ATP synthase, also called F0F1 ATPase, or simply F-ATPase, is the universal protein that terminates oxidative phosphorylation by synthesizing ATP from ADP and phosphate. Nearly identical proteins are found in eukaryotic mitochondria and bacteria, and they all operate on the same principle. Electron-driven ion pumps set up concentration and electrical gradients across a membrane (see Chemiosmotic Coupling and Proton Motive Force). ATP synthase utilizes the energy stored in this electrochemical gradient to drive nucleotide synthesis. It does this in a surprising way by converting the electromotive force into a rotary torque that promotes phosphate binding and liberates ATP from the catalytic site where it was formed. Remarkably, this process can be reversed in certain circumstances. ATP hydrolysis can drive the engine in reverse, so that F-ATPase functions as a proton pump. Indeed, the vacuolar V-ATPases, the most ubiquitous intracellular proton pumps, are structurally similar to ATP synthase and operate according to the same principles.
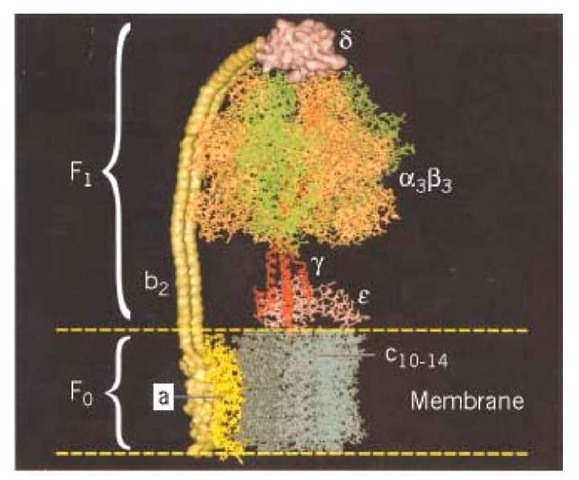
ATP synthase is composed of at least eight subunit types, whose stoichiometries are denoted by the subscripts![]() that combine into two distinct regions. The geometric arrangement of the subunits is shown schematically in Fig. 1(a). The soluble F1 portion consists of a hexamer, a3b3. This hexamer is arranged in an annulus about a central shaft consisting of the coiled- coil g subunit. Subunits d and e are also generally isolated with F |. The FQ portion consists of three transmembrane subunits, a, b2, and c^. The 12 copies of the c-subunit form a disk into which the g and r subunits insert. The remainder of F() consists of the transmembrane subunits a and b2. The latter is attached by the d subunit to an a subunit, so that it anchors the a subunit to F^ Thus there are two "stalks" connecting
that combine into two distinct regions. The geometric arrangement of the subunits is shown schematically in Fig. 1(a). The soluble F1 portion consists of a hexamer, a3b3. This hexamer is arranged in an annulus about a central shaft consisting of the coiled- coil g subunit. Subunits d and e are also generally isolated with F |. The FQ portion consists of three transmembrane subunits, a, b2, and c^. The 12 copies of the c-subunit form a disk into which the g and r subunits insert. The remainder of F() consists of the transmembrane subunits a and b2. The latter is attached by the d subunit to an a subunit, so that it anchors the a subunit to F^ Thus there are two "stalks" connecting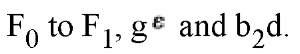
Figure 1. Schematic diagram of the subunit organization of ATP synthase (1). (a) The a3b3 hexamer and a portion of the g shaft. The lower part of g has not been resolved. The c-subunit consists of 12 pairs of transmembrane a-helices, and the a subunit consists of six transmembrane a-helices. The c subunit abuts c and g and interacts with the DELSEED region of b. The a subunit is attached to an a subunit via the b and d subunits. (b) The proton motive force at the a-c interface leads to the functional subdivision into two counterrotating assemblies, usually denoted as the "rotor" and "stator." The rotor consists of subunits![]() and the stator consists of subunits
and the stator consists of subunits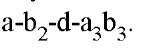
The key to understanding how ATP synthase carries out its catalytic and synthetic roles lies in this geometric organization. The entire protein can be divided into two operational regions denoted suggestively as the "rotor" and the "stator" for reasons that derive from the rotary mechanism by which the protein operates (Fig. 1b). Indeed, it turns out that ATP synthase has two rotary engines in one. The F0 motor converts transmembrane electrochemical energy into a rotary torque on the g shaft, and F1 uses ATP hydrolysis to turn the g shaft in the opposite direction. Because they are connected, one drives the other in reverse. When F0 dominates, the rotor turns clockwise (looking upward in Fig. 1), so that F1 synthesizes ATP. When F1 dominates, so that F0 is driven counterclockwise, it can pump protons against an electrochemical gradient. Deciphering how this remarkable dual energy transduction works is one of the great triumphs of modern chemistry.
1. ATP Synthesis in F1 is Driven by Rotation of the g-Shaft
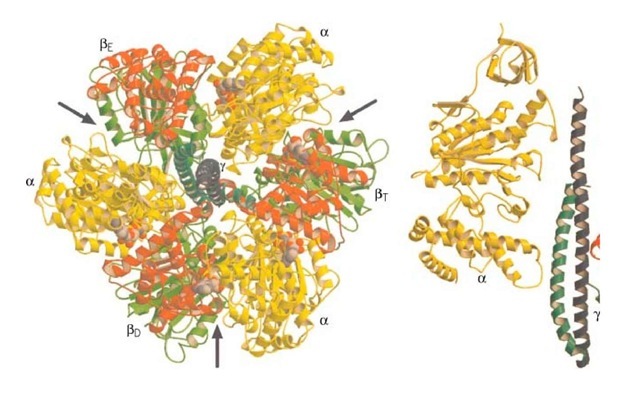
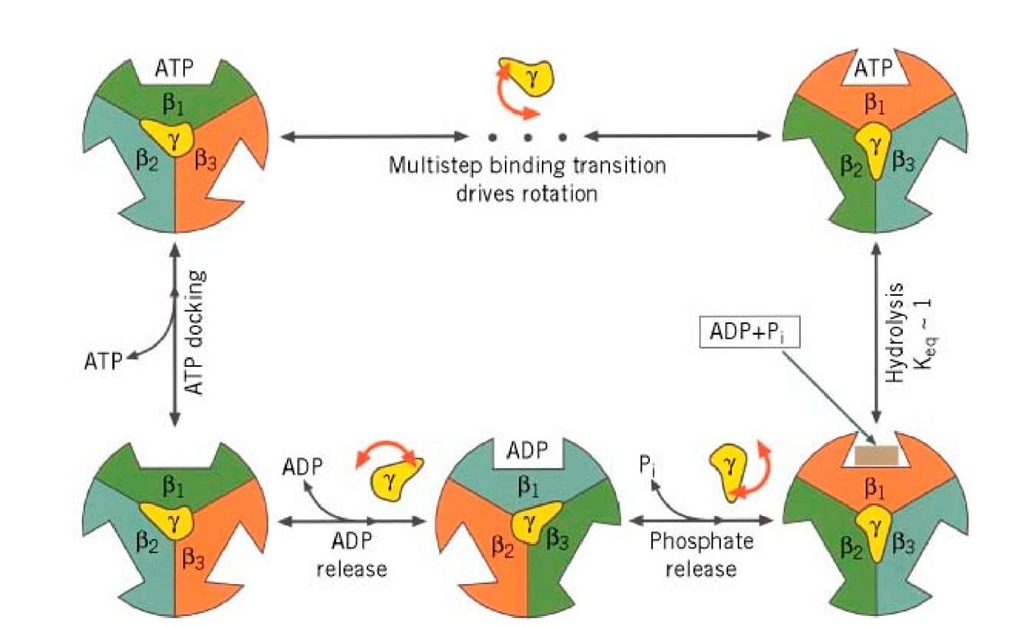
We begin with the F1 motor, because we now know precisely what it looks like. This is due to John Walker and the X-ray crystallography group at Cambridge, who worked out the exact structure of the a3b3 hexamer and most of the g shaft (1). A stereo view of the structure is shown in Fig. 2. Walker was awarded the Nobel Prize in 1997 because his structure revealed essential asymmetries in the molecule’s structure that were the key to understanding how it worked. In the early 1980′s, Paul Boyer at UCLA proposed the surprising theory that, in the catalytic sites of F1, ATP is in chemical equilibrium with its reactants, ADP and phosphate (2). So the formation of ATP is essentially without cost energetically. However, because each ATP hydrolyzed under cellular conditions liberates about 12 kcal/mol, this energetic price must be paid at some point. Boyer proposed that F1 pays this price in the mechanical work necessary to liberate the nucleotide from the catalytic site. Further, release of product (ATP) proceeds sequentially and cyclically around the a3b3 hexamer because the synthetic reactions are synchronized in a fixed-phase relationship by rotation of the g shaft and cooperative coupling between the three catalytic sites. Boyer’s "binding change" mechanism neatly fits the Walker structure, and Boyer shared the 1997 Nobel Prize. A schematic diagram of the binding change mechanism is shown in Fig. 3.
Figure 2. Stereo pair showing the molecular structure of the Fx subunit run through center, coils extending out from mid in light gray, and the two coils of the g subunit medium gray. Its asymmetrical structure is evident. In the b subunit: the s is in dark gray, and the lower hinge segment is medium gray. The structure of the e subunit (not shown) is known, but it the g and c subunits.
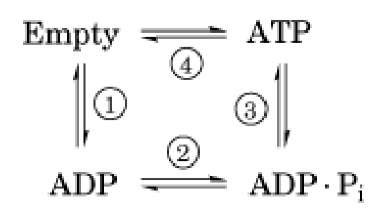
Figure 3. The binding change mechanism. Notation for site occupancies: T = ATP bound, DP = ADP • P; bound, D = AD numbered clockwise. The lengths of the arrows indicate the relative binding affinities. (a) The system starts with either ( ![]() . (b) Clockwise rotation of g increases the binding affinity of ADP in b1, traps ATP in b2, ; (c) Further rotation of g traps ADP and allows Pi binding in b1, releases the tightly bound ATP and allows ADP binding
. (b) Clockwise rotation of g increases the binding affinity of ADP in b1, traps ATP in b2, ; (c) Further rotation of g traps ADP and allows Pi binding in b1, releases the tightly bound ATP and allows ADP binding
There are actually six nucleotide-binding sites on the a3b3 hexamer. All lie at the interfaces between the a and b subunits. The catalytic sites lie mostly in the b subunit, whereas the noncatalytic sites lie mostly in the a subunit. The role of the noncatalytic sites is uncertain, but they may help to hold the hexamer together. Each catalytic site traverses the synthetic cycle sequentially:
In steps 1 and 2, a site binds ADP and phosphate (not necessarily in that order). While trapped in the catalytic site in step 3, the reactants (ADP and Pj) and product (ATP) are in chemical equilibrium.
Step 4 requires the input of mechanical torque from F0 on g to trap the reactants in the ATP state and to pry open the site, releasing the tightly bound ATP. Most of the 12 kcal/mol price of synthesis is paid in step 4. The way in which this works is found in the shape of the a3b3 hexamer and the g shaft.
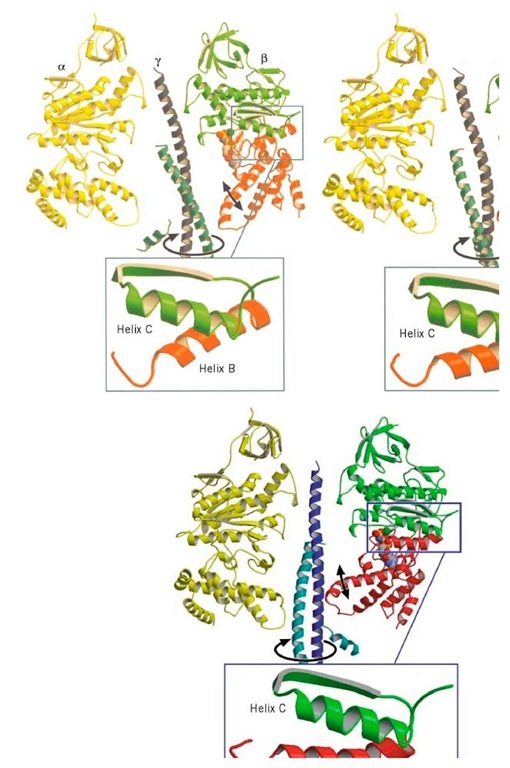
The g subunit is asymmetric and bowed. It fits into a central annulus in a3b3, which is itself asymmetric (Fig. 4). At the top of the a3b3 hexamer is a hydrophobic "sleeve" in which the g shaft rotates. Further down, however, the annulus is offset from the center, so that as g rotates clockwise, it sequentially pushes outward on each catalytic site. In addition, the e subunit is located eccentrically and attached to the g and c subunits, so that as g rotates, it comes into contact sequentially with each b subunit in a conserved region called the DELSEED sequence (named for the single-letter abbreviation of its constituent amino acid residues). Together, this asymmetrical rotation exerts stress on the catalytic site, loosening its grip on ATP, so that thermal fluctuations can free it into solution.
Figure 4. Cross section of F1 showing the conformational changes in the b subunits that drive rotation of g. The a subunit is on the left and the stationary barrel region of b is on the right. During the hydrolytic cycle, the lower segment of b undergoes a hinge-bending motion that rotates it about 30° inward. This motion pushes on the eccentric g coiled coil, causing it to rotate within the barrel bearing. During synthesis, the rotation of g pushes on each catalytic site. The panels show three snapshots of the motion during a 180° rotation.
(3). These interactions may mediate phosphate- and nucleotide-binding, the necessary precursors to synthesis. In addition, the catalytic sites are elastically coupled, so that the occupancy of one site affects the affinity of the other two sites. The consequence of this coupling is that when ATP concentrations are low enough so that only one site is occupied, hydrolysis proceeds much more slowly than when more than one site is occupied.
Together with the F1 molecular structure, the binding change model strongly supports the idea that catalysis involves rotation of the g subunit. However, dramatic visual confirmation was provided by in vitro experiments in which the a3b3g subunits were isolated and attached to a bead. A fluorescently tagged actin filament was attached to the g shaft, and when ATP was supplied, the filament could be clearly seen rotating. In fact, a complete revolution takes place in three steps and consumes a single ATP per step (4).
The viscous drag on the actin filament was estimated, which allowed computing the torque developed by the F1 motor and comparing it with the free energy available from ATP hydrolysis.
The startling result was that the motor generates an average torque of more than 40 piconewton nanometers![]() more than six times the maximum force developed by kinesin or myosin. More impressively, the motor operates near 100% mechanical efficiency, this precludes any sort of heat engine, which would be limited by the Carnot efficiency (4). Several models have been proposed that address the issue of torque generation and efficiency (4-6).
more than six times the maximum force developed by kinesin or myosin. More impressively, the motor operates near 100% mechanical efficiency, this precludes any sort of heat engine, which would be limited by the Carnot efficiency (4). Several models have been proposed that address the issue of torque generation and efficiency (4-6).
The energy to drive this motion derives from the hydrolytic cycle of ATP at the catalytic site. Moreover, the conformational change that drives the hydrolysis motor must be nearly the reverse of the motion that frees ATP from the catalytic site during synthesis. Examination of the structure reveals that the major conformational change is a hinge-bending motion in the b subunits. The bottom portion of each b subunit below the nucleotide-binding site rotates inward approximately 30°, during which it pushes on the bowed g subunit, turning it much like one cranks an automobile jack (Fig. 4).
2. F0 Converts Proton Motive Force into Rotary Torque
Currently, there have been no direct observations of rotation in the F0 portion of ATP synthase (7).
However, current thinking is that the F0 assembly converts the energy contained in the transmembrane proton motive force into a rotary torque at the interface of the a and c subunits (Fig. 1). This torque turns the rotor (the c, g, and r subunits), which couples to the F1 synthetic machine.
The c assembly consists of 12 subunits, each consisting of two transmembrane alpha-helices (8). There is one essential acidic amino acid (Asp61 in the Escherichia coli ATP synthase) which binds protons. Because variants of ATP synthase can operate on sodium rather than protons, the interaction between the c subunit and the translocated ion has the property of an electrostatic carrier mechanism (9).
The a subunit consists of six transmembrane a-helices that contain at least one essential basic residue (Arg210 in E. coli) (8, 10). The electrostatic interaction of these rotor and stator charges is essential for torque generation, and several proposals have been put forward for the way this could work (11-14). Whatever the mechanism, the F0 motor must generate a torque sufficient to liberate three ATP’s from the three catalytic sites in F1 for each revolution. If the proton flux through the stator is tightly coupled to the rotation of the c subunit, then a rotation of 2p/3 carries four protons down the electromotive potential of 230 mV typical of the mitochondrial inner membrane (14). This is sufficient to account for the mechanical energy required for synthesis of one ATP.
Under anaerobic conditions, the ATP synthase of the bacteria E. coli can reverse its operation, hydrolyzing ATP and turning the c subunit backward so that it functions as a proton pump. This is not surprising because the F-ATPases are structurally similar to the most common proton pumps, the vacuolar, or V-ATPases (7). These pumps may have been the evolutionary precursors of ATP synthase (15). A striking difference between the two is that the F-ATPases have 12 acidic rotor charges, whereas the V-ATPases have six. It can be shown that this enables the V-ATPases to function more efficiently as ion pumps at the expense of relinquishing their capability to synthesize ATP.
3. Summary
Both the F1 and F0 motors can operate in both directions. F1 is a hydrolytically driven three-piston engine that can be driven in reverse to synthesize ATP from ADP and phosphate. F0 is an ion- driven rotary engine that can be driven in reverse to function as an ion pump. The F-ATPases are structurally similar to and presumably evolutionarily related to the V-ATPase ion pumps (15). It is thought that most ion pumps function by an "alternating access" mechanism, whereby an ion is first bound strongly on the dilute side, then energy is supplied to move the ion so that it communicates with the concentrated side and weakens its binding affinity (16). In contrast with other ion pumps, however, the F and V-ATPases accomplish this by a rotary mechanism that is driven indirectly by nucleotide hydrolysis, rather than by direct phosphorylation (17). It is thought that the F0 motor is also related to the bacterial flagellar motor. Both can operate on sodium, although the flagellar motor has eight or more "stators" and develops far more torque than F0 (18, 19).
The mechanism driving the F1 hydrolytic motor may carry hints for other nucleotide hydrolytically fueled motors, such as kinesin, myosin, and dynein. However, important structural differences may make the comparison difficult (4). For example, the motors previously mentioned all "walk" along a polymer track to which they bind tightly during a portion of their mechanochemical cycle. The power stroke of the F1 motor is driven by the b subunit, which pushes on the g shaft, but does not bind tightly to it, that is, it does not "walk" around the g shaft. Moreover, no other motor operates with nearly the efficiency of the F1 motor, implying that there are important entropic steps in other motors that are absent in the F1 motor.