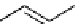Chemistry Reference
In-Depth Information
carboxylations. Furthermore, the carboxylic acids could be converted to esters or amides with good rCys (>40%) and
within short timeframes.
Ureas are common functional groups found in many drug-like molecules and therefore present valuable targets for C-11
labelling in the carbonyl position. Labelling of ureas has been previously achieved using C-11 phosgene and
11
CO (see
below); however, the use of
11
CO
2
for C-11 carbonyl urea synthesis is potentially more advantageous because of the avail-
ability of
11
CO
2
. A route to unsymmetrical carbonyl C-11 labelled ureas using
11
CO
2
has been recently developed using tri-
phenylphosphinine precursors [16]. initially, [
11
C]phenylisocyanate is formed via the reaction of triphenylphosphinimine
and
11
CO
2,
following reaction of the [
11
C]phenylisocyanate with a range of amines a series of small molecule [
carbonyl
-
11
C]
ureas were obtained. The highest rCys were achieved when basic primary amines were used (45-49%), but disappointingly
low yields were obtained when aniline was used as the amine source. This method may be useful for the preparation of a
wide range of [
11
C]ureas by variation of the phosphinimine and amine precursors; however, it has wider limitations due to
the sensitivity of phosphinimine reagents.
recently, the groups of Wilson [17, 18] and Fowler [19] have reported the use
11
CO
2
trapping reagents for the direct fixa-
tion of CO
2
to form labelled [
carbonyl
C-11] carbamate molecules. Both methods rely on the use of a strong amine base, DBU
(1,8-diazabicycloundec-7-ene) or BEMP (2-
tert
-butylimino-2-diethylamino-1,3-dimethylperhydro-1,3,2-diazaphosphorine),
to sequester and trap the
11
CO
2
from its nitrogen carrier gas stream. Exceptionally good trapping efficiencies were obtained
when model alkyl primary or secondary amines were used; however, yields diminished when aniline derivatives were
employed. Both groups were able to label PET imaging agents with high specific activities (>100 gBq μmol
-1
).
4.2.2
[
11
c]methylation reactions
Of the secondary labelling shown in Figure 4.1, [
11
C]methyl iodide is by far the most popular and commonly used C-11 pre-
cursor molecule. it is a highly versatile reagent capable of efficiently labelling a wide range of organic compounds. reliable
synthesis protocols and commercially available production units have further increased its popularity for C-11 labelling.
There are two common synthetic routes to generate
11
CH
3
i: the so-called 'wet method,' which is performed in an organic
solvent and involves LiAlH
4
reduction of
11
CO
2
followed by reaction with hydroiodic acid [20], or the 'gas phase' free rad-
ical iodination reaction of
11
CH
4
at high temperature [21, 22]. The gas phase method has proven to be most popular because
it of its ease of production and reliability. Today, several types of commercial units are available for
11
CH
3
i production. in
recent years, [
11
C]methyl triflate,
11
CH
3
OTf, has become an important alternative to
11
CH
3
i because of its much greater reac-
tivity [23], Typically,
11
CH
3
OTf will be used when
11
CH
3
i fails to give satisfactory yields [24-28]. [
11
C]methyl triflate can be
prepared by passing gaseous
11
CH
3
i through a column of silver triflate at 200 °C [29]. Other
11
C-alkyating agents have been
developed, such as [
11
C]ethyliodide, [
11
C]propyliodide, [
11
C]butyliodide, and [
11
C]benzyliodide; however, these have not
proven to be as popular as the methylating reagents [30, 31].
Nucleophilic substitution is the standard route by which
11
CH
3
is reacted with the precursor molecule to form a tracer.
N
-,
O
-, and
S
- methylation reactions of amines, alcohols, and thiols labelled primary or secondary amines, ethers, or thioethers
are common. Technically, this method is straightforward and usually involves passing a gas stream of
11
CH
3
i or
11
CH
3
OTf
through a basic solution of the precursor; the solution may occasionally need to be heated to improve reaction times.
Numerous
11
C-methylation procedures have been reported [32], and there are many technical variations of this method using
various automated systems or captive solvent 'loop' methods [33]. As mentioned above, all PET labelling reactions have a
vast stoichiometric excess of cold precursor reagent compared to the isotopically labelled precursor regent. This can be ben-
eficial for the labelling of amine precursors. Oversubstitution, typical of 'cold' stoichiometric scale amine-alkylation
reactions, is never observed at the concentrations used in typical PET labelling reactions. such labelling reactions always
result in exclusive labelling of only one C-11 methyl per molecule.
Methylation reactions are commonly used for the production of many of the key
11
C-tracers (scheme 4.5) including
Pittsburgh Compound B (PiB) [28, 34-36], raclopride (scheme 4.5) [33, 37, 38], [
11
C]
N
-methylspiperone [39], (NMsP) [40],
[
11
C]
N
-methylpiperidin-4-yl propinoate (PMP) [41], [
11
C]flumazenil [42], and [
11
C]carfentanil [43-45].
H
3
11
C
OH
O
O
O
11
CH
3
I
Cl
Cl
N
N
H
H
Room temperature
OH
OH
Cl
Cl
scheme 4.5
Formation of [
11
C]raclopride (
5
) via
O
-[
11
C]methylation reaction using
11
CH
3
i .