years; once symptoms appear, the animal dies within weeks.
the polyprotein and functions both in cis and in trans. This
Animals that die of this disease are found to be infected by
protease requires a virally encoded cofactor, NS2B in fla-
viviruses and NS4A in pestiviruses and hepaciviruses. Npro
two types of BVDV. One is the normal wild-type virus,
which is noncytopathic in cultured cells. The second type of
is an autoprotease whose only known cleavage in normal
BVDV is a new strain that is cytopathic in cultured cells. The
infection is to release itself from the polyprotein. The NS2
cytopathic BVDV strain is derived from the wild-type strain
protease described here also functions as an autoprotease
by recombination, which occurs during the persistent infec-
that makes only one cleavage, that between NS2 and NS3.
tion. Several different cytopathic BVDV strains have been
CSFV is epidemic in pig populations and causes serious
sequenced, and they all have in common that NS23 (for-
illness, with different isolates differing in their virulence.
merly called p125) is cleaved to produce NS3 (also called
Infection of pregnant sows can lead to abortion or to birth of
p80). It is the production of NS3 that renders the virus cyto-
persistently infected piglets, which soon die. BDV also can
lytic in culture and causes lethal mucosal disease in cattle. As
cause congenital infection, which can lead to abortion or to
illustrated in Fig. 3.37, the cleavage to produce NS3 can be
birth of animals that display a number of defects.
induced in several different ways. In at least three cytopathic
BVDV strains, cellular ubiquitin sequences were inserted (in
FAMILY CORONAVIRIDAE
different ways) within the sequence encoding this protein,
such that a cellular enzyme that cleaves specifically after
ubiquitin cleaves the BVDV polyprotein to produce NS3.
The name Coronaviridae comes from the Latin word
Another mechanism to produce NS3 was the insertion of the
meaning crown, from the appearance of the array of spikes
BVDV Npro autoprotease immediately upstream of the NS3
around the enveloped virion. The family is composed of a
sequence. A third mechanism, not illustrated in the figure,
number of RNA-containing animal viruses currently clas-
is the insertion of cellular sequences derived from a protein
sified into two genera, the genus Coronavirus (whose
called Jiv.
members will here be called coronaviruses) and the genus
Why the production of NS3 renders the virus cytopathic
Torovirus (whose members will be referred to as torovi-
and capable of causing lethal disease in cattle is a fascinating
ruses). A representative listing of viruses in the two genera
story of self-imposed limitation on virus growth not unlike
is found in Table 3.13. The family is classified together with
the story of alphavirus downregulation described earlier in
the Arteriviridae and the Roniviridae (described later) in the
this chapter. For BVDV to persist in nature it must be able
Order Nidovirales, after the Latin word nido meaning nest,
to establish persistent infection because persistently infected
because they produce a nested set of mRNAs. Coronaviruses
animals that continue to shed the virus are an important res-
are somewhat larger in size (120160 nm) than the torovi-
ervoir for the virus. Cytopathic viruses are not able to estab-
ruses (120140 nm) and have a larger genome (about 30 kb
lish persistent infection and come to a dead end when they
compared to 20 kb). In contrast to other (+)RNA viruses,
arise. It turns out that cleavage to form NS3 is essential for
the nucleocapsids of Coronaviridae are constructed using
virus replication, and cleavage occurs early after infection
helical symmetry. The coronaviruses have a helical nucleo-
by all BVDV strains, noncytopathic as well as cytopathic.
capsid 1020 nm in diameter, whereas the toroviruses have
Cleavage is effected by a protease in NS2, and this protease,
a tubular nucleocapsid that appears toroidal in shape in the
like the NS3 protease of all members of the family, requires
virion. The coronavirus virion is roughly spherical, whereas
a cofactor for function. This cofactor is a cellular protein,
the torovirus virion is disk shaped or rod shaped. The viruses
however, not a virally encoded protein. This cellular cofac-
mature by budding through intracytoplasmic membranes.
tor is the protein Jiv, which forms a stable (but noncovalent)
The coronaviruses have been well studied, whereas the toro-
complex with NS23. The amounts of Jiv in the infected cell
viruses, which are composed of one pathogen of horses, one
are limited; however, and it is soon titrated out. Once no free
pathogen of cattle, a presumptive human torovirus, and a
Jiv remains, NS23 cleavage cannot occur and no further
possible torovirus of swine, have attracted less attention.
increase in viral replication is possible, allowing the estab-
lishment of persistent infection with only limited amounts of
Genus Coronavirus
virus being produced. If NS23 cleavage continues to occur
because new protease sites have been introduced or because
The coronaviruses have the largest RNA genome known,
the virus encodes its own Jiv, virus replication continues to
2732 kb in size. The genome size of RNA viruses is thought
accelerate until it overwhelms the cell and the cell dies. As
to be limited by the mutation rate during RNA synthesis.
an aside, it is possible that the virus host range is controlled
Because there is no proofreading during RNA synthesis, an
inherent mistake frequency results that is in the order of 10-4.
by the presence or absence in cells of sufficient Jiv able to
act as a cofactor.
Thus, error-free replication of an RNA genome becomes
Thus, pestiviruses encode three proteases. The NS3 pro-
impossible once the genome becomes too large. The 30-kb
tease common to all Flaviviridae makes many cleavages in
genome of coronaviruses may represent this upper limit. It is
TABLE 3.13 Coronaviridae
Virus name
Usual
World
Genus/members
abbreviation
host(s)
Transmission
Disease
distribution
Coronavirus
Group 1
Transmissable gasteroenteritis
TGEV
Swine
Contact
Gastroenteritis
United States, Europe
Human coronaviruses 229,
HCoV
Humans
Aerosols
Common cold
Americas, Europe
NL63
Group 2A
Human coronaviruses OC43, HKU-1
HCoV
Humans
Aerosols
Common cold
Americas, Europe
Murine hepatitis
MHV
Mice
Aerosols, contact
Gastroenteritis, hepatitis
Laboratory mouse
colonies worldwide
Group 2B
Batsa, Humans
Severe acute repiratory
SARS
Aerosols, contact
Fever, pneumonia,
Asia, Americas
syndrome
severe respiratory
disease
Group 3
Infectious bronchitis
IBV
Birds
Mechanical,
Bronchitis
Worldwide
oralfecal
Torovirus
Berne (equine torovirus)
EqTV
Horses
Oralfecal
Diarrhea
Europe, Americas
Breda (bovine torovirus)
BoTV
Cattle
Oralfecal
Diarrhea
?
Human torovirus
HuTV
Humans
?
Diarrhea
?
a
Bats have been identified as the vertebrate reservoir, but disease is primarily in humans.
also possible that because the coronaviruses undergo high-
infectious bronchitis virus, turkey coronavirus, and recently
frequency recombination, as described later, they may be
described viruses of geese, pigeons, and ducks. Where
able to accommodate these large genomes because recombi-
known, the viruses in these different groups use different
nation offers a possible mechanism for correcting defective
receptors to enter cells (see Table 1.3). A number of group
genomes. Intriguingly, coronaviruses and other members
1 viruses use aminopeptidase N, also called CD13. Several
of the Nidovirales encode a number of RNA-processing
group 2A viruses are known to use carcinoembryonic anti-
enzymes including a 3′-to-5′ exonuclease that could con-
gen-related adhesion molecules, which are members of the
ceivably make proofreading possible during RNA replica-
Ig superfamily. SARS virus uses angiotensin-converting
tion. However, there is as yet no evidence that the mutation
enzyme 2.
frequency during coronaviral RNA replication is less than
that occurring during replication of other RNA viruses.
Translation of the Viral Genome:
The coronaviruses are grouped into three clades called
The Nonstructural Proteins
groups 1, 2, 3, and examples are given in Table 3.13.
Assignments were first based on serological cross-reactivity
The coronavirus genome is, as in the case of all plus-
but more recently on sequence relatedness. Group 1 viruses
strand RNA viruses, a messenger, and the naked RNA is in-
include porcine epidemic diarrhea virus, porcine trans-
fectious. The organization of the 27.6-kb genome of avian
missible gastroenteritis virus, canine coronavirus, feline
infectious bronchitis virus (IBV) is shown in Fig. 3.38 as
infectious peritonitis virus, and two human viruses, human
an example for the genus. The RNA, which is capped and
coronaviruses 229E and NL63. Group 2 viruses are subdi-
polyadenylated, is translated into two polyproteins required
vided into two clades. Group 2A contains murine hepatitis
for the replication of the viral RNA and the production of
virus (MHV), bovine coronavirus, rat sialodacryoadeni-
subgenomic mRNAs. The first polyprotein terminates at a
stop codon 12.4 kb from the 5′ end of the RNA. Ribosomal
tis virus, porcine hemagglutinating encephalomyelitis virus,
canine respiratory coronavirus, equine coronavirus, and one
frameshifting occurs frequently, however, and in the shift-
human virus, human coronavirus OC43. Group 2B con-
ed frame, translation continues to the end of the RNA
tains severe acute respiratory syndrome coronavirus (SARS
replicase-encoding region at 20.4 kb. The resulting polypro-
HCoV). Group 3 contains a number of avian viruses, avian
teins are cleaved by virus-encoded proteases, as illustrated in
ORF1b
ORF1a
CAP
An
Genome
RNA
An
S
An
E
An
mRNAs
M
An
An
N
0
2
10
20
22
24
26
28
kilobases
p100
p87
p35
Protease Cleavages
Enzyme Motifs
Coding Domains
Polymerase (GDD)
pro 2
Nonstructural proteins
Helicase
pro 2 (predicted)
Nucleocapsid protein
pro1
Zinc finger
Virion transmembrane
3C-like cysteine protease (pro2)
proteins
Papain protease (pro1)
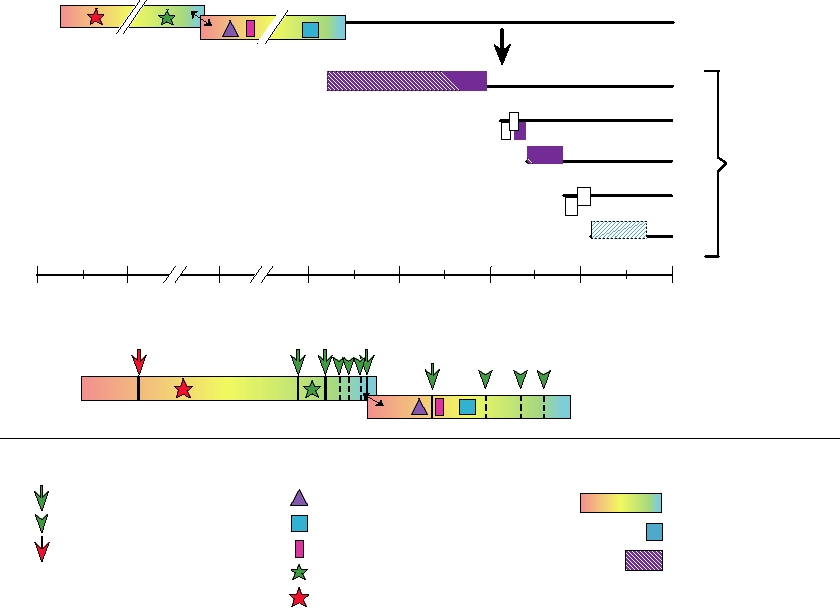
FIGURE 3.38 Upper panel: genome organization of the coronavirus, avian infectious bronchitis virus (IBV). ORF1a and
ORF1b encode components of the viral replicase, and are translated as two polyproteins, with ribosomal frameshifting at
the double-headed arrow. The remaining viral components are encoded in a nested set of mRNAs. The hatched proteins are
polypeptides found in virions. White boxes are open reading frames of unknown function. E is a minor virion component,
but essential for virus assembly. Lower panel: proteolytic processing of the IBV ORF1ab polyprotein. Motifs of papain-like
proteases (pro1), 3C-like cysteine protease (pro2), RNA polymerase (GDD), zinc finger, and helicase are indicated with
various symbols. Arrows at cleavage sites are color coded according to the protease responsible. Green arrowheads are
predicted cleavage sites for pro2. Adapted from de Vries et al. (1997) with permission.
Fig. 3.38B. All coronaviruses possess at least two proteases,
an RNA polymerase, an RNA helicase, and enzymes involved
one papain-like and the other serine-like (but with cysteine
in capping that must perform functions similar to the cor-
at the active site as in poliovirus), and some encode a sec-
responding enzymes in other (+)RNA viruses. The papain-
ond papain-like protease so that they encode three proteases.
like protease, however, has another function in addition to
Processing is complicated, as indicated in the figure.
processing some of the sites in the nonstructural polyprotein.
Nidoviruses differ fundamentally from other RNA viruses
It is a deubiquitinating enzyme (DUB) whose precise role in
in the number of nonstructural enzymes that they encode for
virus infection is unknown. Ubiquitin and ubiquitin-like pro-
the synthesis of the viral RNAs or for the purpose of enabling
teins (UBLs) are small proteins that are covalently attached
vigorous viral replication. The size of the RNA devoted to
to other proteins by ubiquitinating enzymes, either as single
encoding these proteins in the coronaviruses is 2030 kb,
molecules or as branched chains. The role of ubiquitination
larger than the entire genome of other RNA viruses, and the
is only incompletely understood but plays an important role
number of cleaved products produced from the polyprotein
in many cellular processes. One role of ubiquitination is to
precursors is large, on the order of 16. Perhaps the large size
target proteins for degradation by the proteosome, a well-
of the genome requires this. It is known that at least some
studied phenomenon. Ubiquination is also involved in mem-
of these proteins are devoted to countering host defenses
brane protein trafficking, in the activation of the transcription
factor NFκB, in DNA repair, and in autophagy, a response to
against viral infection, which is surely important for the per-
sistence of the viruses in nature.
starvation in which double membrane structures are assem-
The nonstructural proteins encoded in this domain of the
bled that might serve as viral replication sites. Thus a viral
genome include the two or three proteases described before,
DUB might stabilize proteins that enhance viral replication,
or might be important for the induction of NFκB, an impor-
synthetase to reinitiate synthesis at any of the several sub-
tant transcription factor during viral infection, or it might be
genomic promoters in the (-)RNA template. Evidence for
important for constructing viral replication sites. There are
this model includes the fact that each subgenomic RNA has
at its 5′ end the same 60 nucleotides that are present at the 5′
also at least 10 UBLs derived from the same common ances-
tor as ubiquitin that are also conjugated to proteins to control
end of the genomic RNA, and that there is a short sequence
cellular activities in ways that are but incompletely under-
element present at the beginning of each gene that could act as
stood. One of these is the product of interferon-stimulated
an acceptor for the primer (this sequence, e.g., is ACGAAC
gene 15 (ISG15). This protein is induced by interferon and
in the SARS CoV). A recent model proposes that the bulk of
plays an unknown role in regulating the immune response to
the subgenomic mRNAs are produced by independent repli-
viral infection. It is not known if the viral DUB might also
cation of the subgenomic RNAs as replicons. Such replica-
remove conjugated ISG15, but it is known that some other
tion is thought to be possible because the mRNAs contain
both the 5′ and 3′ sequences present in the genomic RNA,
viruses target ISG conjugation. Influenza B virus produces
a protein that binds to ISG, preventing the ISGlation of pro-
and therefore possess the promoters required for replication.
teins. In addition, African swine fever virus, a large DNA
Evidence for this model includes the fact that both plus-sense
virus, has a DUB that is thought to block the production of
and minus-sense subgenomic RNAs are present in infected
interferon by unknown mechanisms.
cells. The model favored is that the subgenomic RNAs are
In addition to these gene products, nidoviruses encode
first produced during synthesis of minus-strand RNA from
the genomic RNA. In this model, synthesis initiates at the 3′
distant homologues of at least five cellular enzymes associ-
end of the genome and then jumps to the 5′ leader at one of
ated with RNA processing. These are an endoribonuclease
that cleaves after uridine residues, the 3′-to-5′ exonuclease
the junctions between the genes. Once produced, the subg-
mentioned earlier, a methyltransferase that might be part of
enomic RNAs begin independent replication.
the capping complex, an adenosine diphosphate-ribose 1′-
Coronaviruses undergo high-frequency recombination
phosphatase, and cyclic phosphodiesterase. Most coronavi-
in which up to 10% of the progeny may be recombinant. It
ruses encode all five of these enzymes whereas roniviruses
is proposed that the mechanism for generation of the sub-
encode only three and arteriviruses only one. The functions
genomic RNAs, which requires the polymerase to stop at
of these enzymes in the virus life cycle are unknown.
defined sites and then reinitiate synthesis at defined promot-
ers, may allow the formation of perfect recombinants at high
frequency.
Production of Subgenomic RNAs
The members of the Nidovirales produce a nested set of
Envelope Glycoproteins
subgenomic mRNAs (Fig. 3.38), which are capped and poly-
adenylated. The number produced depends on the virus but is
Coronaviruses possess three envelope proteins--a spike
5 to 8 for most. Each subgenomic RNA is a messenger that is
protein (S), a membrane protein (M), and an envelope pro-
translated into one to three proteins from the 5′ ORF(s) in the
tein (E). The spike protein is a large protein (e.g., 1255 resi-
mRNA. The five subgenomic mRNAs of IBV and the pro-
dues in the SARS CoV) that is heavily glycosylated (more
teins translated from them are illustrated in Fig. 3.38A. Four
than 10 carbohydrate chains attached) and anchored in the
of the subgenomic mRNAs are translated into the structural
membrane of the virion by a transmembrane domain near
proteins in the virion, S, E, M, and N, found in that order
the C terminus, with a C-terminal cytoplasmic tail of about
in the genomes of all coronaviruses. Four small accessory
40 residues. It forms trimers that project from the surface
proteins of unknown function are also produced, two from
of the membrane and give coronaviruses their characteristic
the E mRNA and two from RNA 5. Coronaviruses encode
corona. These spikes possess the receptor-binding activity,
variable numbers of such accessory proteins which are not
the major neutralizing epitopes, and the fusion activity of the
conserved as to sequence or to number among the various
virion. S contains two domains of about equal size called S1
members of the family and whose function in unknown. It is
(N terminal) and S2 (C terminal), and in some, but not all,
also not known how multiple proteins are translated from a
coronaviruses these two domains are separated into different
single mRNAs in the case of the coronaviruses.
proteins by proteolytic cleavage of S. S1 contains the recep-
Two mechanisms have been proposed for the production
tor-binding region and S2 contains the fusion domain. S is
of these subgenomic RNAs. The first mechanism proposed
not well conserved, with only about 30% sequence identity
was primer-directed synthesis from the (-)RNA template
among S proteins of coronaviruses belonging to different
(i.e., from the antigenome produced from the genomic RNA).
groups.
In this model, a primer of about 60 nucleotides is transcribed
The M protein is smaller, 221 residues in SARS CoV,
from the 3′ end of the template, which is therefore identical
and spans the lipid bilayer three times such that it has only
to the 5′ end of the genomic RNA. The primer is proposed to
a small fraction of its mass exposed outside the bilayer. The
dissociate from the template and to be used by the viral RNA
E protein is quite small, only 76 residues in SARS CoV, and
Search WWH :