Chemistry Reference
In-Depth Information
R
R
H
H
R
n
n
n
(i) Co
2
(CO)
8
/CH
2
Cl
2
(ii) TMANO
O
O
O
O
+
R'
O
R
'
O
R'
n
R
R
R
n
n
75
Rac
-
76
Rac
-
77
Cl
Br
O
N
N
H
TDSO
NH
TDSO
N
N
N
O
O
O
7steps
(37 %)
7steps
(28 %)
TMS
O
OH
OH
Rac-
78
76a
Rac-
79
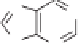
Scheme 4.20
Diastereoselective Pauson-Khand construction of bicyclic cyclopentenone
derivatives
76
.
These compounds are valuable building blocks for the synthesis of structurally di-
verse carbocyclic nucleoside analogs that have a high biological potential as apoptosis-
inducing agents. Starting from PK product
rac
-76
, the synthesis of 4
-trialkylsilyloxyethy
l-substituted nucleosides
rac
-78
and
rac
-79
with 5-bromouracil and 6-chloropurine as
nucleobases, respectively, was accomplished in seven steps (28 and 37% overall yields,
respectively)
Another example of a diastereoselective PK reaction was reported by Honda
et al.
in
2007.
23
They established a diastereoselective formal synthesis of a monoterpene alkaloid,
(-)-incarvilline
83
, that incorporates the intramolecular Pauson-Khand reaction of the cor-
responding enyne amide, that is, (
S
)-
N
-[(
E
)-2-butenyl]-
N
-(3-butynyl-2-methoxymethoxy)-
p-toluenesulfonamide,
80
. In this synthesis, the stereochemistry at the 7 and 7a positions
of the target compound was controlled through the reflection of the stereochemistry at the
4-position to providing the desired absolute configuration; this was achieved by employing
E-olefin as the starting material. The optimal result was obtained when the reaction was
carried out with 1.05 equivalents of Co
2
(CO)
8
in refluxing DCE in the presence of 3.5
equivalents of
tert
-butyl methyl sulfide as a promoter for 2.5 h under an atmosphere of CO
to furnish
81
and
82
in 73 and 8% yields, respectively (Scheme 4.21).
CH
3
OMOM
OMOM
H
H
H
OMOM
Co
2
(CO)
8
(1.05 eq
.
)
t-BuSMe (3.5 eq.)/
DCE
HO
O
O
+
NMe
NTs
NTs
NTs
H
H
H
H
3
C
H
3
C
H
3
C
(-)
-incarvilline
(
83
)
80
81
82
Scheme 4.21
Pauson-Khand reaction used in diastereoselective synthesis of (-)-incarvilline.
In the same year, the application of the PK reaction to a cyclopropene moiety was
described by Fox
et al.
24
In this study, the enantioselective synthesis of (-)-pentalenene
87