CONCEPT
One of the most amazing aspects of physics is the electromagnetic spectrum—radio waves, microwaves, infrared light, visible light, ultraviolet light, x rays, and gamma rays—as well as the relationship between the spectrum and electromagnetic force. The applications of the electromagnetic spectrum in daily life begin the moment a person wakes up in the morning and “sees the light” Yet visible light, the only familiar part of the spectrum prior to the eighteenth and nineteenth centuries, is also its narrowest region. Since the beginning of the twentieth century, uses for other bands in the electromagnetic spectrum have proliferated. At the low-frequency end are radio, short-wave radio, and television signals, as well as the microwaves used in cooking. Higher-frequency waves, all of which can be generally described as light, provide the means for looking deep into the universe—and deep into the human body.
HOW IT WORKS
ELECTROMAGNETISM
The ancient Romans observed that a brushed comb would attract particles, a phenomenon now known as static electricity and studied within the realm of electrostatics in physics. Yet, the Roman understanding of electricity did not extend any further, and as progress was made in the science of physics—after a period of more than a thousand years, during which scientific learning in Europe progressed very slowly—it developed in areas that had nothing to do with the strange force observed by the Romans.
The fathers of physics as a serious science, Galileo Galileo (1564-1642) and Sir Isaac Newton (1642-1727), were concerned with gravitation, which Newton identified as a fundamental force in the universe. For nearly two centuries, physicists continued to believe that there was only one type of force. Yet, as scientists became increasingly aware of molecules and atoms, anomalies began to arise—in particular, the fact that gravitation alone could not account for the strong forces holding atoms and molecules together to form matter.
Foundations of electromagnetic theory
At the same time, a number of thinkers conducted experiments concerning the nature of electricity and magnetism, and the relationship between them. Among these were several giants in physics and other disciplines—including one of America’s greatest founding fathers. In addition to his famous (and highly dangerous) experiment with lightning, Benjamin Franklin (1706-1790) also contributed the names “positive” and “negative” to the differing electrical charges discovered earlier by French physicist Charles Du Fay (1698-1739).
In 1785, French physicist and inventor Charles Coulomb (1736-1806) established the basic laws of electrostatics and magnetism. He maintained that there is an attractive force that, like gravitation, can be explained in terms of the inverse of the square of the distance between objects. That attraction itself, however, resulted not from gravity, but from electrical charge, according to Coulomb.
A few years later, German mathematician Johann Karl Friedrich Gauss (1777-1855) developed a mathematical theory for finding the magnetic potential of any point on Earth, and his contemporary, Danish physicist Hans Christian Oersted (1777-1851), became the first scientist to establish the existence of a clear relationship between electricity and magnetism. This led to the foundation of electromagnetism, the branch of physics devoted to the study of electrical and magnetic phenomena.
French mathematician and physicist Andre Marie Ampere (1775-1836) concluded that magnetism is the result of electricity in motion, and, in 1831, British physicist and chemist Michael Faraday (1791-1867) published his theory of electromagnetic induction. This theory shows how an electrical current in one coil can set up a current in another through the development of a magnetic field. This enabled Faraday to develop the first generator, and for the first time in history, humans were able to convert mechanical energy systematically into electrical energy.
Maxwell and electromagnetic force.
A number of other figures contributed along the way; but, as yet, no one had developed a “unified theory” explaining the relationship between electricity and magnetism. Then, in 1865, Scottish physicist James Clerk Maxwell (1831-1879) published a groundbreaking paper, “On Faraday’s Lines of Force,” in which he outlined a theory of electromagnetic force— the total force on an electrically charged particle, which is a combination of forces due to electrical and/or magnetic fields around the particle.
Maxwell had thus discovered a type of force in addition to gravity, and this reflected a “new” type of fundamental interaction, or a basic mode by which particles interact in nature. Newton had identified the first, gravitational interaction, and in the twentieth century, two other forms of fundamental interaction—strong nuclear and weak nuclear—were identified as well.
In his work, Maxwell drew on the studies conducted by his predecessors, but added a new statement: that electrical charge is conserved. This statement, which did not contradict any of the experimental work done by the other physicists, was based on Maxwell’s predictions regarding what should happen in situations of electro-magnetism; subsequent studies have supported his predictions.
ELECTROMAGNETIC RADIATION
So far, what we have seen is the foundation for modern understanding of electricity and magnetism. This understanding grew enormously in the late nineteenth and early twentieth centuries, thanks both to the theoretical work of physicists, and the practical labors of inventors such as Thomas Alva Edison (1847-1931) and Serbian-American electrical engineer Nikola Tesla (18561943). But our concern in the present context is with electromagnetic radiation, of which the waves on the electromagnetic spectrum are a particularly significant example.
Energy can travel by conduction or convection, two principal means of heat transfer. But the energy Earth receives from the Sun—the energy conveyed through the electromagnetic spectrum—is transferred by another method, radiation. Whereas conduction of convection can only take place where there is matter, which provides a medium for the energy transfer, radiation requires no medium. Thus, electromagnetic energy passes from the Sun to Earth through the vacuum of empty space.
Electromagnetic waves.
The connection between electromagnetic radiation and electromagnetic force is far from obvious. Even today, few people not scientifically trained understand that there is a clear relationship between electricity and magnetism—let alone a connection between these and visible light. The breakthrough in establishing that connection can be attributed both to Maxwell and to German physicist Heinrich Rudolf Hertz (18571894).
Maxwell had suggested that electromagnetic force carried with it a certain wave phenomenon, and predicted that these waves traveled at a certain speed. In his Treatise on Electricity and Magnetism (1873), he predicted that the speed of these waves was the same as that of light— 186,000 mi (299,339 km) per second—and theorized that the electromagnetic interaction included not only electricity and magnetism, but light as well. A few years later, while studying the behavior of electrical currents, Hertz confirmed Maxwell’s proposition regarding the wave phenomenon by showing that an electrical current generated some sort of electromagnetic radiation.
In addition, Hertz found that the flow of electrical charges could be affected by light under certain conditions. Ultraviolet light had already been identified, and Hertz shone an ultraviolet beam on the negatively charged side of a gap in a

The Hubble Space Telescope includes an ultravidlet light instrument called the Goddard High Resolution Spectrograph that it is capable of observing extremely distant objects.
current loop. This made it easier for an electrical spark to jump the gap. Hertz could not explain this phenomenon, which came to be known as the photoelectric effect. Indeed, no one else could explain it until quantum theory was developed in the early twentieth century. In the meantime, however, Hertz’s discovery of electromagnetic waves radiating from a current loop led to the invention of radio by Italian physicist and engineer Guglielmo Marconi (1874-1937) and others.
Light: Waves or Particles?
At this point, it is necessary to jump backward in history, to explain the progression of scientists’
understanding of light. Advancement in this area took place over a long period of time: at the end of the first millennium A.D., the Arab physicist Alhasen (Ibn al-Haytham; c. 965-1039) showed that light comes from the Sun and other self-illuminated bodies—not, as had been believed up to that time—from the eye itself. Thus, studies in optics, or the study of light and vision, were— compared to understanding of electromagnetism itself—relatively advanced by 1666, when Newton discovered the spectrum of colors in light. As Newton showed, colors are arranged in a sequence, and white light is a combination of all colors.
Newton put forth the corpuscular theory of light—that is, the idea that light is made up of particles—but his contemporary Christiaan Huygens (1629-1695), a Dutch physicist and astronomer, maintained that light appears in the form of a wave. For the next century, adherents of Newton’s corpuscular theory and of Huygens’s wave theory continued to disagree. Physicists on the European continent began increasingly to accept wave theory, but corpuscular theory remained strong in Newton’s homeland.
Thus, it was ironic that the physicist whose work struck the most forceful blow against corpuscular theory was himself an Englishman:
Thomas Young (1773-1829), who in 1801 demonstrated interference in light. Young directed a beam of light through two closely spaced pinholes onto a screen, reasoning that if light truly were made of particles, the beams would project two distinct points onto the screen. Instead, what he saw was a pattern of interference—a wave phenomenon.
By the time of Hertz, wave theory had become dominant; but the photoelectric effect also exhibited aspects of particle behavior. Thus, for the first time in more than a century, particle theory gained support again. Yet, it was clear that light had certain wave characteristics, and this raised the question—which is it, a wave or a set of particles streaming through space?
The work of German physicist Max Planck (1858-1947), father of quantum theory, and of Albert Einstein (1879-1955), helped resolve this apparent contradiction. Using Planck’s quantum principles, Einstein, in 1905, showed that light appears in “bundles” of energy, which travel as waves but behave as particles in certain situations. Eighteen years later, American physicist Arthur Holly Compton (1892-1962) showed that, depending on the way it is tested, light appears as either a particle or a wave. These particles he called photons.
Wave Motion and Electromagnetic Waves
The particle behavior of electromagnetic energy is beyond the scope of the present discussion, though aspects of it are discussed elsewhere. For the present purposes, it is necessary only to view the electromagnetic spectrum as a series of waves, and in the paragraphs that follow, the rudiments of wave motion will be presented in short form.
A type of harmonic motion that carries energy from one place to another without actually moving any matter, wave motion is related to oscillation, harmonic—and typically periodic— motion in one or more dimensions. Oscillation involves no net movement, but only movement in place; yet individual waves themselves are oscillating, even as the overall wave pattern moves.
The term periodic motion, or movement repeated at regular intervals called periods, describes the behavior of periodic waves: waves in which a uniform series of crests and troughs follow each other in regular succession. Periodic waves are divided into longitudinal and transverse waves, the latter (of which light waves are an example) being waves in which the vibration or motion is perpendicular to the direction in which the wave is moving. Unlike longitudinal waves, such as those that carry sound energy, transverse waves are fairly easy to visualize, and assume the shape that most people imagine when they think of waves: a regular up-and-down pattern, called “sinusoidal” in mathematical terms.
Parameters of wave motion
A period (represented by the symbol T) is the amount of time required to complete one full cycle of the wave, from trough to crest and back to trough. Period is mathematically related to several other aspects of wave motion, including wave speed, frequency, and wavelength.
Frequency (abbreviated f) is the number of waves passing through a given point during the interval of one second. It is measured in Hertz (Hz), named after Hertz himself: a single Hertz (the term is both singular and plural) is equal to one cycle of oscillation per second. Higher frequencies are expressed in terms of kilohertz (kHz; 103 or 1,000 cycles per second); megahertz (MHz; 106 or 1 million cycles per second); and gigahertz (GHz; 109 or 1 billion cycles per second.)
Wavelength (represented by the symbol X, the Greek letter lambda) is the distance between a crest and the adjacent crest, or a trough and an adjacent trough, of a wave. The higher the frequency, the shorter the wavelength; and, thus, it is possible to describe waves in terms of either. According to quantum theory, however, electromagnetic waves can also be described in terms of photon energy level, or the amount of energy in each photon. Thus, the electromagnetic spectrum, as we shall see, varies from relatively long-wavelength, low-frequency, low-energy radio waves on the one end to extremely short-wavelength, high-frequency, high-energy gamma rays on the other.
The other significant parameter for describing a wave—one mathematically independent from those so far discussed—is amplitude. Defined as the maximum displacement of a vibrating material, amplitude is the “size” of a wave. The greater the amplitude, the greater the energy the wave contains: amplitude indicates intensity. The amplitude of a light wave, for instance, determines the intensity of the light.
A right-hand rule
Physics text topics use a number of “right-hand rules”: devices for remembering certain complex physical interactions by comparing the lines of movement or force to parts of the right hand. In the present context, a right-hand rule makes it easier to visualize the mutually perpendicular directions of electromagnetic waves, electric field, and magnetic field.
A field is a region of space in which it is possible to define the physical properties of each point in the region at any given moment in time. Thus, an electrical field and magnetic field are simply regions in which electrical and magnetic components, respectively, of electromagnetic force are exerted.
Hold out your right hand, palm perpendicular to the floor and thumb upright. Your fingers indicate the direction that an electromagnetic wave is moving. Your thumb points in the direction of the electrical field, as does the heel of your hand: the electrical field forms a plane perpendicular to the direction of wave propagation. Similarly, both your palm and the back of your hand indicate the direction of the magnetic field, which is perpendicular both to the electrical field and the direction of wave propagation.
THE ELECTROMAGNETIC SPECTRUM
As stated earlier, an electromagnetic wave is transverse, meaning that even as it moves forward, it oscillates in a direction perpendicular to the line of propagation. An electromagnetic wave can thus be defined as a transverse wave with mutually perpendicular electrical and magnetic fields that emanate from it.
The electromagnetic spectrum is the complete range of electromagnetic waves on a continuous distribution from a very low range of frequencies and energy levels, with a correspondingly long wavelength, to a very high range of frequencies and energy levels, with a correspondingly short wavelength. Included on the electromagnetic spectrum are radio waves and microwaves; infrared, visible, and ultraviolet light; x rays, and gamma rays. Though each occupies a definite place on the spectrum, the divisions between them are not firm: as befits the nature of a spectrum, one simply “blurs” into another.
Frequency range of the electromagnetic spectrum.
The range of frequencies for waves in the electromagnetic spectrum is from approximately 102Hz to more than 1025 Hz. These numbers are an example of scientific notation, which makes it possible to write large numbers without having to include a string of zeroes. Without scientific notation, the large numbers used for discussing properties of the electromagnetic spectrum can become bewildering.
The first number given, for extremely low-frequency radio waves, is simple enough—100— but the second would be written as 1 followed by 25 zeroes. (A good rule of thumb for scientific notation is this: for any power n of 10, simply attach that number of zeroes to 1. Thus 106 is 1 followed by 6 zeroes, and so on.) In any case, 1025 is a much simpler figure than
10,000,000,000,000,000,000,000,000—or 10 trillion trillion. As noted earlier, gigahertz, or units of 1 billion Hertz, are often used in describing extremely high frequencies, in which case the number is written as 1016 GHz. For simplicity’s sake, however, in the present context, the simple unit of Hertz (rather than kilo-, mega-, or gigahertz) is used wherever it is convenient to do so.
Wavelengths on the electromagnetic spectrum
The range of wavelengths found in the electromagnetic spectrum is from about 108 centimeters to less than 10-15 centimeters. The first number, equal to 1 million meters (about 621 mi), obviously expresses a great length. This figure is for radio waves of extremely low frequency; ordinary radio waves of the kind used for actual radio broadcasts are closer to 105 centimeters (about 328 ft).
For such large wavelengths, the use of centimeters might seem a bit cumbersome; but, as with the use of Hertz for frequencies, centimeters provide a simple unit that can be used to measure all wavelengths. Some charts of the electromagnetic spectrum nonetheless give figures in meters, but for parts of the spectrum beyond microwaves, this, too, can become challenging. The ultra-short wavelengths ofgamma rays, after all, are equal to one-trillionth of a centimeter. By comparison, the angstrom—a unit so small it is used to measure the diameter of an atom—is 10 million times as large.
Energy levels on the electromagnetic spectrum.
Finally, in terms of photon energy, the unit of measurement is the electron volt (eV), which is used for quantifying the energy in atomic particles. The range of photon energy in the electromagnetic spectrum is from about 10-13 to more than 1010 electron volts. Expressed in terms of joules, an electron volt is equal to 1.6 • 10-19 J.
To equate these figures to ordinary language would require a lengthy digression; suffice it to say that even the highest ranges of the electromagnetic spectrum possess a small amount of energy in terms of joules. Remember, however, that the energy level identified is for a photon— a light particle. Again, without going into a great deal of detail, one can just imagine how many of these particles, which are much smaller than atoms, would fit into even the smallest of spaces. Given the fact that electromagnetic waves are traveling at a speed equal to that of light, the amount of photon energy transmitted in a single second is impressive, even for the lower ranges of the spectrum. Where gamma rays are concerned, the energy levels are positively staggering.
REAL-LIFE APPLICATIONS
The Radio Sub-Spectrum
Among the most familiar parts of the electromagnetic spectrum, in modern life at least, is radio. In most schematic representations of the spectrum, radio waves are shown either at the left end or the bottom, as an indication of the fact that these are the electromagnetic waves with the lowest frequencies, the longest wavelengths, and the smallest levels of photon energy. Included in this broad sub-spectrum, with frequencies up to about 107 Hertz, are long-wave radio, short-wave radio, and microwaves. The areas of communication affected are many: broadcast radio, television, mobile phones, radar—and even highly specific forms of technology such as baby monitors.
Though the work of Maxwell and Hertz was foundational to the harnessing of radio waves for human use, the practical use of radio had its beginnings with Marconi. During the 1890s, he made the first radio transmissions, and, by the end of the century, he had succeeded in transmitting telegraph messages across the Atlantic Ocean—a feat which earned him the Nobel Prize for physics in 1909.
Marconi’s spark transmitters could send only coded messages, and due to the broad, long-wavelength signals used, only a few stations could broadcast at the same time. The development of the electron tube in the early years of the twentieth century, however, made it possible to transmit narrower signals on stable frequencies. This, in turn, enabled the development of technology for sending speech and music over the airwaves.
Broadcast Radio
The development of am and fm
A radio signal is simply a carrier: the process of adding information—that is, complex sounds such as those of speech or music—is called modulation. The first type of modulation developed was AM, or amplitude modulation, which Canadian-American physicist Reginald Aubrey Fessenden (1866-1932) demonstrated with the first United States radio broadcast in 1906. Amplitude modulation varies the instantaneous amplitude of the radio wave, a function of the radio station’s power, as a means of transmitting information.
By the end of World War I, radio had emerged as a popular mode of communication: for the first time in history, entire nations could hear the same sounds at the same time. During the 1930s, radio became increasingly important, both for entertainment and information. Families in the era of the Great Depression would gather around large “cathedral radios”—so named for their size and shape—to hear comedy programs, soap operas, news programs, and speeches by important public figures such as President Franklin D. Roosevelt.
Throughout this era—indeed, for more than a half-century from the end of the first World War to the height of the Vietnam Conflict in the mid-1960s—AM held a dominant position in radio. This remained the case despite a number of limitations inherent in amplitude modulation: AM broadcasts flickered with popping noises from lightning, for instance, and cars with AM radios tended to lose their signal when going under a bridge. Yet, another mode of radio transmission was developed in the 1930s, thanks to American inventor and electrical engineer Edwin H. Armstrong (1890-1954). This was FM, or frequency modulation, which varied the radio signal’s frequency rather than its amplitude.
Not only did FM offer a different type of modulation; it was on an entirely different frequency range. Whereas AM is an example of a long-wave radio transmission, FM is on the microwave sector of the electromagnetic spectrum, along with television and radar. Due to its high frequency and form of modulation, FM offered a “clean” sound as compared with AM. The addition of FM stereo broadcasts in the 1950s offered still further improvements; yet despite the advantages of FM, audiences were slow to change, and FM did not become popular until the mid- to late 1960s.
Signal Propagation
AM signals have much longer wavelengths, and smaller frequencies, than do FM signals, and this, in turn, affects the means by which AM signals are propagated. There are, of course, much longer radio wavelengths; hence, AM signals are described as intermediate in wavelength. These intermediate-wavelength signals reflect off highly charged layers in the ionosphere between 25 and 200 mi (40-332 km) above Earth’s surface. Short-wavelength signals, such as those of FM, on the other hand, follow a straight-line path. As a result, AM broadcasts extend much farther than FM, particularly at night.
At a low level in the ionosphere is the D layer, created by the Sun when it is high in the sky. The D layer absorbs medium-wavelength signals during the day, and for this reason, AM signals do not travel far during daytime hours. After the Sun goes down, however, the D layer soon fades, and this makes it possible for AM signals to reflect off a much higher layer of the ionosphere known as the F layer. (This is also sometimes known as the Heaviside layer, or the Ken-nelly-Heaviside layer, after English physicist Oliver Heaviside and British-American electrical engineer Arthur Edwin Kennelly, who independently discovered the ionosphere in 1902.) AM signals “bounce” off the F layer as though it were a mirror, making it possible for a listener at night to pick up a signal from halfway across the country.
The Sun has other effects on long-wave and intermediate-wave radio transmissions. Sunspots, or dark areas that appear on the Sun in cycles of about 11 years, can result in a heavier buildup of the ionosphere than normal, thus impeding radio-signal propagation. In addition, occasional bombardment of Earth by charged particles from the Sun can also disrupt transmissions.
Due to the high frequencies of FM signals, these do not reflect off the ionosphere; instead, they are received as direct waves. For this reason, an FM station has a fairly short broadcast range, and this varies little with regard to day or night. The limited range of FM stations as compared to AM means that there is much less interference on the FM dial than for AM.
Distribution of radio Frequencies
In the United States and most other countries, one cannot simply broadcast at will; the airwaves are regulated, and, in America, the governing authority is the Federal Communications Commission (FCC). The FCC, established in 1934, was an outgrowth of the Federal Radio Commission, founded by Congress seven years earlier. The FCC actually “sells air,” charging companies a fee to gain rights to a certain frequency. Those companies may in turn sell that air to others for a profit.
At the time of the FCC’s establishment, AM was widely used, and the federal government assigned AM stations the frequency range of 535 kHz to 1.7 MHz. Thus, if an AM station today is called, for instance, “AM 640,” this means that it operates at 640 kHz on the dial. The FCC assigned the range of 5.9 to 26.1 MHz to shortwave radio, and later the area of 26.96 to 27.41 MHz to citizens’ band (CB) radio. Above these are microwave regions assigned to television stations, as well as FM, which occupies the range from 88 to 108 MHz.
The organization of the electromagnetic spectrum’s radio frequencies—which, of course, is an entirely arbitrary, humanmade process—is fascinating. It includes assigned frequencies for everything from garage-door openers to deep-space radio communications. The FCC recognizes seven divisions of radio carriers, using a system that is not so much based on rational rules as it is on the way that the communications industries happened to develop over time.
THE SEVEN FCC DIVISIONS
Most of what has so far been described falls under the heading of “Public Fixed Radio Services”: AM and FM radio, other types of radio such as shortwave, television, various other forms of microwave broadcasting, satellite systems, and communication systems for federal departments and agencies. “Public Mobile Services” include pagers, air-to-ground service (for example, aircraft-to-tower communications), offshore service for sailing vessels, and rural radio-telephone service. “Commercial Mobile Radio Services” is the realm of cellular phones, and “Personal Communications Service” that of the newer wireless technology that began to challenge cellular for market dominance in the late 1990s.
“Private Land Mobile Radio Service” (PMR) and “Private Operational-Fixed Microwave Services” (OFS) are rather difficult to distinguish, the principal difference being that the former is used exclusively by profit-making businesses, and the latter mostly by nonprofit institutions. An example of PMR technology is the dispatching radios used by taxis, but this is only one of the more well-known forms of internal electronic communications for industry. For instance, when a film production company is shooting a picture and the director needs to speak to someone at the producer’s trailer a mile away, she may use PMR radio technology. OFS was initially designated purely for nonprofit use, and is used often by schools; but banks and other profit-making institutions often use OFS because of its low cost.
Finally, there is the realm of “Personal Radio Services,” created by the FCC in 1992. This branch, still in its infancy, will probably one day include video-on-demand, interactive polling, online shopping and banking, and other activities classified under the heading of Interactive
Video and Data Services, or IVDS. Unlike other types of video technology, these will all be wireless, and, therefore, represent a telecommunications revolution all their own.
Microwaves
Microwave Communication
Though microwaves are treated separately from radio waves, in fact, they are just radio signals of a very short wavelength. As noted earlier, FM signals are actually carried on microwaves, and, as with FM in particular, microwave signals in general are very clear and very strong, but do not extend over a great geographical area. Nor does microwave include only high-frequency radio and television; in fact, any type of information that can be transmitted via telephone wires or coaxial cables can also be sent via a microwave circuit.
Microwaves have a very narrow, focused beam: thus, the signal is amplified considerably when an antenna receives it. This phenomenon, known as “high antenna gain,” means that microwave transmitters need not be highly powerful to produce a strong signal. To further the reach of microwave broadcasts, transmitters are often placed atop mountain peaks, hilltops, or tall buildings. In the past, a microwave-transmitting network such as NBC (National Broadcasting Company) or CBS (Columbia Broadcasting System) required a network of ground-based relay stations to move its signal across the continent. The advent of satellite broadcasting in the 1960s, however, changed much about the way signals are beamed: today, networks typically replace, or at least augment, ground-based relays with satellite relays.
The first worldwide satellite TV broadcast, in the summer of 1967, featured the Beatles singing their latest song “All You Need Is Love.” Due to the international character of the broadcast, with an estimated 200 million viewers, John Lennon and Paul McCartney wrote a song with simple, universal lyrics, and the result was just another example of electronic communication uniting large populations. Indeed, the phenomenon of rock music, and of superstardom as people know it today, would be impossible without many of the forms of technology discussed here. Long before the TV broadcast, the Beatles had come to fame through the playing of their music on the radio waves—and, thus, they owed much to Maxwell, Hertz, and Marconi.
Microwave ovens
The same microwaves that transmit FM and television signals—to name only the most obviously applications of microwave for communication—can also be harnessed to cook food. The microwave oven, introduced commercially in 1955, was an outgrowth of military technology developed a decade before.
During World War II, the Raytheon Manufacturing Company had experimented with a magnetron, a device for generating extremely short-wavelength radio signals as a means of improving the efficiency of military radar. While working with a magnetron, a technician named Percy Spencer was surprised to discover that a candy bar in his pocket had melted, even though he had not felt any heat. This led him to considering the possibilities of applying the magnetron to peacetime uses, and a decade later, Raytheon’s “radar range” hit the market.
Those early microwave ovens had none of varied power settings to which modern users of the microwave—found today in two-thirds of all American homes—are accustomed. In the first microwaves, the only settings were “on” and “off,” because there were only two possible adjustments: either the magnetron would produce, or not produce, microwaves. Today, it is possible to use a microwave for almost anything that involves the heating of food that contains water—from defrosting a steak to popping popcorn.
As noted much earlier, in the general discussion of electromagnetic radiation, there are three basic types of heat transfer: conduction, convection, and radiation. Without going into too much detail here, conduction generally involves heat transfer between molecules in a solid; convection takes place in a fluid (a gas such as air or a liquid such as water); and radiation, of course, requires no medium.
A conventional oven cooks through convection, though conduction also carries heat from the outer layers of a solid (for example, a turkey) to the interior. A microwave, on the other hand, uses radiation to heat the outer layers ofthe food; then conduction, as with a conventional oven, does the rest. The difference is that the microwave heats only the food—or, more specifically, the water, which then transfers heat throughout the item being heated—and not the dish or plate. Thus, many materials, as long as they do not contain water, can be placed in a microwave oven without being melted or burned. Metal, though it contains no water, is unsafe because the microwaves bounce off the metal surfaces, creating a microwave buildup that can produce sparks and damage the oven.
In a microwave oven, microwaves emitted by a small antenna are directed into the cooking compartment, and as they enter, they pass a set of turning metal fan blades. This is the stirrer, which disperses the microwaves uniformly over the surface of the food to be heated. As a microwave strikes a water molecule, resonance causes the molecule to align with the direction of the wave. An oscillating magnetron causes the microwaves to oscillate as well, and this, in turn, compels the water molecules to do the same. Thus, the water molecules are shifting in position several million times a second, and this vibration generates energy that heats the water.
Radio Waves for Measurement and Ranging
Radar
Radio waves can be used to send communication signals, or even to cook food; they can also be used to find and measure things. One of the most obvious applications in this regard is radar, an acronym for RAdio Detection And Ranging.
Radio makes it possible for pilots to “see” through clouds, rain, fog, and all manner of natural phenomena—not least of which is darkness. It can also identify objects, both natural and manmade, thus enabling a peacetime pilot to avoid hitting another craft or the side of a mountain. On the other hand, radar may help a pilot in wartime to detect the presence of an enemy. Nor is radar used only in the skies, or for military purposes, such as guiding missiles: on the ground, it is used to detect the speeds of objects such as automobiles on an interstate highway, as well as to track storms.
In the simplest model of radar operation, the unit sends out microwaves toward the target, and the waves bounce back off the target to the unit. Though the speed of light is reduced somewhat, due to the fact that waves are traveling through air rather than through a vacuum, it is, nonetheless, possible to account for this difference. Hence, the distance to the target can be calculated using the simple formula d = vt, where d is distance, v is velocity, and t is time.
Typically, a radar system includes the following: a frequency generator and a unit for controlling the timing of signals; a transmitter and, as with broadcast radio, a modulator; a duplexer, which switches back and forth between transmission and reception mode; an antenna; a receiver, which detects and amplifies the signals bounced back to the antenna; signal and data processing units; and data display units. In a monostatic unit—one in which the transmitter and receiver are in the same location—the unit has to be continually switched between sending and receiving modes. Clearly, a bistatic unit—one in which the transmitter and receiver antennas are at different locations—is generally preferable; but on an airplane, for instance, there is no choice but to use a monostatic unit.
In order to determine the range to a target— whether that target be a mountain, an enemy aircraft, or a storm—the target itself must first be detected. This can be challenging, because only a small portion of the transmitted pulse comes back to the receiving antenna. At the same time, the antenna receives reflections from a number of other objects, and it can be difficult to determine which signal comes from the target. For an aircraft in a wartime situation, these problems are compounded by the use of enemy counter-measures such as radar “jamming.” Still another difficulty facing a military flyer is the fact that the use of radar itself—that is, the transmission of microwaves—makes the aircraft detectable to opposing forces.
Telemetry
Telemetry is the process of making measurements from a remote location and transmitting those measurements to receiving equipment. The earliest telemetry systems, developed in the United States during the 1880s, monitored the distribution and use of electricity in a given region, and relayed this information back to power companies using telephone lines. By the end of Worl d War I, electric companies used the power lines themselves as information relays, and though such electrical telemetry systems remain in use in some sectors, most modern telemetry systems apply radio signals.
An example of a modern telemetry application is the use of an input device called a transducer to measure information concerning an astronaut’s vital signs (heartbeat, blood pressure,
body temperature, and so on) during a manned space flight. The transducer takes this information and converts it into an electrical impulse, which is then beamed to the space monitoring station on Earth. Because this signal carries information, it must be modulated, but there is little danger of interference with broadcast transmissions on Earth. Typically, signals from spacecraft are sent in a range above 1010 Hz, far above the frequencies of most microwave transmissions for commercial purposes.
Light: Invisible, Visible, and Invisible Again
Between about 1013 and 1017 Hz on the electromagnetic spectrum is the range of light: infrared, visible, and ultraviolet. Light actually constitutes a small portion of the spectrum, and the area of visible light is very small indeed, extending from about 4.3 • 1014 to 7.5 • 1014 Hz. The latter, incidentally, is another example of scientific notation: not only is it easier not to use a string of zeroes, but where a coefficient or factor (for example, 4.3 or 7.5) is other than a multiple of 10, it is preferable to use what are called significant figures—usually a single digit followed by a decimal point and up to 3 decimal places.
Infrared light lies just below visible light in frequency, and this is easy to remember because of the name: red is the lowest in frequency of all the colors. Similarly, ultraviolet lies beyond the highest-frequency color, violet. Visible light itself, by far the most familiar part of the spectrum— especially prior to the age of radio communications—is discussed in detail elsewhere.
Infrared light
Though we cannot see infrared light, we feel it as heat. German-English astronomer William Herschel (17381822), first scientist to detect infrared radiation from the Sun, demonstrated its existence in 1800 by using a thermometer. Holding a prism, a three-dimensional glass shape used for diffusing beams of light, he directed a beam of sunlight toward the thermometer, which registered the heat of the infrared rays.
Eighty years later, English scientist Sir William Abney (1843-1920) developed infrared photography, a method of capturing infrared radiation, rather than visible light, on film. By the mid-twentieth century, infrared photography had come into use for a variety of purposes. Military forces, for instance, may use infrared to
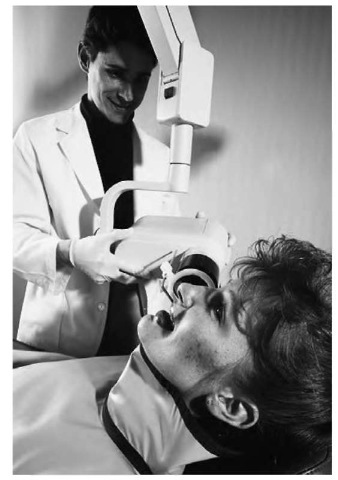
“Soft” x-ray machines, such as this one being used by a dentist to photograph the patient’s teeth, operate at relatively low frequencies and thus don’t harm the patient.
detect the presence of enemy troops. Medicine makes use of infrared photography for detecting tumors, and astronomers use infrared to detect stars too dim to be seen using ordinary visible light.
The uses of infrared imaging in astronomy, as a matter of fact, are many. The development in the 1980s of infrared arrays, two-dimensional grids which produce reliable images of infrared phenomena, revolutionized infrared astronomy. Because infrared penetrates dust much more easily than does visible light, infrared astronomy makes it easier to see regions of the universe where stars—formed from collapsing clouds of gas and dust—are in the process of developing. Because hydrogen molecules emit infrared radiation, infrared astronomy helps provide clues regarding the distribution of this highly significant chemical element throughout the universe.
Ultraviolet Light
Very little of the Sun’s ultraviolet light penetrates Earth’s atmosphere—a fortunate thing, since ultraviolet (UV) radiation can be very harmful to human skin. A suntan, as a matter of fact, is actually the skin’s defense against these harmful UV rays. Due to the fact that Earth is largely opaque, or resistant, to ultraviolet light, the most significant technological applications of UV radiation are found in outer space.
In 1978 the United States, in cooperation with several European space agencies, launched the International Ultraviolet Explorer (IUE), which measured the UV radiation from tens of thousands of stars, nebulae, and galaxies. Despite the progress made with IUE, awareness of its limitations—including a mirror of only 17 in (45 cm) on the telescope itself—led to the development of a replacement in 1992.
This was the Extreme Ultraviolet Explorer (EUVE), which could observe UV phenomena over a much higher range of wavelengths than those observed by IUE. In addition, the Hubble Space Telescope, launched by the United States in 1990, includes a UV instrument called the God-dard High Resolution Spectrograph. With a mirror measuring 8.5 ft (2.6 m), it is capable of observing objects much more faint than those detected earlier by IUE.
Ultraviolet astronomy is used to study the winds created by hot stars, as well as stars still in the process of forming, and even stars that are dying. It is also useful for analyzing the densely packed, highly active sectors near the centers of galaxies, where both energy and temperatures are extremely high.
X Rays
Though they are much higher in frequency than visible light—with wavelengths about 1,000 times shorter than for ordinary light rays—x rays are a familiar part of modern life due to their uses in medicine. German scientist Wilhelm Rontgen (1845-1923) developed the first x-ray device in 1895, and, thus, the science of using x-ray machines is called roentgenology.
The new invention became a curiosity, with carnivals offering patrons an opportunity to look at the insides of their hands. And just as many people today fear the opportunities for invasion of privacy offered by computer technology, many at the time worried that x rays would allow robbers and peeping toms to look into people’s houses. Soon, however, it became clear that the most important application of x rays lay in medicine.
KEY TERMS
Amplitude: The maximum displacement of a vibrating material. In wave motion, amplitude is the “size” of a wave, an indicator of the energy and intensity of the wave.
Cycle: One complete oscillation. In wave motion, this is equivalent to the movement of a wave from trough to crest and back to trough.
Electromagnetic force: The total force on an electrically charged particle, which is a combination of forces due to electrical and/or magnetic fields around the particle. Electromagnetic force reflects electromagnetic interaction, one of the four fundamental interactions in nature.
Electromagnetic spectrum: The complete range of electromagnetic waves on a continuous distribution from a very low range of frequencies and energy levels, with a correspondingly long wavelength, to a very high range of frequencies and energy levels, with a correspondingly short wavelength. Included on the electromagnetic spectrum are long-wave and short-wave radio; microwaves; infrared, visible, and ultraviolet light; x rays, and gamma rays.
Electromagnetic wave: A transverse wave with electrical and magnetic fields that emanate from it. The directions of these fields are perpendicular to one another, and both are perpendicular to the line of propagation for the wave itself.
Electromagnetism: The branch of physics devoted to the study of electrical and magnetic phenomena.
Field: A region of space in which it is possible to define the physical properties of each point in the region at any given moment in time.
Frequency: In wave motion, frequency is the number of waves passing through a given point during the interval of one second. The higher the frequency, the shorter the wavelength. Measured in Hertz, frequency is mathematically related to wave speed, wavelength, and period.
Fundamental interaction: The basic mode by which particles interact. There are four known fundamental interactions in nature: gravitational, electromagnetic, strong nuclear, and weak nuclear.
Harmonic motion: The repeated movement of a particle about a position of equilibrium, or balance.
Hertz: A unit for measuring frequency, named after nineteenth-century German physicist Heinrich Rudolf Hertz (1857-1894). High frequencies are expressed in terms of kilohertz (kHz; 103 or 1,000 cycles per second); megahertz (MHz; 106 or 1 million cycles per second); and gigahertz (GHz; 109 or 1 billion cycles per second.)
Intensity: Intensity is the rate at which a wave moves energy per unit of cross-sectional area.
Oscillation: A type of harmonic motion, typically periodic, in one or more dimensions.
Period: For wave motion, a period is the amount of time required to complete one full cycle. Period is mathematically related to frequency, wavelength, and wave speed.
Periodic motion: Motion that is repeated at regular intervals. These intervals are known as periods.
Periodic wave: A wave in which a uniform series of crests and troughs follow one after the other in regular succession.
PHOTON: A particle of electromagnetic radiation carrying a specific amount of energy, measured in electron volts (eV). For parts of the electromagnetic spectrum with a low frequency and long wavelength, photon energy is relatively low; but for parts with a high frequency and short wavelength, the value of photon energy is very high.
Propagation: The act or state of traveling from one place to another.
Radiation: The transfer of energy by means of electromagnetic waves, which require no physical medium (for example, water or air) for the transfer. Earth receives the Sun’s energy, via the electromagnetic spectrum, by means of radiation.
Scientific notation: A method used by scientists for writing extremely large numbers. This usually involves a coefficient, or factor, of a single digit followed by a decimal point and up to three decimal places, multiplied by 10 to a given exponent. Thus, instead of writing 75,120,000, the preferred scientific notation is 7.512 • 107. To visualize the value of very large multiples of 10, it is helpful to remember that the value of 10 raised to any power n is the same as 1 followed by that number of zeroes. Hence 1025, for instance, is simply 1 followed by 25 zeroes.
Transverse wave: A wave in which the vibration or motion is perpendicular to the direction in which the wave is moving.
Wavelength: The distance between a crest and the adjacent crest, or the trough and an adjacent trough, of a wave. Wavelength, symbolized X (the Greek letter lambda) is mathematically related to wave speed, period, and frequency.
Wave motion: A type of harmonic motion that carries energy from one place to another without actually moving any matter.
How a medical x-ray machine works
Due to their very short wavelengths, x rays can pass through substances of low density—for example, fat and other forms of soft tissue—without their movement being interrupted. But in materials of higher density, such as bone, atoms are packed closely together, and this provides x rays with less space through which to travel. As a result, x-ray images show dark areas where the rays traveled completely through the target, and light images of dense materials that blocked the movement of the rays.
Medical x-ray machines are typically referred to either as “hard” or “soft.” Soft x rays are the ones with which most people are more familiar. Operating at a relatively low frequency, these are used to photograph bones and internal organs, and provided the patient does not receive prolonged exposure to the rays, they cause little damage. Hard x rays, on the other hand, are designed precisely to cause damage—not to the patient, but to cancer cells. Because they use high voltage and high-frequency rays, hard x rays can be quite dangerous to the patient as well.
Other applications
X-ray crystallography, developed in the early twentieth century, is devoted to the study of the interference patterns produced by x rays passing through materials that are crystalline in Structure. Each of these discoveries, in turn, transformed daily life: insulin, by offering hope to diabetics, penicillin, by providing a treatment for a number of previously fatal illnesses, and DNA, by enabling scientists to make complex assessments of genetic information.
In addition to the medical applications, the scanning capabilities of x-ray machines make them useful for security. A healthy person receives an x ray at a doctor’s office only once in a while; but everyone who carries items past a certain point in a major airport must submit to x-ray security scanning. If one is carrying a purse or briefcase, for instance, this is placed on a moving belt and subjected to scanning by a low-power device that can reveal the contents.
Gamma Rays
At the furthest known reaches of the electromagnetic spectrum are gamma rays, ultra high-frequency, high-energy, and short- wavelength forms of radiation. Human understanding of gamma rays, including the awesome powers they contain, is still in its infancy.
In 1979, a wave of enormous energy passed over the Solar System. Though its effects on Earth were negligible, instruments aboard several satellites provided data concerning an enormous quantity of radiation caused by gamma rays. As to the source of the rays themselves, believed to be a product of nuclear fusion on some other body in the universe, scientists knew nothing.
The Compton Gamma Ray Observatory Satellite, launched by NASA (National Aeronautics and Space Administration) in 1991, detected a number of gamma-ray bursts over the next two years. The energy in these bursts was staggering: just one of these, scientists calculated, contained more than a thousand times as much energy as the Sun will generate in its entire lifetime of 10 billion years.
Some astronomers speculate that the source of these gamma-ray bursts may ultimately be a distant supernova, or exploding star. If this is the case, scientists may have found the supernova; but do not expect to see it in the night sky. It is not known just how long ago it exploded, but its light appeared on Earth some 340,000 years ago, and during that time it was visible in daylight for more than two years. So great was its power that the effects of this stellar phenomenon are still being experienced.
