FAMILY BUNYAVIRIDAE
Other Orthomyxoviruses
Thogoto virus is present in regions of Africa, southern
The family Bunyaviridae contains more than 300 viruses
Europe, and Asia. It is a tick-borne virus that is primarily
grouped into five genera. A representative sampling of these
known from infection of livestock such as cattle, camels,
viruses is shown in Table 4.9. Members of four genera,
and sheep. There is significant amino acid sequence identity
Orthobunyavirus, Nairovirus, Phlebovirus, and Hantavirus,
between some of the Thogoto proteins and their counterparts
infect vertebrates and contain important human pathogens,
in influenza so these viruses are fairly closely related. It has
whereas viruses belonging to the genus Tospovirus infect
only six genomic segments, however. Human infection is
plants. The human pathogens in the family variously cause
known to occur in endemic areas.
hemorrhagic fever, hantavirus pulmonary syndrome which
Infectious salmon anemia virus infects salmonid fish.
can be fatal, encephalitis, or milder febrile illnesses, as
Atlantic salmon are particularly susceptible to the virus and
shown in the table. Some of these pathogens were listed in
the virus is a particular problem for fish farming. Farm pop-
Table 3.11, which contains a partial listing of arboviruses that
ulations can suffer 100% mortality in outbreaks. The virus
cause disease in humans. All members of the Bunyaviridae
has eight gene segments.
TABLE 4.9 Bunyaviridae
Virus name
Transmission/
Disease in
World
a
Genus/members
abbreviation
Usual host(s)
vector
humans
distribution
Orthobunyavirus (~48 viruses)
Bunyamwera
BUNV
Rodents, rabbits
Aedes mosquitoes
Febrile illness
Worldwide
La Crosse
LACV
Humans, rodents
Aedes triseriatis
Encephalitis
Midwest United States
Snowshoe hare
SSHV
Lagomorphs
Mosquitoes (Culiseta
Rarely infects
Northern United States
and Aedes)
humans
California encephalitis
CEV
Rodents, rabbits
Aedes melanimon,
Encephalitis (rare)
Western United States,
Ae. dorsalis
Canada
Jamestown Canyon
JCV
White-tailed deer
Aedes species,
Increasing
North America
C. inornata
Hantavirus (~22 viruses)
Hantaan
HTNV
Apodemus agrarius
Feces, urine, saliva
Hemorrhagic fever
Eastern Asia,
Eastern Europe
Seoul
SEOV
Rattus species
Feces, urine, saliva
Hemorrhagic fever
Worldwide
Prospect Hill
PHV
Microtus
?
None?
United States
pennsylvanicus
Sin Nombre
SNV
Peromyscus
Feces, urine, saliva
Pulmonary syndrome
Western United States
maniculatus
and Canada
Nairovirus (~7 viruses)
Dugbe
DUGV
Sheep, goats
Tick-borne
?
Africa
Crimean-Congo
C-CHFV
Humans, cattle,
Tick-borne
Hemorrhagic fever
Africa, Eurasia
hemorrhagic fever
sheep, goats
Phlebovirus (~9 viruses)
Rift Valley fever
RVFV
Sheep, humans,
Mosquitoes, also
Hemorrhagic fever
Africa
cattle, goats
contact, aerosols
Sandfly fever Sicilian
SFSV
Humans
Phlebotomous flies
Nonfatal febrile
Mediterranean
illness
Uukuniemi
UUKV
Birds
Tick-borne
??
Finland
Tospovirus (~8 viruses)
Tomato spotted wilt
TSWV
Plants
Thrips
None
Australia, Northern
hemisphere
a
Representative members of each genus are shown; the first virus listed is the type species.
except the hantaviruses are transmitted to their vertebrate
All bunyavirus genomes consist of three segments of RNA,
or plant hosts by arthropods, and transovarial transmission
referred to as S(mall), M(edium), and L(arge), that together
is important in the maintenance of many of the arboviruses
total from 11 to 19 kb, depending on the virus (Table 4.10).
in nature. The hantaviruses, in contrast, are associated with
The S segment encodes the nucleocapsid protein, M the
rodents and are transmitted to humans by aerosolized excreta
two surface glycoproteins, and L the polymerase protein.
from infected rodents. Thus, their epidemiology resembles
In addition, viruses belonging to three of the genera encode
that of the arenaviruses considered later, rather than that of
two nonstructural proteins, NSs in segment S and NSm in
other bunyaviruses. In the following discussion, the term
seg-ment M.
bunyavirus refers to any member of the family unless indi-
Replication of bunyavirus genomes and the synthesis
cated otherwise.
of mRNAs take place in the cytoplasm. The L protein and
N protein are required components of the RNA synthesis
machinery. Like influenza viruses, these viruses engage in
Replication of the Bunyaviridae
cap-snatching in order to prime mRNA synthesis. In bun-
yaviruses, however, the caps are captured from cytoplasmic
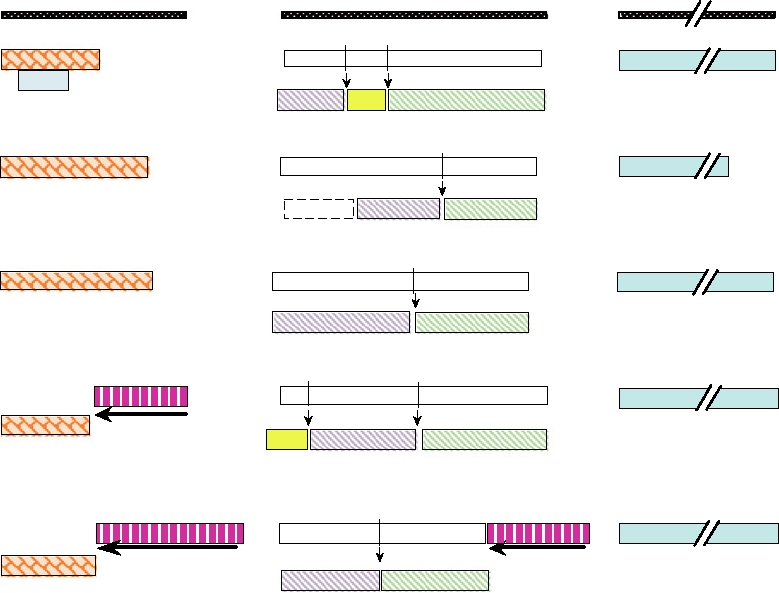
Genome Organization
mRNAs rather than from nuclear pre-mRNAs. The promoter
The genomes of representative viruses belonging to the
for mRNA synthesis and for RNA replication involves nucle-
five genera of the Bunyaviridae are illustrated in Fig. 4.21.
otides located at both ends of the genomic template, which
Minus strand
S RNA
M RNA
L RNA
genome segments
(0.96-2.9 kb)
(3.2-4.8 kb)
(6.4-12.2kb)
(range of sizes in kb)
5
3
5
3
5
3
N
ORTHOBUNYA-
VIRUS
Co-translational cleavage
L
(BUNV)
NSs
Nested reading frames
NSm
Gn
Gc
NAIROVIRUS
Co-translational cleavage
(C-CHFV)
N
L
?
Gn
Gc
HANTAVIRUS
(HTNV)
Post-translational cleavage
N
L
Gn
Gc
NSs
C
N
PHLEBOVIRUS
(RVFV)
L
Co-translational cleavage
N
Ambisense transcription and translation NSm
Gn
Gc
NSm
NSs
C
N
C
N
TOSPOVIRUS
(TSWV)
L
Co-translational cleavage
N
Gn
Gc
Ambisense transcription and translation
Ambisense transcription and translation
FIGURE 4.21 Genome organization of five genera of Bunyaviridae. Protein products encoded in each of the three genome
segments and the various strategies used to produce these proteins are shown. Unless otherwise noted the mRNA (not shown)
would extend 5' to 3' from left to right, and the protein product is shown N terminal to C terminal in the same direction. The
products are illustrated roughly to scale. Structural proteins are N (the nucleocapsid protein) and the two glycoproteins,
named Gn and Gc according to their proximity to the N or C termini of the precursor polyprotein; white boxes are precursor
proteins. Arrows indicate the direction of synthesis and translation of ambisense mRNAs (magenta hatch). Note that not all
phleboviruses encode an NSm protein on the M segment. Virus abbreviations are as follows: BUNV, Bunyamwera; RVFV, Rift
Valley fever; TSWV, tomato spotted wilt; C-CHFV, Crimean-Congo hemorrhagic fever; HTNV, Hantaan.
Deduced Sizes (kD) of Proteins Encoded by Bunyaviruses
TABLE 4.10
Genus
RNA (nts)
Protein (kD)
Orthobunyavirus
Hantavirus
Nairovirus
Phlebovirus
Tospovirus
L Segment
68756980
65506562
12,255
64046423
87768897
L protein (RNA Polymerase)
259263
246247
459
238241
330332
M Segment
44584526
36163696
4888
32314215
48214972
Glycoprotein Gn
2941
6876
3045
5072
4658
Glycoprotein Gc
108120
5258
7284
5575
7278
Precursor preG
--
--
7885, 92115
--
--
Nonstructural NSm
1518
None
None
None or 78
3437
S Segment
961980
16962059
1712
16901869
29162992
Nucleoprotein N
1026
4854
4854
2430
29
Nonstructural NSs
1013
None
None
2932
52
Sizes of precursor proteins are shown in blue, nonstructural proteins are shown in green, and those translated from ambisense transcripts are in red.
are complementary and form hairpin circles, but with some
otides so that the mRNA attached to the primer begins pre-
cisely at the 3¢ end of the template. This "prime and realign"
unpaired nucleotides that are thought to be important recog-
nition signals (Table 4.11). Thus, as with many other RNA
strategy (Fig. 4.22) works because of the repeat triplets at the
3¢ end of the RNA template (Table 4.11). Transcription con-
viruses, the viral RNA polymerase must interact with both
ends of the RNA template in order to initiate synthesis, and
tinues to near the end of the template, but the termination of
complementarity between nucleotides at the 5¢ and 3¢ ends
an mRNA does not appear to be precise and the exact mech-
is required for promoter recognition. The terminal comple-
anism used for termination of the mRNAs is not known. No
poly(A) is added to the 3¢ end of the mRNA upon its release
mentary sequences are highly conserved within each genus
of bunyaviruses but differ between genera (Table 4.11).
and, thus, the mRNAs are capped but not polyadenylated.
Perhaps because of this, reassortment occurs only between
During replication of the genome, an exact complemen-
viruses belonging to the same genus.
tary copy, called cRNA or vcRNA, is produced. This RNA
During initiation of mRNA, as studied in hantaviruses,
serves as a template for producing genomic RNA and, in
the L protein cleaves the 5¢-terminal 718 nucleotides from
the case of ambisense segments, for producing the mRNA
a cellular mRNA. Cleavage is after a G residue, which pairs
for producing the ambisense-encoded protein. The switch to
with the C residue at position 3. The primer is elongated by
replication is assumed to use the same mechanisms as used
a few residues, and there is then a backward shift of 3 nucle-
by other (-)RNA viruses.
TABLE 4.11
Terminal Sequences of the Genome Segments
Expression of Proteins Encoded in S
of the Five Genera of Bunyaviridae
The S segment of bunyaviruses encodes one or two pro-
Nucleotide sequences of the L, M,
teins (Fig. 4.21). In the Hantavirus and Nairovirus genera,
Genus
and S segments
S encodes only N. In the other genera, S encodes both N and
NSs, using one of two different mechanisms. In the genus
5¢ U A G U A G U A ...
Hantavirus
Orthobunyavirus, the two proteins are translated from a
3′ A U C A U C A U C U G ...
single mRNA using two different start codons in different
5¢ U C A U C A C A U G A ...
Orthobunyavirus
reading frames. The coding region for NSs is completely
3′ A G U A G U G U G C U ...
contained within that for N. In the phleboviruses and tospo-
5¢ A G A G U U U C U ...
Nairovirus
viruses, however, an ambisense coding strategy is used for
3′ U C U C A A A G A ...
the two proteins (ambi = both). In this strategy, the two
5¢ U G U G U U U C ...
Phlebovirus
genes encoded in a genomic segment are linked tail to tail
3′ A C A C A A A G ...
so that they are in different polarities, as illustrated in Fig.
4.23. The gene for N is present at the 3¢ end of the genomic
5¢ U C U C G U U A ...
Tospovirus
3′ A G A G C A A U ...
S segment in the minus-sense orientation, and synthesis
of the mRNA for N occurs from the genome segment.
Expression of this gene occurs early because its mRNA is
Repeated sequences are underlined.
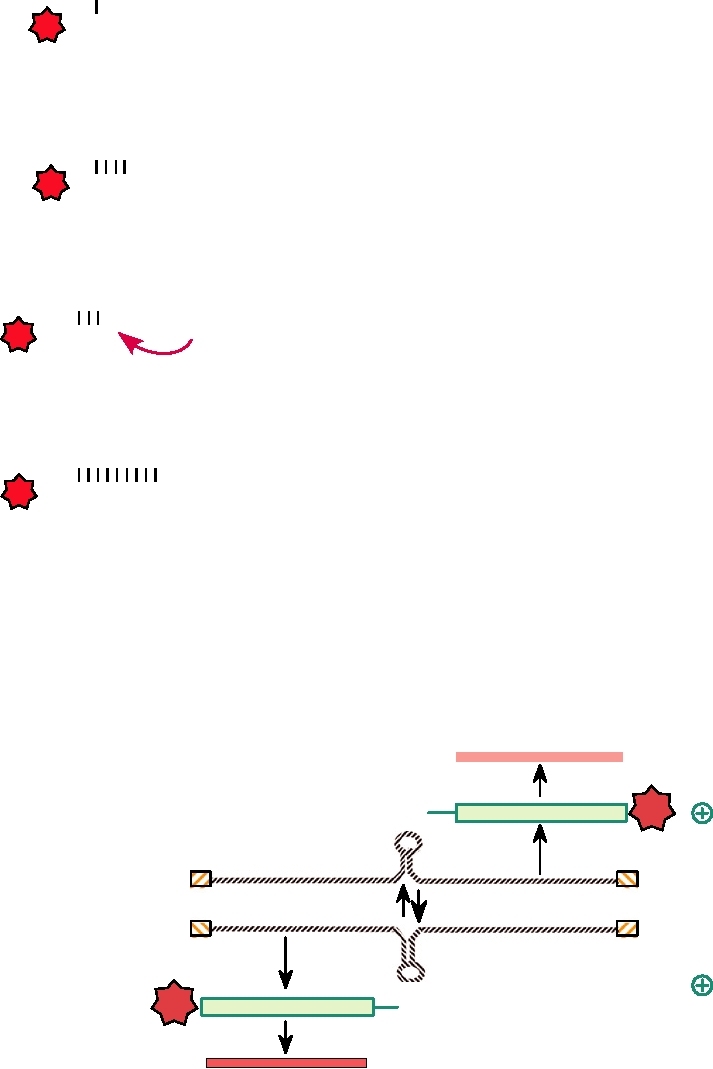
Step 1 - Priming
synthesized from the entering genome by the polymerase
activity present in viral nucleocapsids. The gene for NSs is
3'
AUCAUCAUC...
3' terminus of minus-strand
plus sense within the genome, but the genomic RNA does
genome RNA
not serve as mRNA. Instead, an mRNA for NSs is synthe-
...G
CAP
Capped host-derived primer
sized from the antigenomic RNA. Thus, NSs is expressed
late because its mRNA can only be made after replication
of the incoming genomic RNA to produce the antigenomic
Step 2 - Initial Elongation
RNA. Termination of either mRNA occurs at a secondary
structure between the genes for N and NS, which appears
3'
3' terminus of minus-strand
AUCAUCAUC...
to cause the polymerase to fall off and release the mRNA.
genome RNA
. . . GUAG
N has a number of functions in viral infection. It encap-
Nascent mRNA
CAP
sidates the viral RNA, interacts with L to synthesize viral
RNAs, and is believed to interact with one of the glyco-
proteins during virus assembly. In at least some viruses
Step 3 - Realigning
the protein also modifies cellular metabolism, presumably
3'
AUCAUCAUC...
antagonizing antiviral defenses of the cell or otherwise sub-
verting cellular processes to support viral replication. The N
. . . G UAG
CAP
protein of hantaviruses, which is larger than those of other
bunyaviruses except for that of the nairoviruses, interacts
with a number of cellular proteins. The best studied of these
Step 3 - Final Elongation
are proteins in the small ubiquitin-like protein (SUMO)
3'
pathways such as Ubc-9 (which conjugates SUMOs to pro-
3' terminus of minus-strand
AUCAUCAUC...
teins), SUMO-1 itself, and Daxx (to which SUMOs are con-
genome RNA
. . . G UAGUAGUAG...
Nascent mRNA
CAP
jugated). Sumolation of proteins is an important regulatory
process in cellular metabolism.
FIGURE 4.22 Steps in the primealign mechanism of transcription
The NSs protein of Rift Valley fever virus, and presumably of
of mRNAs by hantaviruses. This mechanism is made possible by the
other viruses as well, inhibits host mRNA synthesis, including
nucleotide repeats in the 3′ and 5′ termini of bunyaviruses (see Table 4.11).
the mRNA for interferon-α and -β. Thus it suppresses the host
Adapted from Kukkonen et al. (2005).
immune response and is a major virulence factor.
NSs protein
Translation
39
59
CAP
NSsmRNA
mRNA
59
39
synthesis
vcRNA
Replication
39
59
Genome RNA
mRNA
synthesis
NmRNA
CAP
39
59
Translation
N protein
FIGURE 4.23
Ambisense coding strategy of the S RNA of a phlebovirus, family Bunyaviridae. The mRNA for the
N protein is synthesized from the S genome segment using primers derived by cap-snatching (similar to the mechanism
for influenza mRNA priming in Fig. 4.13) from cytoplasmic host mRNAs. The mRNA for the NSs protein is formed in
the same way, but with vcRNA as the template. Diagonally striped boxes are the self-complementary termini. The loops
in the middle of the viral genomic and antigenomic RNAs indicate a secondary structure in the RNAs which terminates
synthesis of the mRNAs. No poly(A) is added to the 3¢ terminus of the mRNAs.
came from a different bunyavirus. This reassortant virus,
Expression of Proteins Encoded in M
named Garissa virus, thus caused a disease different from
Two glycoproteins, at one time usually called G1 and G2
Bunyamwera virus, perhaps due to the different properties
but now called GN and GC, are translated from mRNA made
of the M segment.
from M (Fig. 4.21). They are produced as a polyprotein that
Viruses belonging to the California encephalitis group,
is cleaved by a cellular protease to separate the two glyco-
of which La Crosse virus is the best known, are also of
proteins, analogous to what happens in some of the (+)RNA
medical interest. La Crosse virus was named for the town
viruses that have envelopes (e.g., coronaviruses and flavi-
of La Crosse, Wisconsin, where it was first identified as the
viruses). GN is N terminal in the polyprotein and GC is C
causative agent of encephalitis, primarily in children. About
terminal. Where studied, the enzyme responsible for the
100 cases per year of encephalitis are caused by La Crosse
cleavage is a subtilase, SKI-1/S1P or a related enzyme. GN
virus, concentrated in the Midwest. Mortality is low (0.3%)
and GC form a heterodimer that is transported to the Golgi
but 10% of patients suffer neurological sequelae. No vac-
apparatus. Virus budding occurs at the Golgi membrane.
cine exists for the virus and control measures have involved
Heterodimerization recalls the processes that occur in the
control of the mosquito vector. The principal vector of La
assembly of alphaviruses and flaviviruses, and like E1 or
Crosse is Aedes triseriatus. This mosquito breeds in tree
E of these viruses, respectively, the GC protein is a class II
holes, but abandoned tires filled with rainwater constitute
fusion protein.
an important breeding area for it close to human habitation.
The M segments of hantaviruses and nairoviruses encode
Such abandoned tires serve as a beautiful incubator for the
only the two glycoproteins, but in the other three genera M
development of mosquito larvae, and efforts to eliminate
encodes a third protein called NSm (Fig. 4.21 and Table 4.10).
this source of mosquitoes, as well as the institution of other
In phleboviruses and members of the genus Bunyavirus,
mosquito control measures, has resulted in a reduction in the
NSm forms part of the polyprotein translated from the single
number of cases of disease.
mRNA produced from M. NSm is formed during posttrans-
Abandoned tires are important in the transmission
lational processing of the polyprotein. In tospoviruses, an
of other arboviruses as well. Old tires are abundant in
ambisense strategy is used to encode NSm and the translation
Puerto Rico, for example, and contribute to the endemic
strategy is the same as that shown in Fig. 4.23. The function
transmission of dengue virus, all four serotypes of which
of NSm is not known.
are present on the island. Old tires have also been respon-
There is no matrix protein in bunyaviruses. Budding at
sible for the introduction into the United States of Aedes
the Golgi membrane is assumed to involve a direct interac-
albopictus, the so-called Asian tiger mosquito that is the
tion between the glycoproteins and the nucleocapsid pro-
vector of dengue virus in Asia. Loads of old tires that
tein. The virion is spherical, 80120 nm in diameter. The
were brought from Asia to Houston for recycling con-
three nucleocapsids are circular when isolated from the
tained eggs or larvae of the mosquito. After its introduc-
virion.
tion into the Houston area, this mosquito spread over
large areas of the United States and there is fear that it
might become an efficient vector of arboviral disease in
Genus Orthobunyavirus
this country.
There are about 50 currently recognized species in the
genus Orthobunyavirus, of which the majority have several
Genus Phlebovirus
distinct strains that are often given separate names. Together,
these viruses have a worldwide distribution. Most of these
The ICTV currently recognizes 9 species of phlebovi-
viruses are mosquito-borne, although some are tick-borne
ruses but there are an additional 16 tentative species. Many
and some may be transmitted by culicoid flies or phleboto-
of the species have a number of strains that are given their
mines. They are true arboviruses, replicating in the arthro-
own names. All are arboviruses transmitted by mosquitoes,
pod vector as well as in vertebrates.
phlebotomine flies, or ticks. The most important of these is
Bunyamwera virus, the prototype member of the genus,
Rift Valley fever virus, an African virus that was first iso-
was first isolated in Uganda in 1943. It causes a febrile ill-
lated in 1930 in the Rift Valley of East Africa. The virus
ness accompanied by headache, arthralgia, rash, and occa-
is transmitted by mosquitoes and causes hemorrhagic fever
sional nervous system involvement. There are 24 named
in humans. It also causes disease in domestic animals, and
subtypes in the bunyamwera serogroup. Of interest is a
many widespread epidemics in cattle, sheep, and humans
reassortant virus isolated during an epidemic of hemor-
have occurred over the years in Africa. In 19771978, for
rhagic fever in Kenya and Somalia in 1998. Rift Valley
example, an epizootic in Egypt infected 2550% of cattle
fever virus was responsible for some of the cases, but many
and sheep in some areas, and 200,000 human cases resulted
were caused by a bunyavirus whose L and S segments were
in at least 600 deaths. A more recent large epidemic in
derived from a Bunyamwera virus but whose M segment
East Africa in 19971998 was associated with the heavi-
est rainfall in 35 years, 60100 times normal in some areas.
species have a number of named strains. Many hantaviruses
As described before, the epidemic was caused by Rift Valley
cause serious human disease, including hemorrhagic fevers
fever virus and by Garissa virus. Losses of 70% of sheep and
and hantavirus pulmonary syndrome. Unlike other members
goats and 2030% of cattle and camels were reported, and
of the Bunyaviridae, they are not arboviruses. The hantavi-
there were hundreds of cases of human hemorrhagic fever.
ruses are associated with rodents, which form their natural
Contact with livestock was statistically associated with
reservoir, and are transmitted to humans through contact
acute infection with Rift Valley fever virus, indicating that
with aerosolized urine or feces from infected rodents. Each
during epidemics contact transmission becomes important
hantavirus establishes persistent infections in one particular
as a means of spread to humans. Laboratory-acquired cases
species of rodent and is maintained in nature in this way.
contracted through aerosols are also known.
Humans are not an important host for the virus and do
Sand fly fever virus is transmitted by phlebotomine flies
not contribute to its maintenance in nature. Related to this
and causes an acute, nonfatal influenza-like disease in man.
is the fact that the viruses do not cause serious disease in
It is found in the Mediterranean area, North Africa, and
their rodent hosts, but many cause quite serious illness in
Southwest Asia. Related viruses are found in South America.
humans.
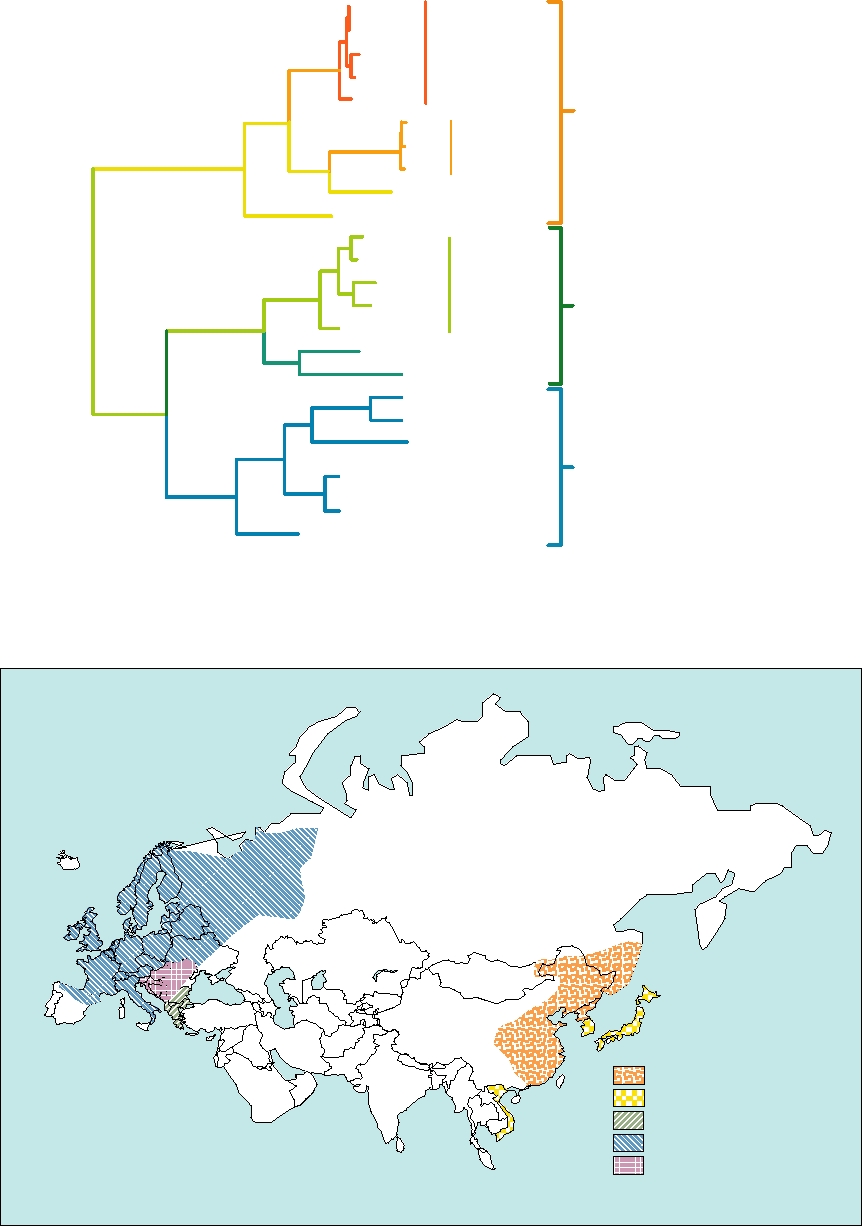
An evolutionary tree of hantaviruses is shown in Fig.
4.24. The rodent hosts for the viruses are also indicated.
Genus Nairovirus
The viruses assort by host rather than by geographical
The nairoviruses have a much larger genome than mem-
proximity. All of the viruses whose hosts belong to the
bers of the other genera, primarily because the L segment is
order Murinae group together, as do those that use rodents
twice the size of those of the other genera of animal viruses
in the order Arvicolinae and those that use rodents in the
(Table 4.10). They are named for Nairobi sheep disease
order Sigmodontinae. As one example, consider Prospect
virus, now considered a strain of Dugbe virus. There are
Hill virus and New York virus, both found in the north-
seven species recognized, all of which consist of multi-
eastern United States. Prospect Hill virus is associated
ple strains with distinct names. The viruses are tick-borne
with rodents of the genus Microtus, order Arvicolinae, and
although a few can also be transmitted by culicoid flies or
is more closely related to Puumala virus of Europe, which
mosquitoes. Nairobi sheep disease virus causes acute gas-
uses Clethrionomys glareolus, order Arvicolinae, than it
troenteritis with hemorrhagic symptoms in sheep and goats,
is to New York virus. New York virus is associated with
with mortality rates over 90% in some populations. It was
rodents in the genus Peromyscus, order Sigmodontinae and
first identified as the causative agent of the disease in 1917
is closely related to Sin Nombre virus of the southwestern
and is transmitted by the tick Rhipicephalus appendicula-
United States, which is associated with Peromyscus man-
tus. Humans can be infected by the virus but suffer only
iculatus. The fact that the evolutionary tree of the hantavi-
mild illness. There is a close relative of the virus called
ruses resembles that of their rodent hosts rather than being
Ganjam virus present in India which also causes disease in
based on geographical proximity is evidence that they
sheep and goats; it is transmitted by the tick Haemaphysalis
have coevolved with their rodent hosts over a very long
intermedia.
period of time.
Crimean-Congo hemorrhagic fever virus (CCHF) is the
The first of the hantaviruses to be identified was the caus-
most important nairovirus in terms of human disease. It was
ative agent of more than 3000 cases of hemorrhagic fever
first identified in the 1940s in the Crimean region of the
with renal syndrome, now called Korean hemorrhagic fever,
former USSR and in the Democratic Republic of Congo. The
that occurred in U.S. troops during the Korean war. The
virus is now known from at least 30 countries. It is found from
virus was called Hantaan virus after a river in the area where
southern Africa through Eastern Europe and the Middle East
it was isolated. In Korea, Hantaan virus is associated with
to western China. The principal vector is Hyalomma ticks,
the field mouse Apodemus agrarius. The virus also occurs
but Dermacentor and Rhipicephalus ticks can also transmit
in Eastern Europe and China, where it is associated with
the virus. Sheep, goats, cattle, ostriches, wild herbivores, and
Apodemus flavicollis and causes a disease similar to Korean
hares become infected by CCHF but most infections result in
hemorrhagic fever (Fig. 4.25).
subclinical disease. In contrast, infection of humans results in
Viruses related to Hantaan virus have now been isolated
severe hemorrhagic fever with a 30% mortality rate. Humans
from all over the world, including the Americas. Many Old
are infected by the bite of a tick or by contact with blood
World viruses cause hemorrhagic fever in humans, and more
or tissues of infected livestock. Transmission to hospital
than 100,000 cases occur worldwide with a case fatality rate
personnel treating infected patients has occurred.
between 0.1 and 10%, depending on the virus. Puumula virus
occurs in Western Europe (Fig. 4.25) and causes a disease
characterized by acute fever with renal involvement. Seoul
Genus Hantavirus
virus, first identified in Seoul, Korea, is associated with wild
There are 22 species of hantaviruses currently recognized,
urban rats (Rattus norvegicus) and has been found all over
and, as with other genera of the bunyaviruses, many of the
the world because wild urban rats have been inadvertently
Rodent-borne Hantaviruses
Rodent hosts
76-118
cumc-b11
Hantaan
hojo
isolates
lee
hv114
Murinae
b1
(Old world rats and mice,
*
Seoul
sr-11
found in Europe and Asia)
isolates
80-39
Thailand
Dobrava
vindeln
vranica
Puumala
cg1820
isolates
Arvicolinae
sotkamo
(Voles; found in Europe,
90-13
Asia, and the Americas)
Tula
Prospect Hill
Bayou
Black Creek Canal
Laguna Negra
Sigmodontinae
Sin Nombre
(New World rats and mice,
found only in the Americas)
New York
El Moro Canyon
FIGURE 4.24 Phylogenetic tree of rodent-borne hantaviruses derived from the nucleotide sequence of the M RNA
segment. This tree illustrates that hantaviruses have coevolved with their rodent hosts for millions of years. However, note
(*) that in contrast to other members of this group, Seoul virus, which infects Rattus norvegicus, is found worldwide, due
to the widespread distribution of these rats. Adapted from Peters (1998a), Figure 2.
Hantaan virus
Seoul virus
Variant Hantaan
Puumala virus
Variant Hantaan
and Puumala
FIGURE 4.25 Map of Eurasia showing the disjunct distribution of different hantaviruses. Adapted from Porterfield
(1995) p. 276 and data from Lee (1996).
introduced almost everywhere. It causes a mild form of
is associated with the deer mouse Peromyscus maniculatus.
Korean hemorrhagic fever in Seoul but does not cause
Sin Nombre virus or related viruses have now been identi-
apparent illness in most other areas where it has been found.
fied in virtually all states within the United States and into
The discovery of Seoul virus led to an intensive study of rats
Latin America, and fatalities due to infection by the virus
in central Baltimore, where it was found that a high percent-
have occurred in many states. One of the cases in California
age of them were infected with Seoul virus and, furthermore,
is of interest because the person died more than a year before
that a substantial fraction of the people living in the slums
the Four Corners epidemic; retrospective studies of serum
of downtown Baltimore showed evidence of infection by
collected from the patient at the time of his hospitalization
hantavirus. No disease is known to be associated with this
showed that he was infected with a hantavirus. The number
virus, but statistical studies suggest that infection may lead
of cases of HPS in the Americas from 1993 to 1998, totaled
to high blood pressure and, possibly, renal failure.
by country, and the names of the viruses responsible in vari-
The New World hantaviruses that cause disease in humans
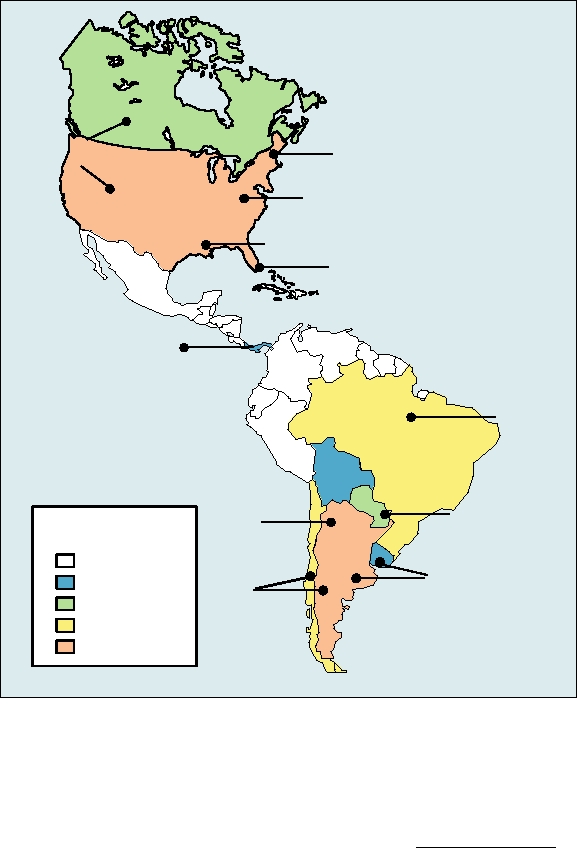
ous areas are shown in Fig. 4.26. Of interest is Andes virus,
cause a syndrome called hantavirus pulmonary syndrome or
which has the potential for human-to-human transmission.
HPS, which has a fatality rate of 2040%. The first such
The mortality rate following infection with Sin Nombre
virus to be identified was Sin Nombre virus, which caused
virus or its close relatives is close to 50%. The mortality in
an epidemic of HPS in the Four Corners area of the United
the earliest cases was even higher because the pulmonary
States in 1993 that resulted in about 25 deaths. The virus
syndrome results from the rapid extravasation of fluids into
Canada
Sin Nombre
New York
Monongahela
United States
Bayou
Black Creek Canal
Choclo
Panama
Juquitiba
Brazil
Bolivia
Paraguay
Laguna Negra
Number of HPS cases
Oran
Argentina
1993-2004
Uruguay
No report
Lechiguanas
11-50
Andes
51-150
151-350
Chile
>351
FIGURE 4.26 Cases of hantavirus pulmonary syndrome (HPS) in the Americas, with locations and names of the viruses
responsible. Case numbers are cumulative totals from the time that HPS was recognized in 1993 in the Four Corners region
of Arizona and New Mexico through 2004. Although several other hantaviruses have been isolated in this region, only
those which have been identified as human pathogens are shown. To the current time, cases of HPS have been diagnosed
in the United States in 30 states, predominantly in the Western United States, with a few cases as far East as Rhode Island.
The total number of cases represented on this map is 1910. Adapted from Peters (1998a) Figure 1 and Table 3, and updated
with data from Yates et al. (2002), and the Pan American Health Organization at http://www.paho.org/.
Search WWH :