ABSTRACT
Hydroxyapatite (HAP) displays very excellent biocompatibility in the body and is for the most part used in biomedical applications thanks to well biocompatibility for replacement of bone. In fact, Boron and Ti containing HAP are the bioactive materials and it can incorporate into bone structures, supporting bone in-growth without breaking down or dissolving, and it interacts with the living tissue due to the presence of free calcium and phosphate compounds. Boron containing HAP are also extensively useful for the manufacturing of bio-ceramics in order to improve the physical and chemical properties of biomaterials. Boron nowadays used in HAP applications is a very successful candidate material for bioceramic engineering. Generally, Al2O3 powder is added to HAP powder in order to obtain high fracture toughness. Al2O3 has good mechanical properties as compared with HAP, and exhibits extremely high stability with human tissues. In this paper, the effect of microwave sintering temperature on the relative density, hardness, and phase purity of compacted bovine Hydroxyapatite (BHA) powder was reported. This research is a comprehensive attempt to develop Hydroxyapatite bio composite ceramics reinforced with alumina – Al2O3, pure metallic titanium and pure pulverised boron powders. A Finite Element (FEM) analysis is also used to simulate the macroscopic behaviour of this material, taking into account the relevant microscopic scales. Generally, microwave-sintered samples showed much small grain size and a uniform microstructure. For this reason, the behaviour of bio ceramics in case of rapid heating in microwave was also discussed. Recent results revealed that microwave processing was a promising method for sintering porous bio ceramics thanks to clean and shorter sintering time regarding to conventional sintering methods.
Keywords: HAP Bio ceramics; Microwave sintering; FE analysis; Interface; Boron; Titanium
Introduction
Biomaterials are designed and fabricated for implantation within or incorporation with a living system to replace or repair the living tissues or organs which form an interface between the living and non-living parts [1, 2]. Biomaterials are based on polymers, metals, ceramics, or composites of these [3]. Hydroxyapatite (HAP), Cai0(PO4)6(OH)2 is a very well known phosphate in the biologically active phosphate ceramic family by virtue of its similarity to natural bone mineral. Synthetic and naturally produced hydroxyapatite finds a variety of biological applications and brings out the formation of an apatite layer at the interface with bone tissue [4, 7, 8]. This property makes HAP extremely attractive and widely used as a material for bone implants. Nevertheless, fracture strength and toughness of this ceramic are low, and it cannot endure practical use. For this reason, HAP is usually reinforced with various biocompatible materials that can improve the strength of the pure HAP. Titanium is a successful biocompatible material that is extensively used for biomedical applications, especially for bone-anchoring systems, such as dental, orthopaedic implants and osteosynthesis applications. It has advantageous bulk mechanical properties such as a low modulus of elasticity, a high strength-to-weight ratio, and passive surface properties i.e. excellent corrosion resistance and low rates of ion release as well as a high degree of biocompatibility which is largely attributed to an inert surface oxide film. Alumina, another material used to make implantable orthopaedic devices, is a very well tolerated material with minimum tissue reaction after implantation. It exhibits high mechanical strength and minimum wearing. Therefore, it is frequently used in high load-bearing sites such as hip prostheses and dental implants [3-8, 10-16].
Additionally, Paraffin is very successful additive in HAP for creating micro porosity in the structure. During the heating, Paraffin is burned and leaved its place as micro porosity homogeneously distributed in the structure. Paraffin was used in this study as a well substitution of naphthalene that is irritant material. Concerning borohydroxyapatite compounds (Boron containing HAP), they are extensively useful for the manufacturing of bio-ceramics in order to improve the physical and chemical properties of biomaterials. Boron very often used in HAP application, is a very successful candidate material for bioceramic applications. Recent scientific researches indicate that Boron will be more useful with other materials in future bioceramic applications.
In this paper, a net shape microwave sintering procedure was used in the frame of the "Bioceramic" research project by using pure metallic titanium and pure pulverised boron powders and a few percent of paraffin to create a micro porous structure that was well replaced the other materials to create porous structure. A special attention was given for the microstructural evolution with recently developed compositions to give practical significance for the application of the biomaterials.
Experimental study
In this preliminary study, a new microwave sintering method (lower energy costs and shorter processing times) has been carried out for manufacturing of bio composite materials that all the details were given in former paper [2]. The compacts geometry was prepared based on the fixed matrix natural HAP + 10% alumina (Al2O3) reinforced with pure metallic titanium and pure pulverised boron with Yttrium and Zirconia (VWR-France). Before process, alumina was doped with MgO (1%). Then, the blended powders homogenized by ball milling during one hour were compacted by uniaxial cold isostatic pressing with a green compact pressure of 300 MPa, intending to produce an initial green density ranging 90%. The aspect ratio of this geometry was between 0.9-1. Cylindrical test specimens were prepared (Height=11mm, Diameter=11mm) according to the British Standards-BS 7253, and then sintered for a short time (up to 30 min) in microwave oven. At the second stage, micro hardness and quasi static compression tests were performed on the sintered samples to study the influence of microstructure and phase composition on the micro-mechanical behaviour of the recently formed compositions. For etching, acetic and lactic acids were used for microstructural surface analysis. For evaluation of fracture surfaces of the specimens, failed during the compression test have been analysed in Scanning Electron Microscope and chemical analysis by "EDS" analysis. Some of the specimens were analyzed by XRD to distinguish the possible phases that can be occurred in high temperatures during sintering. Experimental results were used for a Finite Element (FEM) analysis to create a simple and for an easy understandable model the behaviour of the bioceramic materials during the deformation.
Microwave – Sintering
The application of microwave energy to the processing of various materials such as ceramics, metals and composites offers several advantages over conventional heating methods. Microwave heating results in lower energy costs and decreased processing times for many industrial processes. These advantages include unique microstructure and properties, improved product yield, energy savings, reduction in manufacturing cost and synthesis of new materials. At the beginning of the test, some of the specimens were sintered in an electrical – conventional furnace (High Temperature Furnace) in order to compare the microstructural evolution regarding to the sintered specimens in microwave oven. In this project, the primary aim, however is to use a house type microwave oven in laboratory conditions. For this reason, a house type (2.45 GHz) microwave oven was used by converting some of the conditions of the microwave oven. Temperature measurement system was adapted to the microwave by using a special thermocouples covered with safety insulating cables to prevent the electrical contact of the thermocouples when microwave works. The accuracy of temperature measurement with this device was determined to be within -10°C of the temperature measured. The thermo couples were inserted inside of the alumina crucible that it was very close (2 -3 mm) to the specimens. All of the details for this sintering method in former papers [6, 7, 9]. Compared to the conventional sintering, there was a slight increase in density of the specimens sintered using microwave conditions. Since the microwave sintering took much shorter time, densification rate in the microwave sintering process may be considered to be higher than in the conventional sintering. For effective sintering time for these samples were chosen 20 minutes.
Results and Discussion
Experimental part
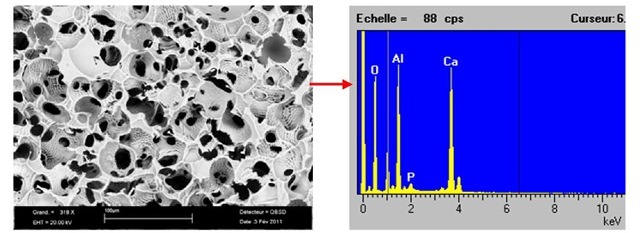
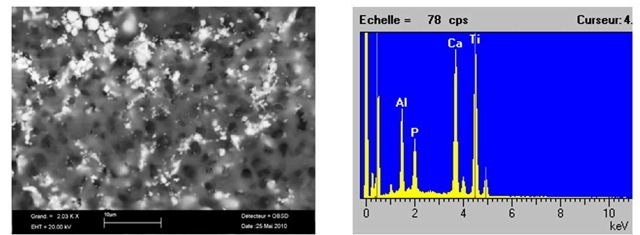
Natural HAP was obtained from calcinated fresh - young bovine bones by following the method developed formerly in SUPMECA/LISMMA-Paris [6, 7-8]; after cutting, the femurs were undergone deproteinized with NaOH treatment. After repeated washing, they were heat treated at 850°C. The treated HAP powder (particle sizes of 1-2^m) was one by one mixed in different percentages. Figures 1 and 2 show the microstructure natural HAP taken from the SEM image with "EDS" analysis, containing 5% and 10% pure pulverised metallic boron powder that was distributed homogeneously in the structure. The average grain size measured of this mixture varies from 1 to 5^m. This structure verifies that the microwave sintering gives a clean, lower energy costs and shorter processing times for these materials.
Fig. 1a. Microstructure of the sample containing only 5%B
Figure 1b shows the distribution map (SEM-cartography) of main elements resulting from Figure 1a. The diffusion of Alumina and Calcium show very uniform distribution, it means that much more improvement has been carried out in the compositions containing boron powder from 5% volume fraction. In reality, alumina shows an easy diffusion even if it does not show high thermodynamic tendency. Doping of Alumina with MgO before sintering process facilitates to react easily with HAP matrix. Looking at the illuminated areas in the back-scattering (BS) SEM images of Figure 1b, it is noted that the Alumina-associated phases were uniformly distributed in the bulk of composite materials.
Fig.1b. Distribution map (SEM) of main elements resulting from Figure 1a (sample containing 5%B)
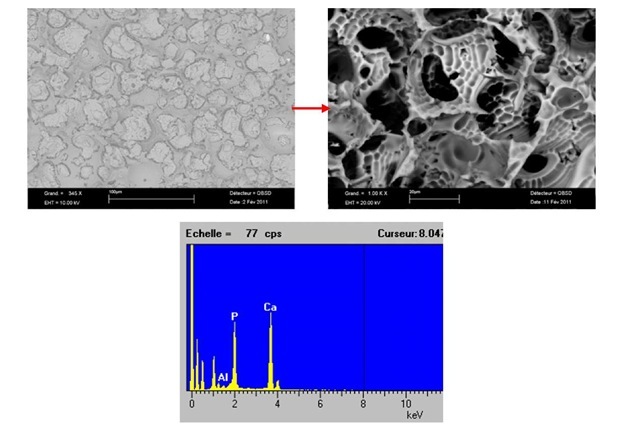
Fig. 2. Microstructure of the sample containing only 10%B
The SEM images shown in the Figures 3 and 4 indicate SEM micrograph of the sample containing 20%B+15%Ti and 20%B+20%Ti respectively. Very perfect sintering has occurred with these composition with very hard structure (see Table 1), and also the formation of certain amount of a glassy phase was identified. Ti always shows a high thermodynamic tendency to diffuse and then to react with HAP matrix easily.
Fig. 3. SEM micrograph containing 20%B+15%Ti+3%Yi203+3%Zr02
Fig. 4. SEM micrograph containing 20%B+20%Ti+3%Yi203+3%Zr02
The phases containing titanium elements were distributed uniformly in the structure even if there are an excess of Ti at the grain boundaries. Finally, a well fusion was observed in the structure and some of the specimen showed a regular micro porosity in the matrix. The diffusion of Ti is much more even inside the matrix compared to Al. As explained in the section of experimental conditions, these compositions have been processed with Yttrium and Zirconium. It seems that both of these elements give a fine and stable structure and noticeable effect on the homogenous distribution of the additional elements in the structure.
More interesting structure has been obtained in the samples containing 40%B having a very high hardness values. They have been processed with natural HAP and they were stabilised by doping with 1% MgO. However, many of the specimens with this composition showed microcracks and also micro porosities in the structures after sintering. From these Figures, it is concluded that the samples containing boron were considerably crystallized at all boron containing compositions. In fact, all the compositions given in this paper indicate that the densification was highly improved. Recently, to evaluate of the machining of the samples containing boron, some of the samples were machined by drilling and turning. The preliminary results have shown that manufacturing of these materials in different shape is possible; they do not show brittleness during machining that have been evaluated in former study [17].
Fig. 5. SEM Micrograph and fracture surface of the sample containing only 40%B
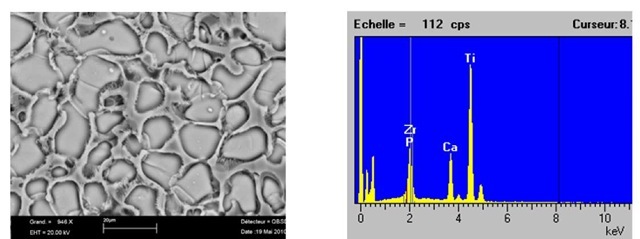
Figure 6 shows SEM Microstructure of the samples containing a) 15%TiO2 and b) 20%Ti02 respectively. These samples have been manufactured for obtaining a micro porous structure by adding 10% paraffin and 3%Yi2O3+3%ZrO2).
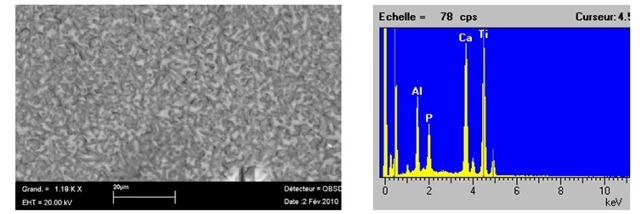
In practical point of view, additions of Yi2O3 and ZrO2 stabilise the microstructure by making fine grain and homogeneous structure. By using paraffin, the natural micro porosity is greatly improved (up to 20%) and homogenous distribution of the natural micro porosity in the structure. It means that an artificial porosity was well created by using Similar results have been obtained in the structures given in Figure 7 that indicates SEM Micrograph of the sample containing 20%Ti02 +10% paraffin+3%Yi2O3+3%ZrO2.
Paraffin wax has a very smooth effect on the size and distribution of the artificial porosity. These results original and gives successfully new results on the bio ceramics containing pure Ti and pure Boron with additive of paraffin.
Fig. 6. a) Microstructure of the sample containing a) 15%Ti02+10% paraffin and b) 20%Ti02+10% paraffin
Experimental results of compressive strength and microhardness of the samples with different compositions (volume fraction) were summarised in Table 1.
At least five specimens were tested for each composition and mean values have been considered. Compressive strength values of high boron containing (>10%) are better than the other composition. For the sake of simplicity, only limited experimental results were used for a FEM study has been carried out to give a practical idea for the manufacturing of bioceramics. Naturally, we could not give more detail on the FEM carried out in the frame of this project going on due to limited page of the SEM proceedings
Fig. 7. SEM Microstructure of the sample containing 20%Ti02 +10% paraffin+3%Yi2O3+3%ZrO2 3.2. A microstructural model and FE simulations
The idealized microstructure considered in this study consists of a random arrangement of cylindrical inclusions embedded in a continuous HAP bio composites, matrix. The volume fraction of inclusions is varied from 10% to 20% and the micro-macro transition schemes are evaluated in many case. However, when the volume fraction increases, nearby inclusions start to interact and this affects the overall mechanical behaviour. The generation of the random distribution follows cylinders filled with identical and aligned cylindrical inclusions. The RVE microstructure is periodic along the 3 directions, allowing us to apply periodic boundary conditions to the external faces of the specimens. The inclusion positioning is constrained by the practical limitation of generating an acceptable FE mesh. A criterion is applied to the minimal distance between each inclusion surface and the external faces of the specimen. The volume of one particular cell inclusion is less than 1 mm3.
The representative cells are meshed with quadratic tetrahedral. FE simulations are performed using ABAQUS (2008) and the whole volume is meshed using 4-node C3D4 tetrahedral in ABAQUS), enabling us to better capture the strain gradients in the matrix. Convergence study was successfully conducted by comparing the predictions (effective response and average inclusion response) to those obtained with finer meshes. Figure 8a show typical meshes for a composite with various % of inclusions and variable geometry of specimen are used. The macroscopic stress predicted by the FE analysis is computed from a volume average of the stress tensor given at each integration point over the RVE of domain i: