane, emptying their contents without any loss of cytoplasm (Fig. 7A) . In contrast, apocrine secretion involves the loss of at least 10% of the apical cytoplasm following the release of secretory vesicles (Fig. 7B). These have previously undergone fusions originating larger vesicles that after release eventually free their contents by solubilization (Fig. 7B) . When the loss of cytoplasm is very small, the secretory mechanism is called microapocrine. Microaprocrine secretion consists of releasing budding double-membrane vesicles (Fig. 7C) or, at least in insect midguts, pinched-off vesicles that may contain a single or several secretory vesicles (Fig. 7D). In both apocrine and microapocrine secretion, the secretory vesicle contents are released by membrane fusion and/or by membrane solubilization due to high pH contents or to the presence of detergents.
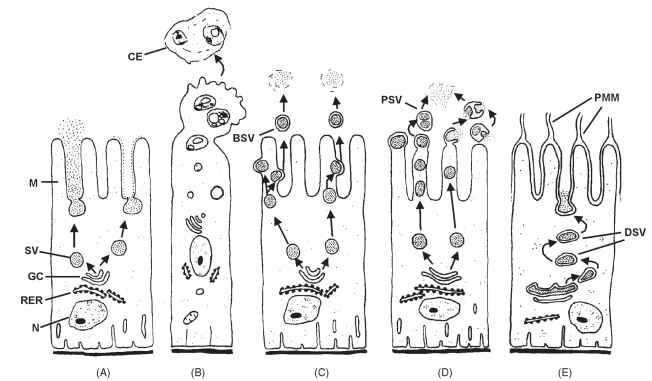
FIGURE 7 Models for secretory processes of insect digestive enzymes; (A) exocytic secretion, (B) apocrine secretion, (C) microapocrine secretion with budding vesicles, (D) microapocrine secretion with pinched-off vesicles, and (E) modified exocytic secretion in hemipteran midgut cell. Abbreviations: BSV, budding secretory vesicle; CE, cellular extrusion; DSV, double-membrane secretory vesicle; GC, Golgi complex; M, microvilli; N, nucleus, PMM, perimicrovillar membrane; PSV, pinched-off secretory vesicle; RER, rough endoplasmic reticulum; SV, secretory vesicle.
Secretion by hemipteran midgut cells displays special features. Double-membrane vesicles bud from modified (double-membrane) Golgi structures (Fig. 7E). The double-membrane vesicles move to the cell apex, their outer membranes fuse with the microvillar membrane, and their inner membranes fuse with the perimicrovillar membranes, emptying their contents (Fig. 3H).
