Diapause is a form of developmental arrest in insects that is much like hibernation in higher animals. It enables insects and related arthropods to circumvent adverse seasons. Winter is most commonly avoided in temperate zones, but diapause is also used to avoid hot, dry summers and periods of food shortage in the tropics. Unlike quiescence, which represents a halt in development elicited immediately at any stage by an adverse condition, diapause is a developmental response that is expressed only during a specific developmental stage, which depends on the species of insect. For example, the commercial silkworm (Bombyx mori) always diapauses as an early embryo, the European corn borer (Ostrinia nubi-lalis) as a fifth instar, the cecropia moth (Hyalophora cecropia) as a pupa, and the Colorado potato beetle (Leptinotarsa decemlineata) as an adult. A few species are capable of entering diapause several times, but this usually occurs only in species living at high latitudes for which several years may be required for the completion of development. If the diapause occurs in response to environmental cues it is referred to as “facultative diapause,” but if it occurs during each generation regardless of the environmental cues it receives, it is considered an “obligatory diapause.” Facultative diapause is by far the more common, but several important species such as the gypsy moth (Lymantria dispar) have an obligatory diapause.
Embryonic diapauses are common in many of the Lepidoptera, in many Hemiptera, and in some Diptera such as mosquitoes. The arrest can occur at any stage of embryonic development, from shortly after fertilization (e.g., commercial silkworm) until after the first instar has already been fully formed (e.g., gypsy moth). Larval diapauses, especially common in the Lepidoptera, are most frequent in the final instar but they sometimes occur in earlier instars as well, for example, the second instar of the spruce budworm (Choristoneura fumiferana) . Pupal diapause is well known for the Lepidoptera and Diptera. Usually the arrest of pupal development occurs in the true pupal stage, but there are a few examples of diapause occurring in pharate adults (after completion of adult differentiation but before adult eclosion). Adult diapause is common in the Coleoptera, Hemiptera and Homoptera, Hymenoptera, Orthoptera, and Neuroptera, as well as some Diptera and Lepidoptera. Adult diapause, sometime referred to as a reproductive diapause, represents a halt in reproduction. Ovaries of females remain small, and the oocytes within the ovarioles contain little or no yolk. In males of some species, the testes remain small during diapause, but in others the testes are well developed and contain sperm. Male accessory glands, the organs that produce spermatophores and factors responsible for sperm activation, usually remain small and inactive during diapause. Mating behavior is strongly suppressed during diapause. In wasps, mating takes place in the autumn; males die soon thereafter and only the females overwinter in diapause. In many other insects, both sexes overwinter and mating takes place in the spring, after diapause has been terminated. Some species, such as lacewings and weevils, mate both before and after diapause.
In preparation for diapause, the insect usually sequesters additional energy reserves and moves to a site that is somewhat protected from the full onslaught of the inclement environmental conditions. Such sites may be underground, beneath debris on the soil, within galls and other plant tissues, or inside cocoons or other structures constructed by the insect. A migratory flight may be a preparatory step for diapause. This may include a short flight to a fence row or a local wooded area, but in the extreme it may be a long-distance flight, as made by the monarch butterfly (Danaus plexippus) when it leaves its summer habitat in Canada and the northern regions of the United States and flies to the highlands of Mexico or California to spend the winter in an adult diapause.
Upon entering diapause, development (or reproduction if it is an adult diapause) is halted and metabolic activity is suppressed. Usually, feeding ceases during diapause; thus, the insect is forced to survive on the energy reserves it has garnered prior to the onset of diapause. It is not unusual for an insect destined for diapause to sequester twice as much lipid reserves as its counterpart that is not programmed to enter diapause. The economic utilization of these reserves is enhanced by the suppression of metabolism, and for poikilotherms such as insects, the low temperatures prevailing during winter further serve to conserve energy reserves. Another challenge faced by diapausing insects is the lack of access to free water. Although some insects may drink during diapause, certain stages such as embryos and pupae do not have this option. This lack of water poses special constraints for an organism as small as an insect. Their large surface-to-volume ratios make insects particularly vulnerable to water loss across the surface of their integument. Two features appear to be common adaptations for maintaining water balance during the long months of diapause. The cuticles of many diapausing insects are coated with extra thick layers of wax that are effective in retarding water loss. In addition, a number of diapausing insects are capable of absorbing atmospheric water vapor directly through their cuticle using a mechanism that is not yet clearly understood.
Color changes are sometimes noted for diapausing individuals. For example, diapausing larvae of the southwestern corn borer, Diatraea grandiosella, are white, whereas their nondiapausing counterparts are brown. Reproductively active adults of a lacew-ing, Chrysopa carnea , are green but turn brown when they enter diapause in the autumn. In the spring, when the lacewings become reproductively active, they again turn green. Such changes presumably serve to camouflage the insect and help it blend with the dominant colors of the seasonal environment.
Flight muscles in many beetles and bugs degenerate when the adults enter diapause. Flight muscles are particularly expensive to maintain, thus their degeneration presumably saves energy that would otherwise be expended for maintenance of this tissue.
Several species that diapause as adults, especially beetles, bugs, and butterflies, are found in aggregations. For species that are distasteful, aggregations are likely to provide protection from predators. Such aggregations, however, may also provide another important function by providing a more stable microenvironment. In diapaus-ing aggregations of a tropical fungus beetle, Stenotarsus rotundus, the beetle’s metabolic rate is inversely related to group size and relative humidity. By forming an aggregation the beetles create a stable, high humidity in their environment, a feature that serves to reduce metabolic rate.
Being in diapause does not, by itself, ensure winter survival. The small size of insects implies that they quickly assume a body temperature close to that of the environment, and their body water is thus vulnerable to freezing. Diapausing insects that live in temperate and polar regions have a host of behavioral, physiological, and biochemical adaptations that enable them to survive at low temperature. A few insects such as the goldenrod gall fly, Eurosta solidaginis,
are freeze tolerant, which implies that they can actually survive body freezing. But, the majority of insects cannot tolerate body freezing. Such freeze-intolerant or freeze-avoiding insects prevent body freezing by several mechanisms. For example, selection of a thermally buffered microhabitat is a first line of defense. Ice nucleators such as food particles or microbes are usually eliminated from the digestive tract to reduce sites for ice formation. Glycerol, sorbitol, or other polyols serve as classic antifreezes that are synthesized and released into the body to suppress the supercooling point. Several proteins, including thermal hysteresis proteins, ice nucleator proteins, and heat-shock proteins, also contribute to cold hardiness. In some insects, such as flesh flies (Sarcophaga), cold hardiness is directly linked to diapause, indicating that the same genetic program that dictates diapause also results in cold hardiness. In other insects, for example, the European corn borer, the two programs are regulated independently: the European corn borer enters diapause without initially being cold hardy, but it becomes cold hardy later in the season in response to prevailing low temperatures.
Diapause thus represents a syndrome of developmental, physiological, biochemical, and behavioral attributes that together serve to enhance survival during seasons of environmental adversity.
ENVIRONMENTAL REGULATION
Obligatory diapause is not elicited by environmental cues. It simply occurs in each generation when the insect reaches a certain developmental stage. In the example of the gypsy moth, diapause occurs when the embryo has completed its development and the first instar is nearly ready to hatch. With the exception of a few aberrant individuals, the gypsy moth always halts development at this time, regardless of the environmental cues they receive. In this example, environmental conditions, mainly temperature, determine when diapause should be terminated but play no role in programming the insect to enter diapause.
This is in contrast to the majority of insects, those with a facultative diapause, which use environmental cues to decide whether to enter diapause. If a certain environmental cue is received during a sensitive period the insect will enter diapause, but if this cue is not received or not received at the correct time, development will proceed without interruption. This design feature enables an insect to track seasonal changes and regulate its development accordingly. Many insects can produce multiple generations each year, and insects with a facultative diapause frequently produce spring and summer generations without diapause and then produce a generation in late summer or autumn that enters an overwintering diapause. The environmental cue used most widely to signal diapause induction is photoperiod, but temperature, food quality, and other factors may contribute to the decision.
Photoperiod
Seasonal change in daylength has all the design features that are desirable in a reliable indicator that can be used for predicting upcoming periods of inclemency. It is mathematically accurate and can be used to effectively foretell the advent of winter or other seasons that are to be avoided. The developmental period that is sensitive to photoperiod usually occurs far in advance of the actual diapause stage. Thus, diapause is not usually an immediate reaction to photoperiod but occurs in response to signals received at an earlier stage. Such early programming offers the insect a period to prepare for diapause by sequestering food reserves and making other preparatory adjustments prior to the actual onset of the developmental arrest.
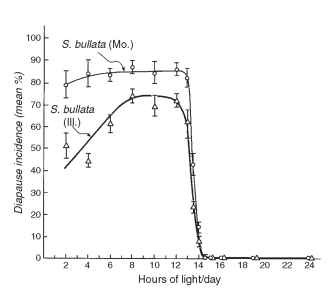
FIGURE 1 Photoperiodic response curves for pupal diapause induction in two populations of the flesh fly, Sarcophaga bullata, from Illinois and Missouri. Fly cultures were maintained at 25°C under the range of daylengths indicated, and the incidence of pupal diapause was recorded. The critical daylength in this case is 13.5 h of light/day.
For many of the insects that overwinter in diapause in the temperate regions, short daylengths dictate the expression of diapause. In the example shown in Fig. 1, flesh flies reared at long daylengths, those longer than 13.5 h, develop without interruption, but at daylengths shorter than 13.5 h, the majority enters diapause as pupae. The daylength marking the transition, 13.5 h in this example, is referred to as the critical daylength. The shape of the photoperiod response curve shown in Fig. 1 is common for temperate species that overwinter in diapause, but the curves may have different forms. For some species, especially those that undergo a summer estivation and reproduce in the autumn, long rather than short daylengths may be used to program diapause. Other species may respond to only a narrow range of daylengths for diapause induction, whereas daylengths both shorter and longer avert diapause.
Near the equator, seasonal changes in daylength are progressively less pronounced; yet insects living as close as 5° north or south of the equator are still capable of using photoperiodic cues to regulate diapause. Diapause still exists in insects living in equatorial regions, but cues derived from temperature, rainfall, and food quality take precedence over photoperiod.
The photoperiodic response controlling diapause varies among geographic populations. Populations living at lower latitudes characteristically respond to shorter critical daylengths. An increase in latitude of 5° results in an increase in critical daylength of approximately 30 min. This pattern of variation is closely related to the latitudinal temperature gradient and is well documented in species of Drosophila that inhabit the Japanese archipelago. The species that occur in the subtropical zone exhibit only a weak diapause or no diapause at all. As one moves northward in the archipeligo, the diapause response becomes more pronounced and the flies use longer critical daylengths for diapause induction.
The period sensitive to photoperiod usually does not encompass the entire prediapause period, but instead a shorter interval, usually well in advance of the actual diapause stage. For example, the pupal
diapause in the flesh fly, Sarcophaga crassipalpis, is programmed during a photosensitive stage that includes the final 2 days of embryonic development and the first 2 days of larval life. In the tobacco hornworm, Manduca sexta, a species that also has a pupal diapause the photosensitive stage is much longer; it begins during embryonic development and continues through the feeding phase of the fifth instar. In the silkworm, B. mori, embryonic diapause is programmed during the mother’s period of embryonic development. This timing of the photosensitive stage thus facilitates the channeling of development toward diapause at an early stage and allows sufficient time for the preparative phase of diapause.
The duration of diapause, often called diapause depth, also may depend on photoperiod. For example, in the lacewing, C. carnea, diapause depth is controlled by photoperiod in such a way that the adult diapause is deeper when it is induced earlier in the autumn, thus preventing an untimely termination of diapause before the onset of winter. And, in the tobacco hornworm, M. sexta, the duration of pupal diapause is a function of the number of short days the embryo and larva have received. Exposure to a few short days, such as would occur in mid- to late summer, results in a long diapause, while exposure exclusively to short days, an event that could occur only in early autumn, results in a short diapause. Such qualitative responses to photoperiod allow the insect to fine tune its development to fit the changing season.
Photoperiodic information is perceived through a receptor in the brain, integrated and stored in the brain, and then translated into the endocrine events that control the induction and maintenance of diapause. The location of the photoreceptor responsible for measurement of daylength has been studied in relatively few insects, but in most of them the compound eyes and ocelli are not the conduit for this information. Surgical destruction of these visual centers or coating the eyes with an opaque paint usually does not interfere with the photoreception involved in the programming of diapause. The photoperiodic signal appears to impinge directly on the brain, but the exact location of these extraretinal photoreceptors has not been elucidated. As in many other plants and animals, the photoperiodic response in insects is primarily a blue-light response. Cryptochromes, proteins involved in photoperiodic responses in a diverse array of organisms, are present in insects and are likely to be implicated in this response. Several important clock genes, including period and timeless, have been identified in insects, and they are emerging as important players in photoperiodism.
The role for photoperiod in the environmental regulation of diapause is mainly in the inductive phase of diapause. There are a few species that use daylength as a direct environmental cue for diapause termination. More commonly, photoperiod may influence the rate of diapause development, which in turn does impact the duration of diapause, but frequently diapause development proceeds at a rate determined by temperature rather than photoperiod.
Temperature
Temperature provides another important seasonal cue for diapause induction, but the daily fluctuations in temperature mean that it is less reliable than photoperiod in this regard. Frequently, a short-day response is enhanced by low temperature. For example, the maximum diapause response observed for flesh flies shown in Fig. 1 is approximately 80%. But, this was for flies reared at 25°C, and if the temperature is lowered to 18°C, the diapause incidence is elevated to nearly 100%. In these flies the critical photoperiod is not influenced by temperature, but in some insects the critical photoperiod may shift as well.
Near the equator, where seasonal changes in daylength are too subtle to be used as environmental cues, temperature may replace photoperiod as the primary environmental regulator of diapause, as it does for flesh flies living in East Africa: daylength has no influence on the expression of diapause, but instead low daytime temperatures experienced in July and August are used to program the flies for pupal diapause.
A period of chilling may be essential for diapause termination. Diapausing insects often cannot resume development or reproduction immediately upon transfer to favorable conditions but require a period of chilling. Although some insects do not absolutely require chilling before initiating development, many will terminate diapause more quickly if they have first been chilled for a few months.
HORMONAL REGULATION
The juvenile hormones (JHs) and ecdysteroids, two of the major families of insect hormones that direct insect development, metamorphosis, and reproduction, are intimately involved in regulating diapause. The JHs, which are isoprenoids secreted by the corpus alla-tum (CA), maintain the juvenile characters during the premetamor-phic molts, while the steroid hormones from the prothoracic gland (PG), ecdysone and related compounds, dictate the decision to molt. In turn, the CA and PG are regulated by both neural and humoral factors from the brain. Brain neuropeptides governing the CA can exert either a stimulatory (allatotropins) or an inhibitory action (alla-toinhibins) on the CA. The dominant regulator of the PG is the brain neuropeptide prothoracicotropic hormone (PTTH). These hormones, together with diapause hormone (DH), a unique neuropeptide that regulates the embryonic diapause of the commercial silkworm, are the key hormonal regulators of insect diapause. In certain situations, the presence of one or more of these hormones promotes diapause, while in others it is the absence of a certain hormone that causes diapause.
Embryonic Diapause
The best-understood hormonal mechanism regulating embryonic diapause is based on the silkworm. In this species diapause intercedes early during embryogenesis, just before segmentation. The developmental fate of the embryo is determined by the presence or absence of DH, a neuropeptide secreted by the mother’s subesophageal ganglion. In the presence of DH, the ovariole produces eggs that enter diapause, and when the hormone is not present the eggs develop without the interruption of diapause. Whether the mother releases DH is dependent upon the photoperiod she was exposed to as an embryo. Thus, the mother’s photoperiodic history dictates whether she will release the DH needed to influence the diapause fate of her progeny.
The structure of DH has been defined, as well as the sequence of the cDNA that encodes the peptide. DH appears to exert its effect on diapause by influencing carbohydrate metabolism. In the presence of DH, the developing oocytes incorporate glycogen stores, which in turn are converted to sorbitol. Sorbitol was originally thought to function simply as a cryoprotectant, but recent work suggests that sorbi-tol may actually be involved in shutting down development in the embryo. The addition of sorbitol to an embryo that is programmed to develop without diapause elicits a developmental arrest; in contrast, the removal of sorbitol from diapause-programmed embryos enables the embryos to develop without diapause.
No other diapauses appear to rely on DH for diapause induction. DH appears to be a hormone uniquely used by the silkworm for this purpose. In the gypsy moth diapause occurs at the end of embryogen-esis, just before hatching of the first instar. The diapause of this species appears to be regulated by maintenance of a high ecdysteroid titer. As long as the ecdysteroid titer remains high, the pharate first instar remains locked in diapause. Only when the ecdysteroid titer drops in the spring is the gypsy moth free to terminate its diapause and hatch. Yet another mechanism seems to operate in the giant silkmoth, Antheraea yamamai. In this insect, an unidentified repressive factor from the mesothorax inhibits the action of a maturation factor from the abdomen. The fact that all three species that have been examined display different endocrine control mechanisms suggests a wealth of mechanisms operating in the regulation of these early stage diapauses.
Larval Diapause
Larval diapause frequently intercedes at the end of larval life, just before the onset of pupation and metamorphosis, but it is not at all uncommon in earlier instars as well. Common to most examples of larval diapause is a shutdown in the brain-PG axis. In the absence of ecdysteroids from the PG, the larva fails to initiate the next molt. The failure of the brain to release ecdysteroids can usually be directly attributed to the brain’s failure to release PTTH. In a number of species, JH may also play a role. For example, in the southwestern corn borer, D. grandiosella, the JH titer remains elevated throughout diapause, and the diapause can be terminated only when the JH titer drops. In some other species such as the European corn borer, O. nubilalis, the JH titer is high in early diapause but then declines and remains low throughout the remainder of diapause. No role for JH is apparent in several other insects: the larval diapause of both the parasitic wasp, Nasonia vitripennis, and the blow fly, Calliphora vicina, can be explained strictly as an ecdysteroid deficiency.
Pupal Diapause
Pupal diapause is the consequence of a shutdown in the brain-PG axis. Thus, in the absence of ecdysteroids from the PG the progression of adult differentiation is halted. At the termination of diapause, ecdysteroids are again released, triggering adult development. In H. cecropia a period of chilling is required before the brain can stimulate the PG to release ecdysteroids. Pupal diapauses can usually be quickly terminated with an injection of 20-hydroxyecdysone. Usually the absence of ecdysteroids can be attributed directly to a failure of the brain to release the neuropeptide PTTH needed to stimulate the PG to synthesize ecdysteroids, but in some insects, for example, Helicoverpa zea, PTTH is released shortly after pupation, but pupa fail to develop until the PG has been chilled adequately. Recently, it was shown that the pupal diapause of moths in the agriculturally important Heliothis—Helicoverpa complex also can be terminated with DH, the hormone known to induce silkworm diapause. Thus, DH elicits two quite opposite effects on diapause in different moth species.
Adult Diapause
A shutdown in JH synthesis is a key feature in the regulation of adult diapause. The corpora allata, the endocrine glands that synthesize and release JH, are characteristically small during diapause. Application of exogenous JH or implantation of active corpora allata into a diapausing individual usually prompts the termination of diapause. Conversely, the surgical extirpation of the corpora allata from a nondiapausing adult causes the adult to enter a diapauselike state. Measurement of the JH titer also supports the idea that adult diapause is the consequence of a shutdown of the corpora allata: the titer of JH typically drops as the insect enters diapause and increases again when diapause is terminated.
It is the brain that regulates the corpora allata, and both nervous and humoral pathways are involved in its regulation. In the Colorado potato beetle, the brain exerts its control over the corpora allata by a humoral mechanism, but in the linden bug, Pyrrhocoris apterus, nervous control is also involved. Ecdysteroids may also be involved in some species. The ecdysteroid titer is nearly twice as high in Colorado potato beetles destined for diapause than in those that are not destined to enter diapause, and an injection of ecdysteroids can terminate adult diapause in Drosophila melanogaster.
MOLECULAR MECHANISMS
The environmental cues that regulate diapause have been well defined, and there is also a fairly good understanding of the downstream hormonal signals that serve to coordinate diapause. But, the molecular underpinning of diapause remains poorly understood. Is diapause simply a shutdown in gene expression or does it represent the expression of a unique set of genes? An examination of the synthesis of brain proteins in flesh flies suggests that far fewer proteins are synthesized in the brain during diapause but, in addition, the brains of diapausing flies synthesize a set of proteins not observed in brains of nondiapausing flies. This suggests that diapause represents both a shutdown in gene expression and the expression of a unique set of genes.
One of the most conspicuous groups of genes that are diapause upregulated is that of the heat-shock proteins. Both heat-shock protein 70 (Hsp70) and one of the small heat-shock proteins (Hsp23) are upregulated in flesh flies during diapause. The Hsps are upregulated upon entry into diapause, remain elevated throughout diapause, and then drop sharply at diapause termination. When expression of the Hsps is knocked down using RNA interference, cold tolerance of the diapausing pupae is lost, suggesting that the Hsps offer protection from environmental stresses during diapause.
Genes that are diapause downregulated are potentially of equal interest. Among the genes in this category is the gene that encodes proliferating cell nuclear antigen, a cell-cycle regulator. The downregu-lation of this gene during diapause may be important in bringing about the cell-cycle arrest. As more genes are examined, it is evident that certain genes are expressed throughout diapause, others are turned off during diapause, while still others are expressed only during early or late diapause or may be expressed intermittently during diapause.
It is still too early to know if common sets of genes are expressed during diapause in different species and different life stages, but preliminary data suggest that the expression patterns of at least some of the genes, those that encode Hsp70, may be shared across species and life stages. Likewise, insulin signaling is emerging as a signaling pathway that is likely to be commonly involved in insect diapause as well as in dormancies of other invertebrates.
