Roots and methods of systematics and classification
There is only one figure in the 1859 first edition of Charles Darwin’s On the Origin of Species; it is what biologists now call a phylogenetic tree. A phylogenetic tree is a diagram that shows how animals have evolved from a common ancestor by branching out from it. Darwin himself did not use the term “phylogeny,” but he referred to his tree as a “diagram of divergence of taxa.” Darwin wrote primarily about evolution in Origin, but he devoted parts of Chapter XIII to classification, in which he gave a clear account of what he considered a natural system for classifying organisms: “I hold the natural system is genealogical in its arrangement, like a pedigree; but the degrees of modification which the different groups have undergone, have to be expressed by ranking them under different so-called genera, sub-families, families, sections, orders, and classes.” The science of systematics, which includes taxonomy, is the oldest and most encompassing of all fields of biology, and in 1859 natural history was largely a matter of classifying. Darwin’s statement may not sound particularly revolutionary to modern adherents of the theory of evolution; however, biological classification before the mid-nineteenth century had essentially been a matter of imposing some kind of order on a complex nature created by God. (Obviously, classifications at that time did not reflect any underlying process simply because the process of evolution was unknown then.) Darwin’s concept quickly won acceptance among biologists, and phylogenetic trees became the standard way to depict the evolution of recent taxa and how taxa have originated from a common ancestor.
If scientists wish to classify living animals to reflect their evolutionary relationships, they must first investigate the phy-logeny of the organisms in question. Darwin did not devise a method for determining phylogenetic relationships other than in very general terms, however. Although phylogenies began to appear in the late nineteenth century, they were based on subjective assessments of the morphological similarities and differences that were then regarded as indications of kinship. Even though many authors have used phylogenetic terms in discussing their systems of classification, one must bear in mind that many of the classifications found in text topics are not based on any explicit phylogenetic analysis. In fact, it was not until the mid-twentieth century that theoretical as well as methodological advances in the field of systematics led biologists to better supported phylogenetic hypotheses. As of 2003, most systematists and evolutionary biologists use these methods, which are known as phylogenetic systematics or cladis-tics, to infer relationships among various animals and present the results in the form of a cladogram or phylogenetic tree.
The basic concept in phylogenetic systematics is mono-phyly. A monophyletic group of species is one that includes the ancestral species and all of its descendants. Thus, a mono-phyletic taxon is a group of species whose members are related to one another through a shared history of descent; that is, a single evolutionary lineage. There are several taxa and names still in use that are not monophyletic; some have survived because they are still in common use—for example, “invertebrates”—and others because we know little about their evolutionary history. The basis for determining evolutionary relationships is homology, a term that refers to similarities resulting from shared ancestry. The cladistic term for this similarity is synapomorphy. It is these homologous characters that point to a common ancestry. For example, the presence of a backbone in birds, lizards and humans indicates that these three groups share a common ancestor and are thus related. Similarity, however, does not always reflect common ancestry; sometimes it points to convergent evolution. The advanced octopus eye, which in many ways resembles the human eye, is not an indication of a relationship between octopods and humans. Cladistic methods are used to distinguish between similarities resulting from a common ancestry and similarities due to other causes.
In the past, biologists used morphological characters as the primary source for investigating relationships. Most of the current taxonomic classification is based on assessment of morphological similarities and differences. Morphological characters alone, however, have obvious limitations in determining phylogeny within the animal kingdom. It is difficult to find similarities between, say, a flatworm and a sponge if the researcher must rely on gross morphology and anatomy. Some taxonomists therefore turned to embryological characteristics; many relationships among animals have been established on the basis of sperm morphology or larval biology. The advent of polymerase chain reaction (PCR) technology and direct nucleotide sequencing has brought about immense changes in the amount of information available for phylogeny evaluation. The finding that animals at all levels share large portions of the genome makes it possible to compare taxa as far apart as vertebrates and nematodes at homologous gene loci. Biologists can feel confident that they are comparing the “same” thing when they look at the base composition of a gene such as 18s rDNA, because there are so many overall genetic similarities in this gene between taxa. A staggering amount of new information has been collected from DNA; at present, all new gene sequences are deposited in banks like GenBank, which makes them available to the worldwide scientific community. Researchers are thus getting closer to finding the actual Tree of Life; electronic databases and information sharing have led them much closer to realizing Darwin’s vision of a classification based on genealogy. On the other hand, these recent advances mean that many traditional views of relationships among various groups of animals are open to question. It is not easy to write about metazoan phy-logeny today, knowing that so many established “truths” have already been overturned, and more will certainly be challenged in the near future.
As of 2003, the classification of animals is defined by the International Code of Zoological Nomenclature, established on January 1, 1758—the year of publication of the tenth edition of Linnaeus’s Systema Naturae. This code regulates the naming of species, genera, and families. It states that a species name should refer to a holotype, which is a designated specimen deposited in a museum or similar institution. In theory, the holotype should be available to anyone who wishes to study it. The genus is defined by the type (typical) species, and the family name is defined by the type genus. Although the convention has developed of using a series of hierarchical categories, these other ranks are not covered by the code and are not defined in the same explicit way. The most inclusive category is kingdom, followed by phylum. Although phyla are not formally defined in the Code, and authors disagree about their definition in some instances, “phylum” is probably the category most easily recognized by nonspecialists. Members of a phylum have a similar bauplan (the German word for an architect’s ground plan for a building) or body organization, and share some obvious synapomorphies, or specialized characters that originated in the last common ancestor. These similarities and shared characters are not always obvious, however; the rank of phylum for some taxa is open to debate. Molecular data have also challenged the monophyletic status of some phyla that were previously unquestioned. It is evident that the cladistic approach to systematics, combined with an ever increasing amount of data from molecular genetics, has ushered in a period of taxonomic turmoil.
Kingdoms of life
The world of living organisms can be divided into two major groups, the prokaryotes and the eukaryotes. The prokary-otes lack membrane-enclosed organelles and a nucleus, while the eukaryotes do possess organelles and a nucleus inside their membranes, and have linear chromosomes. (By 2003, however, an organelle was found in a bacterium—which overturns the assumption that these specialized compartments are unique to the eukaryotes.) The prokaryotes have been subdivided into two kingdoms: Eubacteria (bacteria) and Archea (archaebacteria). The eukaryotic kingdom has been subdivided into the Animalia or Metazoa and the “animal-like organisms” or Protozoa. As of 2003, however, the protozoans are most often referred to as the Kingdom Protista; that is, eukaryotic single-celled microorganisms together with certain algae. This kingdom contains around 18 phyla that include amoebas, dinoflagellates, foraminiferans and ciliates. Kingdom Animalia contains about 34 phyla of heterotrophic mul-ticellular organisms. The number is approximate because there is currently no consensus regarding the detailed classification of taxa into phyla. About 1.3 million living species have been described, but this number is undoubtedly an underestimate. Estimates of undescribed species range from lows of 10-30 million to highs of 100-200 million.
The beginnings of life
Clearly the prokaryotes are the most ancient living organisms, but when did they first appear? There is indirect evidence of prokaryotic organisms in some of the oldest sediments on earth, suggesting that life first appeared in the seas as soon as the planet cooled enough for life as we know it today to exist. There are three popular theories regarding the origin of life on earth. The classic theory, which dates from the 1950s, suggests that self-replicating organic molecules first appeared in the atmosphere and were deposited in the seas by rain. In the seas, these molecules underwent further reactions in the presence of energy from lightning strikes to make nucleic acids, proteins, and the other building blocks of life. More recently, the second theory has proposed that the first synthesis of organic molecules took place near deep-sea hydrothermal vents that had the necessary heat energy and chemical activity to form these molecules. The third theory maintains that organic molecules came to Earth from another planet.
The data suggest that the first eukaryotic cells appeared several billion years ago, but we know very few details about the early evolution of these eukaryotes. Although they appeared early, they probably took a few hundred million more years to develop into multicellular organisms. Eukaryotic cells are appreciably larger than prokaryotic cells and have a much higher degree of organization. Each cell has a membrane-bound nucleus with chromosomes, and a cytoplasm containing various specialized organelles that carry out different functions, including reproduction. An example of an organelle is the mitochondrion. Mitochondria serve as the sites of cell respiration and energy generation. The presence of these or-ganelles, and their similarity to the structures and functions of free-living bacteria, suggest that bacteria were incorporated into the precursors of eukaryotic cells and lost their autonomy in the process. This scenario is referred to as the theory of endosymbiosis; in essence, it defines the eukaryotic cell as a community of microorganisms. The first endosymbionts are believed to have been ancestral bacteria incorporating other bacteria that could respire aerobically. These bacteria subsequently became the mitochondria. The accumulation of free oxygen in the oceans from photosynthesis may have triggered the evolution of eukaryotes; this hypothesis is supported by the coincidental timing of the first eukaryotic cells and a rise
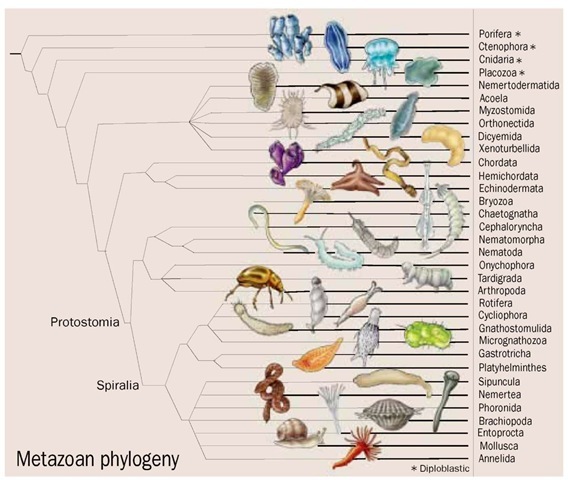
Phylogenetic tree of lower metazoans and protostomes.
in the levels of free oxygen in the oceans. On the basis of fossil findings, scientists think that the forming of these early complex cells took place rather quickly, probably between 2800 and 2100 million years ago (mya), even though the oldest known eukaryote (Grypania, a coiled unbranched filament up to 1.18 in [30 mm] long) comes from rocks that are more than 2100 million years old. The earliest eukaryotic cells known to belong to any modern taxon are red algae, thought to be about 1000 million years old.
Data from the molecular clock suggest that the last common ancestor of plants and animals existed about 1.6 billion years ago, which is long after the first appearance of eukary-otes and long before any definite fossil records of metazoans. The Ediacaran fauna (600-570 mya) contains the first evidence of the existence of many modern phyla. This evidence, however, is largely a matter of trace fossils, which result from animals moving through sediment. The relation of these trace fossils to modern phyla is therefore a matter of debate. As of 2003, biologists tend to regard the entire fauna as including many species now viewed as primitive members of extant phyla. The modern phyla thought to be represented among the Ediacaran fauna include annelid-like forms, Porifera, Cnidaria, Echiura, Onychophora, and Mollusca. There are many fossils from this period that cannot be assigned with certainty to any recent phyla; these forms probably represent high-level taxa that later became extinct. Although most of the Ediacaran organisms were preserved as shallow-water impressions in sandstone, there are around 30 sites worldwide representing deepwater and continental slope communities.

An illustration depicting what the ocean may have looked like during the Jurassic Period; present are an ammonite (Titanites anguiformis), based on fossils from Portland, Dorset, England, and ichthyosaurs (Stenopterygius sp.), based on fossils from Holtzmanaden, Germany.
The Ediacaran fauna was almost entirely soft-bodied, although it also included some animals that palaeontologists place among the mollusks and early arthropod-like organisms. They draw this conclusion from the fossilized remains of chitinous structures thought to be the jaws of annelid-like animals and the radulae (rows of teeth functioning as scrapers) of mollusks. Many of the animals from this period appear to have lacked complex internal structures, but by the late Edi-acara period larger animals appeared that probably had internal organs, considering their size. For example, the segmented sheet-like Dickinsonia, which was probably a poly-chaete, grew as long as 39.3 in (1 m). It is unlikely that an animal of that size could survive without internal structures that digested and metabolized food. It is clear from these fossils that large and complicated metazoan animals already existed 540 mya.
The amount of fossil evidence for bilaterally symmetrical metazoans increased exponentially during the transition between the Precambrian and Cambrian Periods, about 544 mya. This transition is called the Cambrian explosion. The question is whether this sudden appearance of a number of phyla is evidence for a rapid radiation (diversification) of animal forms. Some researchers have suggested that the absence or lack of metazoan life in the early fossils is due to the simple fact that the first animals were small organisms lacking structures (like shells) that fossilized well. Some findings support the view that the first animals were microscopic; however, as has already been mentioned, there were also large animals in this period. Although there are problems with using the molecular clock method of measurement (calibrating the nodes in a phylogenetic tree based on assumptions about the rate of mutations in a molecule), metazoan phylogenies based on molecular data indicate that many recent phyla existed before the Cambrian explosion but did not leave fossil evidence until later. Some researchers have proposed that a more complicated life style, more complex interactions among animals, and especially the advent of predation were a strong selective force for developing such features as shells as an-tipredation devices. While the Ediacaran fauna seems to have consisted of suspension and detritus feeders who were largely passive as well as a very few active predators, animal communities during the Early Cambrian Period included most of the trophic levels found in modern marine communities. According to some authors, it is this second set of interactions that led to structures that could be fossilized. There have also been explanations based on such abiotic factors as atmospheric or geochemical changes. In either case, it is possible that although abundant fossils from the major animal phyla are found in Cambrian strata, the organisms originated in an earlier period. In other words, the so-called “Cambrian explosion” may simply reflect the difficulty of preserving soft-bodied or microscopic animals. Many paleontologists hold that these phyla originated instead during the Neopro-terozoic Period during the 160 million years preceding the Cambrian explosion. Their opinion is based on findings from Ediacaran fauna.
Dating the origins of the metazoan phyla is thus controversial. The resolution of this controversy has been sought in DNA sequence data and the concept of a molecular clock. The molecular clock hypothesis assumes that the evolutionary rates for a particular gene are constant through time and across taxa, or that we can compensate for disparate rates at different times. The results from such studies differ; however, one relatively recent result indicates that protostomes and deuterostomes diverged around 544-700 mya, and that the divergence between echinoderms and chordates took place just before the Cambrian period. This study shows that mol-lusks, annelids, and arthropods had existed for over 100 million years before the Cambrian explosion, and echinoderms and early chordates may have arisen 50 million years prior to this explosion.
Protists
Although the term “protozoa” has been used for a long time, and ranked as a phylum for a hundred years, it is now clear that the name does not define a monophyletic group. “Protozoa” is really a name attached to a loose assemblage of primarily single-celled heterotrophic eukaryotic organisms. The Kingdom Protista contains both organisms traditionally called protozoa as well as some autotrophic groups. (The distinction between heterotrophy and autotrophy is, however, blurred in these organisms.) There are no unique features, or synapomorphies, that distinguish this kingdom from others; protists can be defined only as a grouping of eukaryotes that lack the organization of cells into tissues and organs that is seen in animals (or in fungi and plants for that matter). Current understandings of protist phylogeny and classification are in a state of constant flux. Recent molecular studies have overturned so many established classification schemes that any attempt to describe taxa within this kingdom as of 2003 risks becoming obsolete in a matter of months. In any event, however, the protists are the first eukaryotic organisms, and the forerunners of the multicellular animals known as metazoans.
One example of protists is phylum Ciliophora, the ciliates, which are very common in benthic (sea bottom) and plank-

Dorsal reconstruction of a crustacean (Waptia fieldensis) from the Middle Cambrian Burgess Shale of British Columbia. This small, fairly common, shrimp-like arthropod had gill branches for swimming, tail flaps for steering, and legs for walking on the seafloor. It averaged about 3 in. (7.5 cm) in length.
tonic communities in marine, brackish, and freshwater habitats as well as in damp soils. Several ciliates are important mu-tualistic endosymbionts of such ruminants as goats and sheep, in whose digestive tracts they convert plant material into a form that can be absorbed by the animal. Other examples include the euglenids and their kin. They are now placed in the phylum Kinetoplastida, but used to be part of what was called the phylum Sarcomastigophora, which, at that time, also contained the dinoflagellates (now placed in their own phylum Di-noflagellata). The phylum Kinetoplastida includes two major subgroups. The trypanosomes are the better known of the two, since several species in this group cause debilitating and often fatal diseases in humans. Species of Leishmania cause a variety of ailments collectively known as leishmaniasis, transmitted by the bite of sand flies. Leishmaniasis kills about a thousand people each year and infects over a million worldwide. More serious diseases are caused by members of the genus Trypanosoma which live as parasites in all classes of vertebrates. Chagas’ disease, for example, is caused by a Trypanosoma species transmitted to humans by a group of hemipterans (insects with sucking mouth parts) known as assassin or kissing bugs. These insects feed on blood and often bite sleeping humans (commonly around the mouth, whence the nickname). They leave behind fecal matter that contains the infective stages of the trypanosome, which invades the body through mucous membranes or the insect’s bite wound.
Other protists that cause serious diseases in humans belong to the phylum Apicomplexa. Members of the genus Plasmodium cause malaria, which affects millions of people in over a hundred countries. Malaria has been known since antiquity; the relationship between the disease and swampy land led to the belief that it was contracted by breathing “bad air” (mal aria in Italian). Nearly 500 million people around the world

A trilobite fossil (Ceraurus pleurexanthemus) from the Ordovician Period, found in Quebec, Canada.
are stricken annually with malaria; of these, 1-3 million die from the disease, half of them children. The most deadly species of this genus, Plasmodium falciparum, causes massive destruction of red blood cells, which results in high levels of free hemoglobin and various breakdown products circulating in the patient’s blood and urine. These broken-down cellular fragments lead to a darkening of the urine and a condition known as blackwater fever.
There are around 4000 described species of dinoflagellates, now assigned to their own phylum, Dinoflagellata. Many of these species are known only as fossils. There are fossils that unquestionably belong to this group dating back 240 million years; in addition, evidence from Early Cambrian rocks indicates that they were abundant as early as 540 mya. Some planktonic dinoflagellates occasionally undergo periodic bursts of population growth responsible for a phenomenon known as red tide. During a red tide, the density of these di-noflagellates may be as high as 10-100 million cells per 1.05 quarts (1 liter) of seawater. Many of the organisms that cause red tides produce toxic substances that can be transmitted to humans through shellfish. Another well-known group of pro-tists are the amoebas (phylum Rhizopoda), a small phylum of around 200 described species. The most obvious feature of rhizopodans is that they form temporary extensions of their cytoplasm known as pseudopodia, which are used in feeding and locomotion.
Earliest metazoans
The origin of the metazoan phyla is a matter of debate; several theories are presently proposed. The syncytial theory suggests that the metazoan ancestor was a multinucleate, bilaterally symmetrical, ciliated protist that began to live on sea bottoms. A syncytium is a mass of cytoplasm that contains several or many nuclei but is not divided into separate cells. The principal argument in support of the syncytial theory is the presence of certain similarities between modern ciliates and acoel flatworms. Most of the objections to this hypothesis concern developmental matters and differences in general levels of complexity among the adult animals. Another proposal known as the colonial theory suggests that a colonial flagellated protist gave rise to a planuloid (free-swimming larva) metazoan ancestor. The ancestral protist, according to this theory, was a hollow sphere of flagellated cells that developed some degree of anterior-posterior orientation related to its patterns of motion, and also had cells that were specialized for separate somatic and reproductive functions. This theory has been modified over the years by various authors; most evidence as of 2003 points to the protist phylum Choanoflagellata as the most likely ancestor of the Metazoa. Choanoflagellates possess collar cells that are basically identical to those found in sponges. There are a number of choanoflagellate genera commonly cited as typifying a potential metazoan precursor, for example Proterospongia and Sphaeroeca.
The differences between the two theories may be summarized as follows. There is a ciliate ancestor in the syncytial theory; this ancestor gave rise to one lineage leading to “other protists” and the Porifera (sponges), and another lineage leading to flatworms, cnidarians, ctenophorans, flatworms, and “higher metazoans.” The colonial theory posits three separate lineages: one leading to other protists, a second to Porifera, and a third to the rest of the metazoans. Both theories place Porifera at the base of the phylogenetic tree, probably because sponges are among the simplest of living multicellular organisms. They are sedentary filter feeders with flagellated cells that pump water through their canal system. Sponges are aggregates, or collections, of partially differentiated cells that show some rudimentary interdependence and are loosely arranged in layers. These organisms essentially remain at a cellular grade of organization. Porifera is the only phylum representing the parazoan type of body structure, which means that the sponges are metazoans without true em-bryological germ layering. Not only are true tissues absent in sponges, most of their body cells are capable of changing form and function.
Diploblastic metazoans
The next step in the direction of more complex metazoans was the evolution of the diploblastic phyla, the cnidarians and the comb jellies. “Diploblastic” refers to the presence of two germ layers in the embryonic forms of these animals. Both Cnidaria and Ctenophora are characterized by primary radial symmetry and two body layers, the ectoderm and the endo-derm. One should note that some authors argue for the presence of a third germ layer in the ctenophores that is embryologically equivalent with the other two. Phylum Cnidaria includes jellyfish, sea anemones and corals, together with other less known groups. Cnidarians lack cephalization, which means that they do not reflect the evolutionary tendency to locate important body organs in or near the head.
In addition, cnidarians do not have a centralized nervous system or discrete (separate) respiratory, circulatory, and excretory organs. The primitive nature of the cnidarian bauplan is shown by the fact that they have very few different types of cells, in fact fewer than a single organ in most other meta-zoans. The essence of the cnidarian bauplan is radial symmetry, a pattern that resembles the spokes of a wheel, and places limits on the possible modes of life for a cnidarian. Cnidarians may be sessile, sedentary, or pelagic, but they do not move in a clear direction in the manner of bilateral cephal-ized creatures. The cnidarians, however, have one of the longest metazoan fossil histories. The first documented cnidarian fossil is from the Ediacaran fauna, which contains several kinds of medusae and sea pens that lived nearly 600 mya. There are two major competing theories about the ancestral cnidaria, focusing on whether the first cnidarian was polyploid or medusoid in form. According to one theory, modern Hydrozoa lie at the base of the cnidarian phylogeny (planuloid theory); other theories are inconclusive as to whether the modern Anthozoa or Hydrozoa were the first cnidarians.
Ctenophores, commonly called comb jellies, sea gooseberries, or sea walnuts, are transparent gelatinous animals. Like the cnidarians, the ctenophores are radially symmetrical diploblastic animals that resemble cnidarians in many respects. They differ significantly from cnidarians, however, in having a more organized digestive system, mesenchymal musculature, and eight rows of ciliary plates at some stage in their life history, as well as in some other features. Although the ctenophores and cnidarians are similar in their general construction, it is difficult to derive ctenophores from any existing cnidarian group; consequently, the phylogenetic position of ctenophores is an open question. Traditional accounts of ctenophores describe them as close to cnidarians but separating from them at a later point in evolutionary history. Ctenophores, however, are really quite different from cnidar-ians in many fundamental ways; many of the apparent similarities between the two groups may well reflect convergent adaptations to similar lifestyles rather than a phylogenetic relationship. Ctenophores have a pair of anal pores that have sometimes been interpreted as homologous with the anus of bilaterian animals (worms, humans, snails, fish, etc.). Furthermore, a third tissue layer between the endoderm and ectoderm may be a characteristic reminiscent of the Bilateria. These findings would support the phylogenetic position of ctenophores in comparison to that of the cnidarians, but recent molecular studies in fact point to a plesiomorphic position. “Plesiomorphic” refers to primitive or generalized characteristics that arose at an early stage in the evolution of a taxon. As a result of these studies, the relationship between cnidarians and ctenophores is still unsettled and is an active area of research.
Bilateral symmetry, triploblastic metazoans and the protostomes
Although there are currently several different views regarding the details of metazoan phylogeny, all analyses make a clear differentiation between the lower and diploblastic animals on the one hand and the triploblastic animals on the other. Some time after the radiate phyla evolved, animals with bilateral symmetry (a body axis with a clear front end and back end) and a third germ layer (the mesoderm) appeared. The appearance of bilateral symmetry was associated with the beginning of cephalization as the nervous system was concentrated in the head, and was accompanied by the development of longitudinal nerve cords. There are two fundamentally different patterns of mesoderm development, which are mirrored in the two major lineages of the Bilateria, the Deuterostomia and the Protostomia. This section will discuss the evolution of the protostomes. It must be emphasized, however, that metazoan phylogeny is undergoing continual revision. The molecular data are inconclusive not only regarding the relationships of some metazoan taxa to one another, but also which phyla belong to the two major clades. The following discussion of protostome evolution is derived from the most recent analyses based on combinations of DNA and morphological data.
Most authors assign about 20 phyla to the Protostomia; however, recent molecular data and cladistic analyses based on extended sets of morphological data do not agree as to whether the brachiopods and Phoronida should be included among the deuterostomes. These analyses are also incongru-ent when it comes to the position of several phyla; in addition, they call into question the monophyly of traditionally well-recognized taxa. Still, most authors have so far identified the flatworms (Platyhelminthes) as the first phylum to emerge among the protostomes. Platyhelminths are simple, wormlike animals lacking any apomorphies that distinguish them from the hypothetical protostome ancestor. An apomorphy is a new evolutionary trait that is unique to a species and all its descendants. The absence of apomorphies among the platy-helminths, then, means that there is no unique feature, such as the rhyncocoel found in ribbon worms, that can be used to identify the group as monophyletic. Various hypotheses regarding the origin of flatworms, their relationship to other taxa, and evolutionary patterns within the group have been hotly debated over the years. Recent DNA data even suggests that the phylum Platyhelminthes is not monophyletic and that one of the orders (Acoela) should be placed in a separate phylum. These analyses furthermore place the flatworms in a more apomorphic position on the tree, indicating that what were regarded as primitive and ancestral conditions are really either secondary losses, or that the ancestor was quite different from present notions of it.
Morphological characters still suggest that the first pro-tostomes were vermiform (worm-shaped) animals like the ribbon worms in phylum Nemertea. Other taxa with wormlike ancestral features include the sipunculans (phylum Sipuncula) and the echiurans (phylum Echiura), which are animals that burrow into sediments on the ocean floor by using the large trunk coelom for peristalsis. The other major protostome taxa escaped from infaunal life, perhaps in part through the evolution of exoskeletons or the ability to build a tube. The emergence of the mollusks may be an instance of this transition. The most primitive mollusks are probably the vermiform aplacophorans, or worm mollusks. It seems likely that these animals arose from an early wormlike protostome, indicating that the sipunculans are related to the mollusks. This hypothesis is also supported by morphological data and 18S rDNA sequences. Except for the aplacophorans, all other mollusks have solid calcareous shells produced by glands in their mantles. These shells, which provide structural support and serve as defense mechanisms, vary greatly in size and shape.
The most diverse group of animals with exoskeletons is phylum Arthropoda, which includes over one million described species, most of them in the two classes Crustacea and Insecta. The first arthropods probably arose in Precam-brian seas over 600 mya, and the true crustaceans were already well established by the early Cambrian period. The arthropods have undergone a tremendous evolutionary radiation and are now found in virtually all environments on the planet. The arthropods constitute 85% of all described animal species; this figure, however, is a gross underestimate of the actual number of arthropod species. Estimates of the true number range from 3-100 million species. The arthropods resemble the annelids in being segmented animals; in contrast to the annelids, however, they have hard exoskeletons. This feature provides several evolutionary advantages but clearly poses some problems as well. Being encased in an ex-oskeleton of this kind puts obvious limits on an organism’s growth and locomotion. The fundamental problem of movement was solved by the evolution of joints in the body and appendages, and sets of highly regionalized muscles. The intricate problem of growth within a constraining exoskeleton was solved through the complex process of ecdysis, a specific hormone-mediated form of molting. In this process, the ex-oskeleton is periodically shed to allow for increases in body size. It may be that ecdysis is unique to Arthropoda, Tardigrada and Onychophora and not homologous to the cu-ticular shedding that occurs in several metazoan phyla. This question is unresolved as of 2003. In the phylogeny included here, the arthropods are assigned to the same clade (a group of organisms sharing a specific common ancestor) as other cuticular-shedding taxa. Some authors refer to this clade as Ecdysozoa, a classification that is receiving increasing support from various sources of information.
The development of an exoskeleton clearly conferred a great selective advantage, as evidenced by the spectacular success of arthropods with regard to both diversity and abundance. This success took place despite the need for several coincidental changes to overcome the limitations of the exoskeleton. One of the key advantages of an exoskeleton is protection—against predation and physical injury, to be sure, but also against physiological stress. The morphological diversity among arthropods has resulted largely from the differential specialization of various segments, regions, and appendages in their bodies. It is clear that segmentation, in which body structures with the same genetic and developmental origins arise repeatedly during the ontogeny of an organism, is advantageous in general and leads to evolutionary plasticity. The segmented worms of phylum Annelida exemplify this evolutionary plasticity. This taxon comprises around 16,500 species; annelids have successfully invaded virtually all habitats that have sufficient water. Annelids are found most commonly in the sea, but are also abundant in fresh water. In addition, many annelid species live comfortably in damp terrestrial environments.
Segmentation as expressed in the arthropods and annelids has traditionally been considered a character that indicates a close relationships between these two taxa, and they are often placed in the same clade. Several recent analyses dispute this picture, however; the phylogeny included here assigns Annelida and Arthropoda to two different clades. It also places the flatworms in a much more apomorphic position and not in their customary location at the base of the phylogenetic tree. Systematics and classification are unsettled as of 2003, and several “truths” about evolutionary relationships are likely to be overturned in the near future. These new phylogenies will also lead to the revision of theories regarding the evolution of behavior and characters.
