Bivalvia
(Bivalves)
Phylum Mollusca
Number of families 105
Thumbnail description
Bilaterally symmetrical mollusks, with a reduced head and typically two external shell valves, many of which are commercially important for human consumption and pearl production; some have major impacts on the world economy and environment
Photo: A coquina clam (Donax variabilis) underwater, with its siphons extended.

Evolution and systematics
The evolutionary history of bivalves is represented by an extensive fossil record. It begins in the Cambrian period (544-505 million years ago [mya]) with a laterally compressed stenothecid monoplacophoran (primitive single-shelled marine mollusk) as the most likely immediate ancestor. By the Middle Ordovician period (about 460 mya), recognizable members of all modern subclasses had appeared. Bivalves formed important components of marine communities since they first diversified, especially in shallow-water marine sediments, but also in the intertidal zone, the deep sea, and freshwater habitats. In the Cretaceous period (145-65 mya), epibenthic rudist bivalves formed tropical reef-like structures similar to modern coral reefs. Rudists were massive extinct cemented bivalves that had two different-sized shell valves.
Most authors think that scaphopods and bivalves are closely related taxa, based on the configuration of the nervous system, lateral expansion of the mantle, and elongation of the foot for burrowing. Within the class of bivalves itself, the widely variable shell characteristics (shape, sculpture, color, hinge teeth) have been historically and consistently used in identification and classification at all levels. Various other evolutionary schemes have been based primarily on single organ systems, especially the ligament, stomach, digestive tract, and gills. Elements of each of these continue to be important in modern comprehensive phylogenetic analyses.
Current classification schemes recognize five subclasses of bivalves. The Protobranchia (e.g., families Nuculidae, Solemyidae) are presumably the most primitive, using simple gills solely for respiration and palp proboscides (enlarged labial palps, normally used for sorting food particles) for collecting food from the sediment surface. The Pteriomorphia (e.g., Mytilidae, Pteriidae) include many of the most familiar bivalves, all of which share an epibenthic habitat (byssate or cemented), an unfused mantle edge, and a reduced foot. Byssate bivalves are attached to their substrate by a byssus (bundle of elastic collagen-rich threads) secreted by the foot. The Pale-oheterodonta (e.g., Unionidae, Margaritiferidae) include the freshwater mussels, with their specialized glochidia larvae. Heterodonta (e.g., Veneridae, Donacidae) is the most species-rich and most widely distributed subclass, containing the classic burrowing clams with well-developed hinges, siphons, and active feet. The Anomalodesmata (e.g., Pandoridae, Clavag-ellidae) include the most unusual and most specialized bivalves, featuring modified ctenidia (gills), an edentulous hinge, fused mantle margins, and hermaphroditism. The evolutionary relationships, and thus the accepted classification, among and within bivalve subgroups continue to be revised through the application of phylogenetic analysis. These analyses makes use of a wide range of morphological (especially anatomical) and molecular characters.
Physical characteristics
Most bivalved mollusks have laterally compressed bodies and a shell consisting of two calcareous valves hinged dorsally by interlocking teeth and an elastic ligament. The shell valves are usually similar in size, sculpture, and color, and retain a permanent record of shell growth in their concentric layers. Growth can be traced from the first stage (prodissoconch) at the umbo (rounded or pointed extremity) to the latest stage at the ventral margin. The shells of many bivalve species are rather bland, although some groups (e.g., Pectinidae, Spondyl-idae) show characteristic colors or color patterns. Lining each valve is mantle tissue that secretes the aragonitic or calcitic shell, including its organic outer layer, or periostracum, and the interior aragonitic nacre (mother-of-pearl) present in many species. Between the two shell-mantle layers, an internal mantle (or pallial) cavity contains the ctenidia and visceral mass (containing digestive and reproductive organs), the latter ending in a muscular extensible foot. The foot is usually equipped with a gland that secretes the byssus for attachment to hard substrates.
The posterior mantle edge of many bivalves is fused into incurrent and excurrent siphons that direct water in and out of the mantle cavity for respiration, feeding, and discharge of waste and reproductive products. In most bivalves, water flows in and out of the posterior end; some, however, are secondarily modified for an anterior-posterior flow. Paired adductor muscles connect the inner valve surfaces, enabling the bivalve to close; relaxation of these muscles allows the ligament to open the valves. The head of bivalves is reduced, so that the cephalic eyes, tentacles, and radular teeth typical of most mollusks are absent. The stomach is one of the most complex organs of bivalves, comprising various ciliated sorting areas and ridges as well as the crystalline style, which is an enzymatic rod that rotates against a hardened gastric shield lining the dorsal stomach surface to facilitate extracellular digestion. The bivalve nervous system consists simply of three pairs of ganglia connected by nerve connectives. The circulatory system is equipped with a three-chambered heart, simple vessels, and open hemocoels. The hemostatic pressure of the fluid in these blood cavities is responsible for the expansion and retraction of many bivalve structures, particularly the foot, siphons, and tentacles.
Distribution
Bivalves are found worldwide, including aquatic habitats at high and low latitudes. All are aquatic, requiring water for reproductive processes, respiration, and typically for feeding. One supratidal species (Enigmonia) lives in the tidal spray on mangrove leaves or seawalls in Australia, achieving the most nearly “terrestrial” life mode of any bivalve. Several independent lineages of bivalves have invaded freshwater habitats, where they have diversified to produce one of the most endemic faunas as well as some of the most important biofoulers among invertebrates. Many species have been introduced to non-native locations accidentally or intentionally, the latter often in commercial aquaculture for human use or consumption.
Habitat
All bivalves are aquatic, requiring water for reproductive processes, respiration, and typically for feeding. They range in depth from the intertidal zone to the deep sea; one suprati-dal species (Enigmonia) lives in the tidal spray on mangrove leaves or seawalls in Australia, achieving the most “terrestrial” life-mode of any bivalve. Several independent lineages of bivalves have invaded freshwater habitats, where they have diversified to produce one of the most endemic faunas as well as some of the most important biofoulers among invertebrates. Most bivalves are free-living, either epibenthic (e.g., Pectinidae) or infaunal, burrowing into sand or mud with the muscular foot (e.g., Veneridae, Donacidae). Others are cemented by one valve (e.g., Ostreidae) or permanently attached by byssal threads (e.g., Mytilidae, Dreissenidae). Specialized members of the class burrow into rock or wood (e.g., Pholadidae, Teredinidae), using one or a combination of chemical and mechanical methods. Commensal and parasitic forms (e.g., Galeommatoidea) live associated with, attached to, or within the bodies of other invertebrates.
The habitat of a bivalve is often reflected in the form of its shell. Nestlers and cementing bivalves frequently take the shape of their substrates. Individuals in calm waters often have more delicate or leaflike shell sculpture than their counterparts in fast-flowing currents.
Behavior
Most bivalves are relatively sedentary organisms; however, many are capable of considerable levels of activity. The former class name Pelecypoda means “hatchet-foot,” referring to the laterally compressed foot typically used for burrowing in sand or mud. Bivalves move downward into the substrate by extending the foot into the sediment, anchoring the foot by expanding its tip, and pulling the shell downward toward the anchor by muscular action. Byssally attached bivalves (e.g., Mytilidae, Dreissenidae) can break their byssal threads to relocate, and use the foot to move across a hard substrate in a sequence similar to that used for burrowing. They then produce a new byssus for reattachment. Other bivalves can actively swim by waving the foot or tentacles (e.g., Solenidae, Limidae) or by jet-propelling themselves by rapidly clapping the shell valves together (e.g., Pectinidae).
Although the reduction of the bivalve head has eliminated cephalic eyes and other sense organs, many bivalves (e.g., Ga-leommatidae, Pectinidae) have tentacles and/or photorecep-tors along the mantle margins or in the vicinity of the siphons. These structures allow bivalves to respond to changes in light intensity by retracting the siphons and closing the valves. More sophisticated eyes, equipped with retina and lens, are found in several families of epibenthic bivalves (e.g., Cardi-idae, Pectinidae).
Feeding ecology and diet
Most bivalves are suspension feeders, filtering food particles from the water column. The expansive ctenidia, in addition to functioning in gas exchange, are the main feeding organs. Their cilia-covered surface collects and sorts particles from currents flowing through the mantle cavity, conveying them to marginal food grooves, then anteriorly toward the labial palps flanking the mouth.
The most primitive bivalves were probably deposit feeders, collecting detritus from the sediment surface. This method is
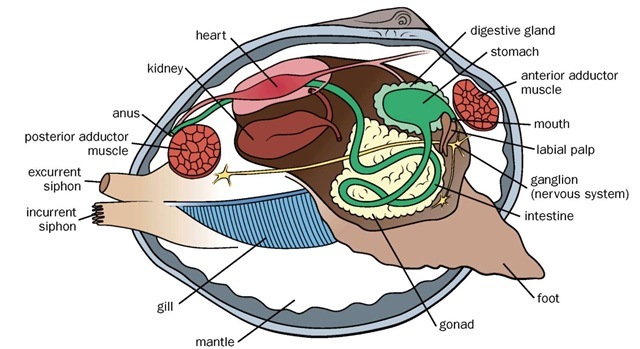
Bivalve anatomy.
still used by living Nuculoida, using specialized structures known as palp proboscides. Other specialists feed by direct absorption of dissolved organic matter, or DOM (e.g., Sole-myidae), or by active capture of small crustaceans and worms through use of a raptorial incurrent siphon (e.g., Cuspidari-idae). Others possess symbiotic organisms, supplementing their energy reserves with by-products from their inhabitants. Examples of symbiotic relationships include chemoau-totrophic bacteria in Solemyidae and Lucinidae that facilitate habitation of anoxic muds, and zooxanthellae in Cardiidae that provide photosynthetic products in shallow eutrophic waters. The wood-eating Teredinidae are enabled by symbiotic cel-lulolytic (cellulose-digesting) bacteria that are stored in pouches along the bivalve’s esophagus.
Reproductive biology
Bivalves are usually dioecious, with eggs and sperm shed into the water column where external fertilization occurs. Some species are consecutive or simultaneous hermaphrodites, with protandry (male phase preceding female phase) most common. Internal fertilization has been recorded for a few groups (Galeommatoidea, Teredinidae), using tentacles or siphons as copulatory organs. External sexual dimorphism is evident in only a few bivalves (Carditidae, Unionoidea).
Larval development is plesiomorphically planktotrophic, with free-swimming veliger larvae that feed in the plankton for a few weeks. Some bivalves brood their larvae in the supra-branchial chamber or in specialized brood pouches, releasing late-stage veligers or direct-developed juveniles through the excurrent opening. Settlement of larvae is time-dependent but is often delayed in the absence of suitable habitat. Freshwater mussels (Unionoidea) are characterized by specialized glochidia larvae that require attachment to the gills or fins of fish to complete their life cycles. Many of these larvae have specialized hooks for attachment, and some bivalve-fish relationships are species-specific. Many unionoideans possess specialized flaps on the mantle edge that they wave in the water column to attract the attention of the required fish; some of these “lures” mimic small fish or the invertebrate prey of the fish host.
Conservation status
Freshwater pearl mussels (Unionoida) are among the world’s most gravely threatened fauna. In eastern North America, the group’s center of evolutionary diversification, 35% of the 297 native species are presumed extinct, with another 69% formally listed as endangered or threatened. Human-introduced pollution, especially from agriculture and industry, as well as other forms of habitat alteration (dredging, damming) have been blamed for much of the decline. Such factors can adversely impact not only the mussels themselves, but also the obligate fish hosts of their larvae, potentially resulting in population declines. Introduced species, especially the Asian clam (Corbicula) and two species of zebra mussels (Dreissena) have further impacted unionoid populations through competition for space and food resources. One-hundred ninety-five species of bivalves have been placed on the 2002 IUCN Red List; all but 10 of these are freshwater pearl mussels. Twenty-nine species of freshwater pearl mussels are protected under CITES.
Marine bivalves are much less affected by human activities. There are no known recent extinctions in this group, and none are currently listed as threatened or endangered. Many species are partially protected by local and national laws regulating the commercial and private harvesting of shellfish. The giant clams (Tridacna, Hippopus; Tridacnidae) are the single marine group regulated internationally, as a result of overcollecting; eight species are included on the 2002 IUCN Red List, and the entire family is protected under CITES.
Significance to humans
Many kinds of bivalves, especially clams, cockles, mussels, oysters, and scallops, have served as important food sources for fish, vertebrates, other invertebrates, and humans. Aboriginal populations of many cultures have left evidence of eating bivalves in their kitchen middens (mounds or deposits of refuse from meals). Recent practices rely both on harvesting wild populations and on aquaculture in either open or closed aquatic systems. Members of the marine Pteriidae and freshwater Unionoidea have been sources of natural pearls and mother-of-pearl shell for centuries. Since the 1950s, cultured pearls have increased the quantity and quality of this biological gem through aquaculture and husbandry. The Japanese perfected the process of culturing pearls using pearl oysters of the species Pinctada fu-cata (Gould, 1850).
Bivalves have also had negative impacts on human activities. Because most bivalves are filter feeders, they are frequent vectors of human disease related to the concentration of bacteria, viruses, pesticides, industrial wastes, toxic metals, and petroleum derivatives from the water column. Shipworms (Tere-dinidae) have a long historical record of bioerosion of such human-made wooden structures as ships and docks. Species introduced in freshwaters of the United States, such as the bio-fouling zebra mussel Dreissena, have required millions of dollars to repair clogged water treatment plants and irrigation systems. Damage caused by such species to the environment, in terms of altered habitat and impact on native species, is irreversible; their spread has been largely unstoppable.

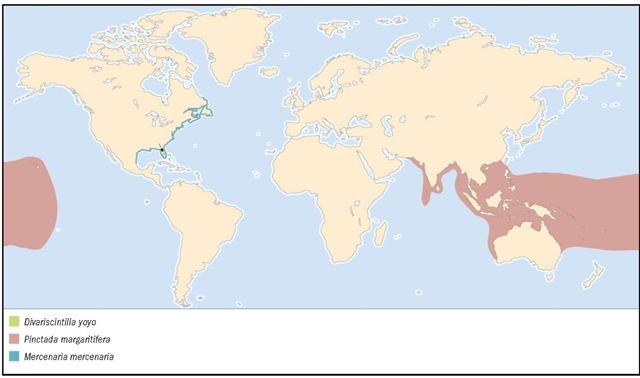
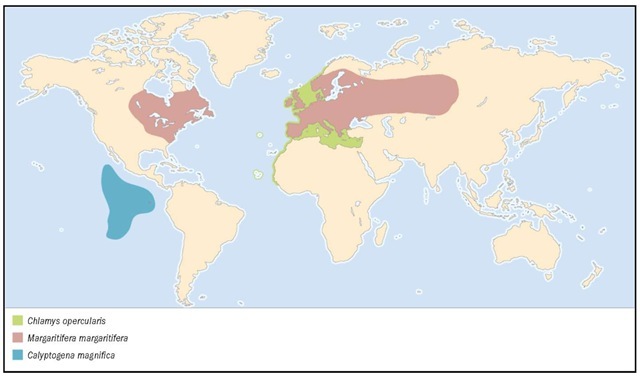
1. Eastern American oyster (Crassostrea virginica); 2. Queen scallop (Chlamys opercularis); 3. Yo-yo clam (Divariscintilla yoyo); 4. Common blue mussel (Mytilus edulis); 5. European pearly mussel (Margaritifera margaritifera); 6. Noble pen shell (Pinna nobilis); 7. Black-lipped pearl oyster (Pinctada margaritifera); 8. P. margaritifera internal view.

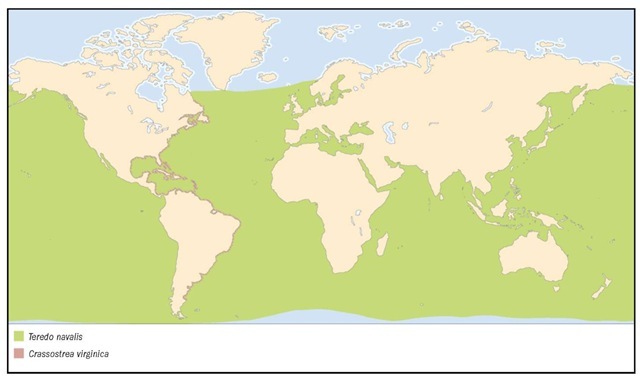
1. Zebra mussel (Dreissena polymorpha); 2. Shipworm (Teredo navalis); 3. Giant vent clam (Calyptogena magnifica); 4. Giant clam (Tridacna gi-gas); 5. Northern quahog (Mercenaria mercenaria); 6. Watering pot shell (Brechites vaginiferus); 7. Coquina clam (Donax variabilis); 8. Coquina clam color morph 1; 9. Coquina clam color morph 2.
Species accounts
Common shipworm
Teredo navalis
ORDER
Myoida
FAMILY
Teredinidae
TAXONOMY
Teredo navalis Linnaeus, 1758, The Netherlands.
OTHER COMMON NAMES
English: Gribble, pileworm, ship’s worm; French: Taret com-mun; German: Schiffsbohrwurm.
PHYSICAL CHARACTERISTICS
Shell is white, triangular in shape, inflated, and coarsely sculptured with ridges used for burrowing; reduced in size and permanently gaping. It covers only the anterior end of a much larger worm-like soft body. Generally 4 in (10 cm) in total length, but may grow as long as 2 ft (60 cm). Mantle secretes a calcareous lining for wood burrow. The posterior end has short siphons and unsegmented shovel-shaped calcareous pallets used to close the burrow. Stomach has a wood-storing caecum or blind pouch.
DISTRIBUTION
Worldwide in temperate seas, spread by wooden vessels and ballast water. Probably native to northeastern Atlantic; definitely introduced to San Francisco Bay, but additional differentiation of native versus introduced range is unclear. Reported from such far-flung locations as southern Brazil, Zaire, South Australia, the Black Sea, New England, and British Columbia.
HABITAT
Burrows into floating or stationary untreated wood in seawater. Tolerates salinities ranging from normal seawater to 4 ppt.
BEHAVIOR
Specialized for boring in wood by using ridged shell valves to rasp into wood surface. During burrowing, the animal’s disklike foot acts as suction cup to hold shell tightly against end of burrow. When disturbed, it withdraws into burrow and seals opening with specially shaped pallets.
FEEDING ECOLOGY AND DIET
Specialized for boring into and digesting wood with the assistance of symbiotic cellulolytic bacteria and specialized wood-storing caecum on stomach. Ctenidia are well-developed and equipped with food groove, indicating retained ability to filter-feed.
REPRODUCTIVE BIOLOGY
Protandric hermaphrodite. Extended excurrent siphon probably (as known in other Teredinidae) used as copulatory organ for sperm transfer to adjacent individual. Larvae are brooded in gills to veliger stage. Juveniles grow to maturity in eight weeks. Several generations produced per year. Capable of invading new wood only at time of larval settlement.
CONSERVATION STATUS
Not listed by the IUCN. Considered a nuisance species due to wood-destroying capabilities.
SIGNIFICANCE TO HUMANS
Called “termites of the sea.” Damage to wooden ships and piers, especially in warm tropical waters, recorded as early as Roman times (A.D. 2). Countered by coating wooden surfaces with tar and pitch in the fifteenth and sixteenth centuries, later by copper plating. Responsible for more than $900 million in damages to wooden piers and quays in San Francisco, California, between 1819 and 1821.
Common blue mussel
Mytilus edulis
ORDER
Mytiloida
FAMILY
Mytilidae
TAXONOMY
Mytilus edulis Linnaeus, 1758, European oceans.
OTHER COMMON NAMES
English: Blue mussel, common mussel; French: Moule bleue; German: Miesmuschel; Japanese: Murasakii-gai; Norwegian: Blaskjell; Spanish: Mejillon; Swedish: Blamussla.
PHYSICAL CHARACTERISTICS
The shell is narrowly elongated and triangular in shape, with the umbo at the pointed anterior end. It is rounded at the posterior end, moderately inflated, smooth, not gaping, with an adherent periostracum varying from dark blue or purple to brown. Generally 2—4 in (5-10 cm) in length, the largest reaching 8 in (20 cm). Shell shape and size influenced by environment, especially tidal level, habitat type, and population density. Interior is faintly nacreous (pearly). Soft body secretes strong stalk of byssal threads exiting shell at ventral margin; mantle edge is yellowish brown; anterior adductor muscle narrow and elongated.
DISTRIBUTION
Northern temperate coastlines from Scandinavia to France; Iceland; maritime Canada to North Carolina; both coasts of southern tip of South America. Raised in aquaculture in China, many European countries, Canada, and United States.
HABITAT
Epibenthic, attached by byssus to hard substrates from high in-tertidal to shallow subtidal waters. Prefers full oceanic salinity, although dwarfed individuals have been recorded at 4-5 ppt. Lower depth usually limited by presence of such predators as seastars, gastropods, and crabs. Forms large populations (mussel beds) on rocky shores that support extensive associated fauna and flora.
BEHAVIOR
Sessile (permanently attached).
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Dioecious; broadcast spawner, with females releasing eggs in short yellow-orange rod-shaped masses that quickly break down in the water column. The veligers are initially

lecithotrophic (nourished by stored yolk), but are later plank-totrophic, spending 1-4 weeks in the water column. Settling is triggered by the presence of a filamentous substrate (bry-ozoans, hydroids, algae). Growth rate in culture may be as much as 3 in (80 mm) per year. Life span may be as long as 24 years. Hybridizes with the Mediterranean Mytilus galloprovin-cialis in northern Europe.
CONSERVATION STATUS
Not listed by the IUCN. Collection for human consumption is regulated by fisheries and public health agencies in most areas.
SIGNIFICANCE TO HUMANS
Human food source worldwide. Recorded as vector of human gastroenteritis, hepatitis A, and viral hepatitis. Used in worldwide “Mussel Watch” programs as an indicator organism for monitoring coastal water quality. Extensively used as model organism in scientific studies.
Eastern American oyster
Crassostrea virginica
ORDER
Ostreoida
FAMILY
Ostreidae
TAXONOMY
Crassostrea virginica (Gmelin, 1791), American and [West] Indian Oceans.
OTHER COMMON NAMES
English: American oyster, Atlantic oyster, cove oyster, eastern oyster; French: Huitre de Virginie; German: Amerikanische Auster. Also has commercial varietal names: Blue points (Long Island, New York), Lynnhavens (Virginia).
PHYSICAL CHARACTERISTICS
Shell is elongated and ovate-triangular in shape, irregular and variable in outline, roughly sculptured with undulating concentric ridges. The left (cemented) valve more deeply cupped, right valve flattened, not gaping. Color is whitish to gray. Hinge teeth reduced, ligament strong. Interior is porcelain white with purple-stained muscle scar. Generally grows about 3 in (8 cm) in length, but specimens have been found as long as 14 in (36 cm). Soft body with central adductor muscle and unfused mantle margins.
DISTRIBUTION
Atlantic coast of North America from Canada to Florida, the Gulf of Mexico, and the West Indies to Brazil and Argentina. Introduced to Europe, the western coast of United States, and Hawaii; populations established only in British Columbia and Hawaii. Raised in aquaculture in Australia, New Zealand, Europe, Chile, Japan, Mexico, and eastern United States.
HABITAT
Epibenthic, forming oyster reefs or beds on hard surfaces in shallow coastal estuaries of reduced salinity. Crassostrea virginica tolerates a wide range of salinity (5-35 ppt) and temperature. Complex physical structure of oyster beds provides refuge for many other organisms.
BEHAVIOR
Tight shell closure allows individuals to survive many days of aerial exposure. The acidity of the shell liquor (commercial term for water within mantle cavity) is mediated by the buffering action of calcium carbonate dissolved from shell.
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Protandric hermaphrodite; broadcast spawner, triggered by warming water temperatures. Veligers are planktonic for about three weeks, settling in response to adult oyster shells. Newly attached juveniles are called “spat.” Hermaphrodites are rare; instances are recorded of secondary sex reversal (female reverting to male). Three to seven years in culture are required to attain market size of 3 in (8 cm).
CONSERVATION STATUS
Not listed by the IUCN. Collection for human consumption is regulated by fisheries and public health agencies in most areas. Many historically important harvesting areas, such as Prince Edward Island, are severely depleted as of 2003.
SIGNIFICANCE TO HUMANS
Human food source, including many famous local varieties (Chesapeake Bay, Apalachicola Bay, Prince Edward Island), both through wild harvest and aquaculture. Oyster reefs stabilize shorelines against erosion and filter estuarine water. Recorded as vector of human disease caused by uptake and concentration of toxic metals and lipophilic organic contaminants.
Queen scallop
Aequipecten opercularis
ORDER
Pterioida
FAMILY
Pectinidae
TAXONOMY
Aequipecten opercularis (Linnaeus, 1758), Mediterranean Sea. Numerous named color forms and varieties.
OTHER COMMON NAMES
French: Vanneau.
PHYSICAL CHARACTERISTICS
Shell is round in outline and compressed, with subequal anterior and posterior auricles (ears) strongly delimited, gaping below each auricle, with about 20 finely sculptured radial ribs. Shell color is highly variable; may be white, red, orange, mottled or solid, with the right valve lighter in color than the left. Interior is white, with grooves reflecting external ribs. May grow as large as 3 in (80 mm) in diameter. Soft body with single central adductor muscle; mantle margin equipped with numerous sensory tentacles and eyes.
DISTRIBUTION
Mediterranean Sea and eastern Atlantic coast from Norway to the Cape Verde Islands, the Azores and the North Sea. Under experimental aquaculture in Spain, France, and United Kingdom.
HABITAT
Epibenthic on all substrates except rocky bottoms. Found in depths from the intertidal zone to 1,312 ft (400 m); most common at about 130 ft (40 m).
BEHAVIOR
Actively swims in response to threat by clapping shell valves together, forcing water to exit mantle cavity in a manner resembling jet propulsion.
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Simultaneous hermaphrodite, broadcast spawner.
CONSERVATION STATUS
Not listed by the IUCN, and not protected except by local fishery regulations. Fished extensively until 1970s, when populations declined and queen scallops became less important in the commercial market.
SIGNIFICANCE TO HUMANS
Human food source (adductor muscle or whole). The queen scallop symbol was originally worn on heraldic insignia to signify that the wearer had made a pilgrimage to the Christian shrine of St. James in Santiago de Compostela, Spain. The symbol later identified its bearer or ancestor as a Crusader or other type of pilgrim.
Watering pot shell
Brechites vaginiferus
ORDER
Pholadomyoida
FAMILY
Clavagellidae
TAXONOMY
Brechites vaginiferus (Lamarck, 1818), Aden, Red Sea. One recognized subspecies or variety, B. vaginiferus australis (Chenu, 1843), Australia.
OTHER COMMON NAMES
English: Australian watering pot, vaginal watering pot.
PHYSICAL CHARACTERISTICS
Shell is narrowly tube-shaped and irregularly sculptured, with the remnant of its 0.2-in (4 mm) larval shell in a lateral “saddle” near the anterior end. The anterior end is flared, comprising a perforated plate (reflected in common name) fringed by open tubules; posterior end has “plaited ruffles.” Color is white and chalky. Grows as long as 12 in (295 mm). Soft body with large bottle-green siphons fringed with white tentacles and ringed by a periostracal band. The apex is coated with sand grains. Adults lack adductor muscles.
DISTRIBUTION
Red Sea and Gulf of Aden southward down the eastern coast of Africa to Zanzibar, northeast to the mouth of Gulf of Oman. The variety australis from western Australia is found from the Kimberly Archipelago to the Abrolhos Islands.
HABITAT
Infaunal, buried vertically in soft sediment with only the open tube at the posterior end protruding into water. Found at depths of 0-66 ft (0-20 m). Cements itself to rock crevices in some habitats. Shell form reflects the habitat—long and thin in sand, wider and shorter in rock crevices.
BEHAVIOR
Tube is adventitious (has a single layer), thought to be produced all at once by the adult animal, analogous to the calcareous burrow lining in the Teredinidae. Shell material is subsequently added at posterior end to produce ruffles and lengthen tube. Animal can withdraw deeply into tube as protection against most predators. Cannot burrow further into sediment or reburrow if dislodged.
FEEDING ECOLOGY AND DIET
Filter feeder; pedal disk (foot) against perforated anterior plate contracts and relaxes to pump interstitial water into mantle cavity.
REPRODUCTIVE BIOLOGY
Simultaneous hermaphrodite; probably broadcast spawner, with planktonic veligers. Prodissoconch phase, pre-tube juveniles, and life span unknown.
CONSERVATION STATUS
Not listed by the IUCN; generally considered rare throughout its range.
SIGNIFICANCE TO HUMANS
None known.
Noble pen shell
Pinna nobilis
ORDER
Pterioida
FAMILY
Pinnidae
TAXONOMY
Pinna nobilis Linnaeus, 1758, Mediterranean Sea.
OTHER COMMON NAMES
English: Fan shell, Mediterranean pen shell; French: La Grande Nacre, jambonneau; German: Grosse Steckmuschel; Spanish: Gran nacra.
PHYSICAL CHARACTERISTICS
Shell is narrowly elongated, compressed, and triangular in shape; umbo at pointed anterior end, bluntly flattened or rounded at gaping posterior end. Surface is fragile, sculptured with dense erect scales in younger specimens that erode to smooth areas in older parts of large adults. Exterior is light tan to orange in color; interior lightly nacreous in anterior portion. P. nobilis is the second largest living bivalve, growing to 35.5 in (90 cm) in length. Soft body with pallial organ; secretes long strong stalk of gold or brown byssal threads exiting shell ven-trally near umbo.
DISTRIBUTION
Mediterranean Sea.
HABITAT
Infaunal, half-buried vertically in sand, mud, gravel, or sea-grass, in waters up to 197 ft (60 m) in depth. Shells form an important hard substrate for biocoenotic aggregations in soft-bottom habitats, especially mollusks and sponges, thereby increasing overall local species richness.
BEHAVIOR
The pallial organ actively clears broken shell and other debris from mantle cavity. Commensal crustaceans live outside the valves, retreating into mantle cavity when threatened; crab’s entry stimulates pen shell to close, thereby protecting the crustacean.
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Protandric hermaphrodite; broadcast spawner. Life span about 20 years.
CONSERVATION STATUS
Not listed by the IUCN. Overcollected by divers; considered endangered throughout the Mediterranean. Protected by national laws in Spain, France, and other coastal European countries.
SIGNIFICANCE TO HUMANS
Human food source (adductor muscle). The historical use of natural pearls, both nacreous and porcelaneous (orange to black), is documented as far back as the third century B.C. Gold-colored byssus threads as long as 2 ft (61 cm) were used in weaving textiles since Roman times, including cloth for gloves, shawls, stockings and cloaks. The mythological Golden Fleece was probably spun from byssus threads of this bivalve.
Black-lipped pearl oyster
Pinctada margaritifera
ORDER
Pterioida
FAMILY
Pteriidae
TAXONOMY
Pinctada margaritifera (Linnaeus, 1758), Indian Ocean. Recognized local varieties include P. m. var galtsoffi; Bartsch, 1931, of Hawaii, and P. m. var cumingii (Saville-Kent, 1890) of French Polynesia.
OTHER COMMON NAMES
English: Tahitian pearl oyster; French: Huitre a levres noires, nacre; German: Schwarzlippigen Perlenauster; Arabic: Sadaf; Italian: Ostrica dalle labbra nere.
PHYSICAL CHARACTERISTICS
Shell is round in outline, compressed, with anterior and posterior auricles faintly delimited. It is ornamented with concentric, radially aligned rows of overlapping fragile lamellae, not gaping. Color is overall blackish with white to green mottling. Generally 6-10 in (15-25 cm) in diameter. Interior is thickly nacreous in a wide range of colors including gray, blue, pink, yellow, and green, but seldom black as common name implies. Soft body with a gray or black foot and expansive gills. The orange mantle margin is unfused. The pearl oyster secretes strong byssal threads that exit the shell through a byssal notch below the anterior auricle.
DISTRIBUTION
Widespread in Indian Ocean and western to central Pacific, including Hawaiian Islands. Raised in pearl cultivation ventures in French Polynesia, Cook Islands, Gilbert Islands, Marshall Islands, Solomon Islands, southern China, northern and western Australia, Seychelles, and the Sudan. Variety galtsoffi (Bartsch, 1931), is cultured in Hawaii for potential restocking of natural habitat.
HABITAT
Epibenthic, bysally attached to hard substrates in atoll lagoons and coral reefs with calm clear (nutrient-poor) waters at depths of 3-130 ft (1-40 m).
BEHAVIOR
Water currents associated with normal physiological processes (respiration, excretion) eliminate most nonconsumable particles and microfauna. Natural pearl formation often results when these processes and muscular movements cannot dislodge foreign particles. Such particles lodge between shell and mantle or within mantle tissue, and are subsequently coated with nacre. More often, natural pearls form around internal parasites.
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Protandric hermaphrodite, broadcast spawner. Cultured pearl industries in South Pacific collect wild free-swimming larvae on spat collectors, subsequently raising juveniles to adult size for nucleation (insertion of a piece of mantle tissue and a pearl bead). Life span in wild is more than 30 years.
CONSERVATION STATUS
Not listed by the IUCN. Collection by diving has been illegal in French Polynesia since the collapse of the mother-of-pearl industry and the concurrent rise of the cultured pearl industry in the 1960s. Collecting has been illegal in Hawaii since 1930, where P. margaritifera was fished to near extinction for natural pearls at Pearl and Hermes Reef in a period of only three years (1927-1930). The population had still not recovered by the early 1990s.
SIGNIFICANCE TO HUMANS
Pinctada margaritifera is historically important as a primary source of mother-of-pearl for carving and inlay. The earliest cases from the Red Sea and Persian Gulf are from sites dating from ancient Sumeria (2300 B.C.) in Iraq and from the Greco-Roman era in Cyprus and Jerusalem. At present P. margaritifera is the source of black Tahitian pearls, both natural and cultured. The latter have been produced commercially and most extensively in French Polynesia and the Cook Islands since the 1960s.
European pearly mussel
Margaritifera margaritifera
ORDER
Unionioida
FAMILY
Margaritiferidae
TAXONOMY
Margaritifera margaritifera (Linnaeus, 1758), boreal (northern) Europe.
OTHER COMMON NAMES
English: Eastern pearl shell, freshwater pearl mussel, river pearl mussel; French: Moule perliere; German: Flufiperl-
muschel; Danish: Flodperlemusling; Japanese: Kawashinju-gai; Norwegian: Elvemusling; Swedish: Flodparlmussla.
PHYSICAL CHARACTERISTICS
Shell is elongated and oval in shape with a subcentral umbo; moderately inflated, not gaping, with brown adherent perios-tracum. Grows to 4-5 in (10-13 cm) in length. Interior is thickly nacreous. Soft body with accessory muscles attaching mantle to shell, with unfused mantle margin forming functional siphons; has no “lure” on mantle margin.
DISTRIBUTION
Freshwater streams in Europe, northwestern Asia and northeastern North America. Raised in aquaculture in Europe to restock diminished populations.
HABITAT
Infaunal; prefers cool, calcium-poor, fast-flowing freshwater streams lined with cobble and gravel. Lives partly buried in substrate in a near-vertical position with posterior end uppermost.
BEHAVIOR
Very sedentary. Can sense presence of host fish in vicinity, releasing clouds of glochidia larvae.
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Dioecious; males release sperm into water column; females collect sperm by normal filtering process; fertilization occurs within female. Larvae are brooded in all four demibranchs (half gills) and released as small hooked glochidia. “Parasitic” larval phase of M. margaritifera is dependent on salmonid fish (trout, salmon) as hosts; the glochidia attach to the host’s gills.
Reaches reproductive maturity at 12-15 yrs; maximum life span 130-200 years.
CONSERVATION STATUS
Harvesting for pearls in Europe was reserved for the church and aristocrats as early as the medieval period. Although early laws were prompted by the commercial value of pearls, they served to protect this long-lived species, most effectively in Saxony (Germany). Less efficiently managed areas (Scotland, Ireland, Norway, Sweden) experienced declines and extirpation. Additional declines have been attributed to poor water quality, habitat alteration, and declines in host fish populations. Classified as Endangered on the 2002 IUCN Red List. Listed on Appendix II of the 1979 Convention on the Conservation of European Wildlife and Natural Habitats.
SIGNIFICANCE TO HUMANS
Primary source of natural Eurasian freshwater pearls, especially in Germany, Scotland, and Russia; largely restricted to royal and religious use. Pearl growth requires 20 years or more in cold waters of native habitat.
Giant clam
Tridacna gigas
ORDER
Veneroida
FAMILY
Cardiidae
TAXONOMY
Tridacna gigas (Linnaeus, 1758), Amboina, Indonesia.

OTHER COMMON NAMES
English: Gigas clam; French: Benitier geant, tridacne geant.
PHYSICAL CHARACTERISTICS
Shell is heavy, fluted with 4—6 rather smooth folds; has central umbo and relatively small byssal notch. Color is whitish. Largest extant bivalve species; the largest existing specimen, in the American Museum of Natural History, is 4 ft, 5.9 in (136.9 cm) long and weighs 579.5 lbs (262.8 kg). Interior is porcelain white. Mantle is brightly colored, yellowish brown to olive-green with iridescent blue-green spots, fused except for the incurrent and excurrent siphons.
DISTRIBUTION
Southwestern Pacific from Philippines to Micronesia. Raised in aquaculture on various Pacific Islands for the aquarium trade.
HABITAT
Epibenthic; found on coral reefs, partly embedded in sand or rubble, at depths of 6—66 ft (2—20 m).
BEHAVIOR
Tridacha gigas is permanently sessile as adult, positioned hinge-down in reef, exposing open edge and mantle to sunlight. Valves remain widely gaping unless disturbed. Reaction to stimuli is rapid, although closure is slow due to great amount of water requiring discharge from mantle cavity. Larger specimens cannot completely close shells. Although little documented evidence exists, folklore claims that divers have drowned after catching a foot or hand in the closing “maws” of giant “killer” clams. Closing speed is slow and the bivalves certainly unaggressive, but the strength of closure and the sharpness of the valve edges can inflict serious injuries on unwary divers.
FEEDING ECOLOGY AND DIET
Mantle tissue exposed to sunlight supports photosynthesis of obligate commensal zooxanthellae (dinoflagellate algae, Symbio-dinium sp.), analogous to the condition in reef corals. The algae provide 90% of the host’s metabolized nutrients. T. gigas supplements nutrition derived from algae by filter feeding and uptake of dissolved organic matter (DOM).
REPRODUCTIVE BIOLOGY
Protandric hermaphrodite, reaching sexual maturity in 5—6 years; broadcast spawner, probably triggered by water temperature. Growth rate is estimated at 2 in (5 cm) per year in young individuals. Life span is uncertain; estimates vary wildely from several decades to 100 years.
CONSERVATION STATUS
Classified as Vulnerable by the IUCN. Populations have been reduced by overharvesting for food, the aquarium trade, and the curio trade. Listed on CITES Appendix II.
SIGNIFICANCE TO HUMANS
Human food source (adductor muscle), harvested by native populations on Pacific Islands. Shells historically used for making tools (mallets, hoes, scrapers); also used intact as water basins (and in churches worldwide as baptismal fonts, suggested by the French vernacular name). Non-nacreous pearls have little commercial value, although the largest pearl on record is the oblong “Pearl of Allah,” 9 in (22.9 cm) long and 14 lbs (6.35 kg) in weight, from a T. gigas specimen collected in the Philippines in 1934.
Coquina clam
Donax variabilis
ORDER
Veneroida
FAMILY
Donacidae
TAXONOMY
Donax variabilis (Say, 1822), Georgia and eastern Florida, United States.
OTHER COMMON NAMES
English: Bean clam, butterfly clam, donax clam, southern co-quina, variable coquina; French: Donax de Floride; Japanese: Kocyo-naminoko.
PHYSICAL CHARACTERISTICS
Shell is unequally triangular, with a subcentral umbo. The shorter anterior end is radially sculptured; the shell is otherwise smooth, not gaping. Polychromic, in wide variety of colors including white, yellow, orange, pink, purple, and blue; frequently radially striped. Interior is non-nacreous, often deep purple, with a denticulate (finely toothed) margin. Grows as long as 1 in (20 mm).
DISTRIBUTION
Eastern coast of North America from Chesapeake Bay to Florida; Gulf of Mexico to Yucatan.
HABITAT
Infaunal, on intertidal sandy beaches with wave action, sometimes numbering in thousands per square meter.
BEHAVIOR
Intertidal migration behavior is well documented. Coquina clams use their muscular foot to repeatedly rebury themselves after being washed from sand by incoming waves. Migration is both vertical (between tide levels) and horizontal (along the beach).
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Dioecious, broadcast spawner. Life span is about 1—2 years.
CONSERVATION STATUS
Not listed by the IUCN.
SIGNIFICANCE TO HUMANS
Human food source, locally in “coquina broth.” Coquina rock, a subfossil conglomerate of Donax shells and sand, was used as a building material by early Spanish settlers in North America. More recently, it has been used in ornamental landscaping.
Zebra mussel
Dreissena polymorpha
ORDER
Veneroida
FAMILY
Dreissenidae
TAXONOMY
Dreissena polymorpha (Pallas, 1771), Caspian Sea and Ural River, Russia.
OTHER COMMON NAMES
French: Moule zebree; German: Dreikantmuschel; Italian: Mitilo zebrato.
PHYSICAL CHARACTERISTICS
Shell is narrowly elongated and triangular in shape, as the animal’s German name suggests. The umbo lies at the pointed anterior end. The shell is rounded at the posterior end, inflated, smooth, narrowly gaping ventrally, with a sharp mid-valve keel producing a flattened ventral surface. The shell is variably banded with dark-brown-and-cream “zebra” markings. Grows to about 2 in (5 cm) in length. Has an internal shelflike septum at the umbo for muscle attachment. Soft body with short, separated siphons; produces byssal threads that exit the shell at a ventral byssal gape.
DISTRIBUTION
Native to the Black and Caspian Seas; introduced to western Europe through canals and inland waterways during the nineteenth century; currently established in most European countries. Introduced to North America in 1985 through ship ballast water released in the Great Lakes; has spread throughout the Mississippi River and other major U.S. drainages.
HABITAT
Epibenthic, in lakes, streams and estuaries of all sizes, byssally attached to any hard surface—including rocks, wood, boat hulls, waste materials, other zebra mussels and, importantly, native freshwater mussels, which are subsequently smothered. Found in waters 0-195 ft (0-60 m) in depth. Concentrated beds of zebra mussels can contain as many as 100,000 individuals per square yard (meter).
BEHAVIOR
Sessile; smaller individuals are mobile, frequently detaching byssus threads, moving to new locations and reattaching. Dispersal is facilitated by human-mediated mechanisms, especially the movement of vessels and release of ship ballast water containing larvae.
FEEDING ECOLOGY AND DIET
Efficient filter feeder, substantially reducing density of suspended matter in the water, which may have a detrimental effect on other filter-feeding organisms.
REPRODUCTIVE BIOLOGY
Dioecious, prolific broadcast spawner, with females releasing 40,000 to one million eggs per season. Free-swimming veliger (unusual for freshwater invertebrates) is initially lecithotrophic, later planktotrophic; larval life 18-28 days; settlement cues nonspecific. Life span about two years.
CONSERVATION STATUS
Not listed by the IUCN. Considered a nuisance species in Europe and North America.
SIGNIFICANCE TO HUMANS
Introduction and spread in Europe and North America have inflicted serious economic and environmental damage due to clogging of power plant intakes and competition with native pearl mussel populations. Attachment to boat motors, docks, buoys, and pipes also has negative impact on recreation industries. Wide-ranging scientific studies are focusing on the biology and ecology of zebra mussels in order to control their spread and proliferation in North America. ♦
Yo-yo clam
Divariscintilla yoyo
ORDER
Veneroida
FAMILY
Galeommatidae
TAXONOMY
Divariscintilla yoyo Mikkelsen and Bieler, 1989, Indian River Lagoon, Fort Pierce Inlet, Florida, United States.
OTHER COMMON NAMES
None known.
PHYSICAL CHARACTERISTICS
Animal is globular, about 0.5 in (10-15 mm) in length, translucent white, with a small wedge-shaped, fragile, permanently gaping internal shell covered nearly entirely by external mantle folds. Anterior cowl is wide and flaring; there are two long retractable anterodorsal “cephalic” tentacles and a single short pallial tentacle next to the excurrent siphon on the posterodor-sal midline. The foot is large and muscular with an elongated narrow posterior portion used in byssal attachment and “hanging” behavior. There are three to seven sensory flower-like organs on the visceral mass near the mouth.
DISTRIBUTION
Restricted to type locality, Indian River Lagoon in eastern Florida, United States.
HABITAT
Epibenthic; bysally attached to smooth walls of shallow-water sand burrows constructed by mantis shrimp Lysiosquilla scabricauda (Lamarck, 1818), in a commensal relationship.
BEHAVIOR
Yo-yo clams crawl in a snail-like fashion on a muscular foot, seeking a suitable vertical surface for attaching a short byssus thread secreted by a gland in the anterior portion of the foot. Once attached, the thread is picked up by a gland at the posterior tip of the foot, from which the animal then “hangs.” Periodic contractions of mantle muscles for clearing the mantle cavity of waste products jerk the entire animal in the manner of a yo-yo.
FEEDING ECOLOGY AND DIET
Filter feeder; presumably benefits from currents generated by host mantis shrimp in its burrow as well as feeding on particles from the shrimp’s predatory activities.
REPRODUCTIVE BIOLOGY
Simultaneous hermaphrodite; broods larvae in outer demi-branch and suprabranchial chamber; releases shelled, swimming veligers. Life span unknown.
CONSERVATION STATUS
Not listed by the IUCN, although restricted range near commercial harbor makes it particularly vulnerable to habitat alteration.
SIGNIFICANCE TO HUMANS
None known.
Giant vent clam
Calyptogena magnifica
ORDER
Veneroida
FAMILY
Kelliellidae
TAXONOMY
Calyptogena magnifica Boss and Turner, 1980, Galapagos Rift.
OTHER COMMON NAMES
English: Magnificent Calypto clam, vesicomyid clam.
PHYSICAL CHARACTERISTICS
Shell is oval with a subcentral umbo; moderately inflated with irregular concentric growth lines, slightly gaping. Color is white and chalky, with a yellow or brown periostracum persistent at margins only. Grows as long as 10 in (26 cm). Interior is porcelain white. Mantle and foot are iridescent pink; siphons short; visceral mass is red due to presence of hemoglobin in blood; byssus present only in juveniles.
DISTRIBUTION
Deep sea, on the Eastern Pacific Rise from 21°N to 22°S and the Galapagos Rift.
HABITAT
Endemic to hydrothermal vent systems of the tropical Pacific Ocean. Epibenthic; restricted to the sulfide-rich hydrothermal vent environment, nestled in crevices and among other mol-lusks around the vents at depths about 8,200 ft (2,500 m).
BEHAVIOR
Observed at hydrothermal vents maintaining nearly vertical position with posterior end upward; not bysally attached. Adults do not respond to crabs or shrimp crawling over shells, but respond experimentally to the manipulator arm of a submersible, suggesting they do not perceive crustaceans as predators. Oc-topods are probable predators.
FEEDING ECOLOGY AND DIET
Dependent upon energy produced by sulfur-oxidizing bacteria in gills. Alimentary tract reduced; ability to filter feed is questionable.
REPRODUCTIVE BIOLOGY
Reproductive mode incompletely known; probably broadcast spawner. Produces large yolky eggs, suggesting the larvae are lecithotrophic with limited dispersal capabilities (but carrying own food reserves), probably utilizing heat and/or sulfide as settling cues. Life span unknown.
CONSERVATION STATUS
Not listed by the IUCN. Relatively inaccessible; observed solely from deep-diving submersibles.
SIGNIFICANCE TO HUMANS
None known.
Northern quahog
Mercenaria mercenaria
ORDER
Veneroida
FAMILY
Veneridae
TAXONOMY
Mercenaria mercenaria (Linnaeus, 1758), Pennsylvania. One recognized brown-zigzagged color variety, M. m. var notata (Say, 1822), coast of the United States.
OTHER COMMON NAMES
English: Cherrystone clam, chowder clam, hard clam, hardshell clam, littleneck clam, quahog; French: Palourde americaine, praire, quahaug; German: Ostamerikanische Venusmuschel; Spanish: Almeja.
PHYSICAL CHARACTERISTICS
Shell is ovate-trigonal in shape, with the umbo curved toward the anterior end; moderately inflated, smooth with concentric growth lines, not gaping. Color is white or gray (with brown zigzag markings in form notata). Grows as long as 6 in (15 cm). Interior is porcelain white with a purple stain at posterior end; moderately deep pallial sinus (to contain retracted siphons). Soft body with muscular foot; mantle is fused to form siphons.
DISTRIBUTION
Eastern North America from Canada to Florida and the Gulf of Mexico. Introduced to California, England, and France. Raised in aquaculture in France and the southeastern United States.
HABITAT
Infaunal, buried in soft shelly substrates containing shells; less abundant in sand and mud. Found in depths from the inter-tidal zone to 50 ft (15 m). Tolerant of estuarine conditions.
BEHAVIOR
Generally sedentary.
FEEDING ECOLOGY AND DIET
Filter feeder.
REPRODUCTIVE BIOLOGY
Protandric hermaphrodite; broadcast spawner, with plank-totrophic larvae settling after two weeks. Reaches reproductive maturity at one year, with maximum life span perhaps 40 years.
CONSERVATION STATUS
Not listed by the IUCN. Collection for human consumption regulated by fisheries and public health agencies in most areas.
SIGNIFICANCE TO HUMANS
Human food source, both through wild harvest and aquaculture. Commercial categories (in order of increasing size) include seed clams, beans, buttons, littlenecks, topnecks, cherrystones, and chowders. Esteemed as food item, reflected in its status as the official state shell of Rhode Island. American aquaculture groups stress the brown-marked notata form, which bears the visible “brand” of a farm-raised product. Drilled shell pieces were historically produced as wampum beads by Native Americans in the eastern United States. Wampum beads were used within tribes for gifts, exchange, and ornaments but not as money; wampum served as currency, however, for European settlers in the original thirteen colonies as late as 1701. Merce-naria mercenaria is presently being investigated for the anti-cancer activity of its digestive gland. Also used in college-level zoology courses to study invertebrate anatomy.
