Abstract
This entry deals with biomass as an energy source. Different types of biomass are described from the energy perspective, focusing on those more interesting for energy application. The main energy conversion technologies available are outlined, as well as the properties of their main products. Finally, an overview over the benefits that come from biomass exploitation for energy purposes is provided.
INTRODUCTION
This work is organized into seven main sections. The first paragraph provides the reader with a general overview on biomass, including the definition, environmental benefits, energetic properties, and a short list of biomass types that can be used as energy sources. The second paragraph illustrates the mechanical processes needed to produce standardized solid biomass fuels. The third paragraph describes one of the major technologies for converting biomass into energy, which is combustion. The fourth paragraph analyzes pyrolysis and gasification as promising techniques for efficient exploitation of biomass still in a demonstration phase. The fifth paragraph is concerned with biochemical processes for producing biogas and biofuels for transportation. The sixth paragraph outlines the major benefits from biomass exploitation for energy purposes. The seventh paragraph is constituted by the concluding remarks.
GENERALITIES ABOUT BIOMASS
In general, biomass is the substance produced or by-produced by biological processes.
Commonly, biomass refers to the organic matter derived from plants and generated through photosynthesis. Biomass not only provides food but also construction materials, fibers, medicines, and energy. In particular, biomass can be regarded as solar energy stored in the chemical bonds of the organic material. Carbon dioxide (CO2) from the atmosphere and water absorbed by the plants roots are combined in the photosynthetic process to produce carbohydrates (or sugars) that form the biomass. The solar energy that drives photosynthesis is stored in the chemical bonds of the biomass structural components. During biomass combustion, oxygen from the atmosphere combines with the carbon and hydrogen in biomass to produce CO2 and water. The process is therefore cyclic because the carbon dioxide is then available to produce new biomass. This is also the reason why bioenergy is potentially considered carbon-neutral, although some CO2 emissions occur due to the use of fossil fuels during the production and transport of biofuels.
Biomass resources can be classified according to the supply sector, as shown in Table 1.
The chemical composition of plant biomass varies among species. Yet, in general terms, plants are made of approximately 25% lignin and 75% carbohydrates or sugars. The carbohydrate fraction consists of many sugar molecules linked together in long chains or polymers. Two categories are distinguished: cellulose and hemi-cellulose. The lignin fraction consists of non-sugar-type molecules that act as a glue, holding together the cellulose fibers.
The Energy Content of Biomass
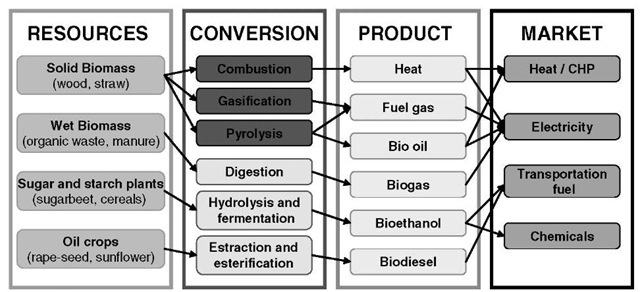
Bioenergy is energy of biological and renewable origin, normally derived from purpose-grown energy crops or by-products of agriculture. Examples of bioenergy resources are wood, straw, bagasse, and organic waste. The term bioenergy encompasses the overall technical means through which biomass is produced, converted, and used. Fig. 1 summarizes the variety of processes for energy production from biomass.
The calorific value of a fuel is usually expressed as the higher heating value (HHV) or the lower heating value (LHV). The difference is caused by the heat of evaporation of the water formed from the combustion of hydrogen in the material and the original moisture. Note that the difference between the two heating values depends on the chemical composition of the fuel.
The most important property of biomass feedstocks with regard to combustion—and to the other thermochemical processes—is the moisture content, which influences the energy content of the fuel. Wood, just after falling, has a typical 55% water content and LHV of approximately 7.1 MJ/kg; logwood after 2-3 years of air-drying may present 20% water content and LHV of 14.4 MJ/kg; pellets show a quite constant humidity content of about 8% with LHV equal to 17 MJ/kg.
MECHANICAL PROCESSES FOR ENERGY DENSIFICATION
Some practical problems are associated with the use of biomass material (sawdust, wood chips, or agricultural residues) as fuel. Those problems are mainly related to the high bulk volume, which results in high transportation costs and requires large storage capacities, and to the high moisture content, which can result in biological degradation as well as in freezing and blocking the in-plant transportation systems. In addition, variations in moisture content make difficult the optimal plant operation and process control. All of those problems may be overcome by standardization and densification. The former consists of processing the original biomass in order to obtain fuels with standard size and heating properties, while the latter consists in compressing the material, which needs to be available in the sawdust size, to give it more uniform properties.
Table 1 Types of biomass for energy use
Table 2 reports the main features of pellets, briquettes, and chips.
Fig. 1 Processes to convert biomass into useful energy, i.e., bioenergy.
Table 2 Comparison of different solid wood fuels
BIOMASS COMBUSTION
The burning of wood and other solid biomass is the oldest energy technology used by man. Combustion is a well-established commercial technology with applications in most industrialized and developing countries, and development is concentrated on resolving environmental problems, improving the overall performance with multi-fuel operation, and increasing the efficiency of the power and heat cycles (CHP).
The devices used for direct combustion of solid bio-mass fuels range from small domestic stoves (1-10 kW) to the large boilers used in power and CHP plants (> 5 MW). Intermediate devices cover small boilers (10-50 kW) used for heating in single-family houses, medium-sized boilers (50-150 kW) used for heating in multifamily houses or buildings, and large boilers (150 kW to over 1 MW) used for district heating. Cofiring in fossil-fired power stations enables the advantages of large size plants (> 100 MWe) that are not applicable for dedicated biomass combustion due to limited local biomass availability.
To achieve complete burnout and high efficiencies in small-scale combustion, downdraft boilers with inverse flow have been introduced, which apply the two-stage combustion principle. An operation at very low load should be avoided as it can lead to high emissions. Hence, it is recommended to couple log wood boilers to a heat storage tank. Because wood pellets are well suited for automatic heating at small heat outputs (as needed for nowadays buildings), pellet furnaces are an interesting application with growing propagation. Thanks to the well-defined fuel at low water content, pellet furnaces can easily achieve high combustion quality. They are applied both as stoves and as boilers, and they are encountering increasing acceptance especially in urban areas because modern pellet stoves are nowadays efficient home heating appliances. While a conventional fireplace is less than 10% efficient at delivering heat to a house, an average modern pellet stove achieves 80%-90% efficiency. Technology development has led to the application of strongly improved heating systems that are automated and have catalytic gas cleaning equipment. Such systems significantly reduce the emissions from fireplaces and older systems while at the same time improving the efficiency.
Understoker furnaces are mostly used for wood chips and similar fuel with relatively low ash content, while grate furnaces can also be applied for high ash and water content. Special types of furnaces have been developed for straw that has very low density and is usually stored in bales. Other than conventional grate furnaces operated with whole bales, cigar burners and other specific furnaces are in operation. Stationary or bubbling fluidized bed (SFB) as well as circulating fluidized bed (CFB) boilers are applied for large-scale applications and are often used for waste wood or mixtures of wood and industrial wastes, e.g., from the pulp and paper industry.
Co-Combustion
Bioenergy production may be hampered by limitations in the supply or by fuel quality. In those cases, the cofiring of several types of biomass or cofiring biomass with coal ensures flexibility in operation, both technically and economically. Several concepts have been developed:
• Co-combustion or direct cofiring. The biomass is directly fed to the boiler furnace, if needed, after physical preprocessing of the biomass such as drying, grinding, or metal removal is applied. This typically takes place in bubbling or CFB combustors. Such technologies can be applied to a wide range of fuels, even for very wet fuels like bark or sludge. Multifuel fluidized bed boilers achieve efficiencies over 90% while flue gas emissions are lower than for conventional grate combustion due to lower combustion temperatures.
• Indirect cofiring. Biomass is first gasified and the fuel gas is cofired in the main boiler. Sometimes the gas has to be cooled and cleaned, which is more challenging and implies higher operation costs.
• Parallel combustion. The biomass is burnt in a separate boiler for steam generation. The steam is used in a power plant together with the main fuel.
Problems in Biomass Combustion
Biomass has a number of characteristics that makes it more difficult to handle and combust than fossil fuels. The low energy density is the main problem in handling and transport of the biomass, while the difficulties in using biomass as fuel relates to its content of inorganic constituents. Some types of biomass used contain significant amounts of chlorine, sulfur, and potassium. The salts—KCl and K2SO4—are quite volatile, and the release of these components may lead to heavy deposition on heat transfer surfaces, resulting in reduced heat transfer and enhanced corrosion rates. Severe deposits may interfere with operation and cause unscheduled shut downs.
In order to minimize these problems, various fuel pretreatment processes have been considered, including washing the biomass with hot water or using a combination of pyrolysis and char treatment.
THERMOCHEMICAL CONVERSION OF BIOMASS
Pyrolysis and gasification are the two most typical thermochemical processes that do not produce useful energy directly because they convert the original bioenergy feedstock into more convenient energy carriers such as producer gas, oil, methanol, and char.[3]
Pyrolysis
Pyrolysis is a process for thermal conversion of solid fuels, like biomass or wastes, in the complete absence of oxidizing agent (air/oxygen) or with such limited supply that gasification does not occur to any appreciable extent. Commercial applications are either focused on the production of charcoal or the production of a liquid product—the bio-oil and pyro-gas. Charcoal is a very ancient product, even if traditional processes (partial combustion of wood covered by a layer of earth) are very inefficient and polluting. Modern processes such as rotary kiln carbonization are presently used in industry. Bio-oil production (or wood liquefaction) is potentially very interesting as a substitute for fuel oil and as a feedstock for production of synthetic gasoline or diesel fuel. Pyro-gas has higher energy density than gasification gas (syngas) because it has been created without oxygen (and nitrogen, if air is employed), hence it does not contain the gaseous products of partial combustion.
The pyrolysis process takes place at temperatures in the range 400°C-800°C and during this process most of the cellulose and hemicellulose and part of the lignin will disintegrate to form smaller and lighter molecules, which are gases at the pyrolysis temperature. As these gases cool, some of the vapors condense to form a liquid, which is the bio-oil and the tars. The remaining part of the biomass, mainly parts of the lignin, is left as a solid, i.e., the charcoal. It is possible to influence the product mix through a control of heating rate, residence time, pressure, and maximum reaction temperature so that either gases, condensable vapors, or the solid charcoal is promoted.
Gasification
Gasification technology has been developing since the 18th century, and it is still in a development phase.[5'6] Gasification is a conversion process that involves partial oxidation at elevated temperature. It is intermediate between combustion and pyrolysis. In fact, oxygen (or air) is present but it is not enough for complete combustion. This process can start from carbonaceous feedstock such as biomass or coal and convert them into a gaseous energy carrier. The overall gasification process may be split into two main stages: the first is pyrolysis stage, i.e., where oxygen is not present but temperature is high, and here typical pyrolysis reactions take place; the second stage is the partial combustion, where oxygen is present and it reacts with the pyrolyzed biomass to release heat necessary for the process. In the latter stage, the actual gasification reactions take place, which consist of almost complete charcoal conversion into lighter gaseous products (e.g., carbon monoxide and hydrogen) through the chemical oxidizing action of oxygen, steam, and carbon dioxide. Such gases are injected into the reactor near the partial combustion zone (normally, steam and carbon dioxide are mutually exclusive). Gasification reactions require temperature in excess of 800°C to minimize tar and maximize gas production. The gasification output gas, called “producer gas,” is composed by hydrogen (18%-20%), carbon monoxide (18%-20%), carbon dioxide (8%-10%), methane (2%-3%), trace amounts of higher hydrocarbons like ethane and ethene, water, nitrogen (if air is used as oxidant agent), and various contaminants such as small char particles, ash, tars, and oils. The incondensable part of producer gas is called “syngas” and it represents the useful product of gasification. If air is used, syngas has a high heating value in the order of 4-7 MJ/m3, which is exploitable for boiler, engine, and turbine operation, but due to its low energy density, it is not suitable for pipeline transportation. If pure oxygen is used, the syngas high heating value almost doubles (approximately 10-18 MJ/m3 high heating value), hence such a syngas is suitable for limited pipeline distribution as well as for conversion to liquid fuels (e.g., methanol and gasoline). However, the most common technology is the air gasification because it avoids the costs and the hazards of oxygen production and usage. With air gasification, the syngas efficiency—describing the energy content of the cold gas stream in relation to that of the input biomass stream—is in the order of 55%-85%, typically 70%.
Comparison of Thermal Conversion Methods of Biomass
Table 3 reports a general overview on specific features of the conversion technologies analyzed here, showing the related advantages and drawbacks.
BIOCHEMICAL CONVERSION OF BIOMASS
Biochemical conversion of biomass refers to processes that decompose the original biomass into useful products. Commonly, the energy product is either in the liquid or in gaseous forms, hence it is called “biofuel” or “biogas,” respectively. Biofuels are very promising for transportation sector, while biogas is used for electricity and heat production. Normally, biofuels are obtained from dedicated crops (e.g., biodiesel from seed oil), while biogas production results from concerns over environmental issues such as the elimination of pollution, the treatment of waste, and the control of landfill greenhouse gas emissions.
Biogas from Anaerobic Digestion
The most common way to produce biogas is anaerobic digestion of biomass. Anaerobic digestion is the bacterial breakdown of organic materials in the absence of oxygen.
This biochemical process produces a gas called biogas, principally composed of methane (30%-60% in volume) and carbon dioxide. Such a biogas can be converted to energy in the following ways:
• Biogas converted by conventional boilers for heating purposes at the production plant (house heating, district heating, industrial purposes).
• Biogas for combined heat and power generation.
• Biogas and natural gas combinations and integration in the natural gas grid.
• Biogas upgraded and used as vehicle fuel in the transportation sector.
• Biogas utilization for hydrogen production and fuel cells.
An important production of biogas comes from landfills. Anaerobic digestion in landfills is brought about by the microbial decomposition of the organic matter in refuse. Landfill gas is on average 55% methane and 45% carbon dioxide. With waste generation increasing at a faster rate than economic growth, it makes sense to recover the energy from that stream through thermal or fermentation processes.
Biofuels for Transport
A wide range of chemical processes may be employed to produce liquid fuels from biomass. Such fuels can find a very high level of acceptance by the market thanks to their relatively easy adaptation to existing technologies (i.e., gasoline and diesel engines). The main potential biofuels are outlined below.
• Biodiesel is a methyl-ester produced from vegetable or animal oil to be used as an alternative to conventional petroleum-derived diesel fuel. Compared to pure vegetable or animal oil, which can be used in adapted diesel engines as well, biodiesel presents lower viscosity and slightly higher HHV.
• Pure vegetable oil is produced from oil plants through pressing, extraction, or comparable procedures, crude or refined but chemically unmodified. Usually, it is
Table 3 Qualitative comparison of technologies for energy conversion of biomass
| Process | Technology | Economics | Environment | Market potential | Present deployment |
| Combustion-heat | + + + | € | + + + | + + + | + + + |
| Combustion-electricity | + +(+) | €€ | + +(+) | + + + | + + |
| Gasification | + (+) | €€€ | + (++) | + + + | (+) |
| Pyrolysis | (+) | €€€€ | (+ + +) | + +(+) | ( +) |
compatible with existing diesel engines only if blended with conventional diesel fuel at rates not higher than 5%-10% in volume. Higher rates may lead to emission and engine durability problems.
• Bioethanol is ethanol produced from biomass or the biodegradable fraction of waste. Bioethanol can be produced from any biological feedstock that contains appreciable amounts of sugar or other matter that can be converted into sugar, such as starch or cellulose. Also, ligno-cellulosic materials (wood and straw) can be used, but their processing into bioethanol is more expensive. Application is possible to modified spark ignition engines.
• Bio-ETBE (ethyl-tertio-butyl-ether) is ETBE produced on the basis of bioethanol. Bio-ETBE may be effectively used to enhance the octane number of gasoline (blends with petrol gasoline).
• Biomethanol is methanol produced from biomass. Methanol can be produced from gasification syngas (a mixture of carbon monoxide and hydrogen) or wood dry distillation (old method with low methanol yields). Most all syngas for conventional methanol production is produced by the steam reforming of natural gas into syngas. In the case of biomethanol, a biomass is gasified first to produce a syngas from which the biomethanol is produced. Application is possible to spark ignition engines and fuel cells. Compared to ethanol, methanol presents more serious handling issues because it is corrosive and poisonous for human beings.
• Bio-MTBE (methyl-tertio-butyl-ether) is a fuel produced on the basis of biomethanol. It is suitable for blends with petrol gasoline.
• Biodimethylether (DME) is dimethylether produced from biomass. Bio-DME can be formed from syngas by means of oxygenate synthesis. It has emerged only recently as an automotive fuel option. Storage capabilities are similar to those of LPG. Application is possible to spark ignition engines.
BENEFITS FROM BIOMASS ENERGY
There is quite a wide consensus that, over the coming decades, modern biofuels will provide a substantial source of alternative energy. Nowadays, biomass already provides approximately 11%-14% of the world’s primary energy consumption (data varies according to sources).
There are significant differences between industrialized and developing countries. In particular, in many developing countries, bioenergy is the main energy source—even if used in very low-efficiency applications (e.g., cooking stoves have an efficiency of about 5%-15%). Furthermore, inefficient biomass utilization is often associated with the increasing scarcity of hand-gathered wood, nutrient depletion, and the problems of deforestation and desertification.
One of the key drivers to bioenergy deployment is its positive environmental benefit, in particular regarding the global balance of green house gas (GHG) emissions. This is not a trivial matter, because biomass production and use are not entirely GHG neutral. In general terms, the GHG emission reduction as a result of employing biomass for energy reads as reported in Table 4.
Bioenergy is a decentralized energy option whose implementation presents positive impacts on rural development by creating business and employment opportunities. Jobs are created all along the bioenergy chain, from biomass production or procurement to its transport, conversion, distribution, and marketing.
Bioenergy is a key factor for the transition to a more sustainable development.
CONCLUSIONS
Biomass refers to a very wide range of substances produced by biological processes. In the energy field, special focus has been and will be placed on vegetable biomass such as wood and agricultural by-products because of the energy potential as well as economic and environmental benefits. Size and humidity standardization of biomass is a necessary step to make it suitable for effective domestic and industrial exploitation. Chips, briquettes, and pellets are modern examples of standard solid fuels.
Biomass can be converted to energy in three may pathways: combustion, thermochemical processing, and biochemical processing. The combustion of solid biomass for the production of heat or electricity is the most viable technology, while pyrolysis and gasification still face economic and reliability issues. Among biochemical processes, anaerobic digestion is often used to reduce the environmental impact of hazardous waste and landfills. Biochemical processes are also concerned with the conversion of biomass into useful fuels for transportation, such as biodiesel, bioethanol, biomethanol, and others. All of them can effectively contribute to the transition to a more sustainable transportation system at zero GHG emissions.
Table 4 Benefits in reduction of green houses gas emissions
| + | Avoided mining of fossil resources |
| - | Emission from biomass production |
| + | Avoided fossil fuel transport (from |
| producer to user) | |
| — | Emission from biomass fuel transport |
| (from producer to user) | |
| + | Avoided fossil fuel utilization |
+, positive; —, neutral.
Biomass represents a viable option for green energy resources of the 21st century.