The study of the rates of enzyme-catalyzed transformations provides invaluable information as to the number of steps and their magnitude in the catalytic process. The most common method is to use steady-state conditions in which the enzyme is at <10_8 M concentration and the substrate(s) \xM or higher. In the simplest case of the conwhere S and P are the substrate and the product, E is the free enzyme, and ES is the associated enzyme ■ substrate complex, also called a Michaelis complex. The rate constants k\, k_1, k2, and k_2 describe the rates of each step in the reaction. Because the concentration of ES is not changing, and so is at the steady state, the kinetic scheme can be solved by relating the initial velocity at a given substrate concentration to both the maximum velocity, Vmax, and the substrate concentration at which the initial velocity reaches one-half the maximum velocity, KM, through the equation
The term KM is the ratio (k_1 + k2)/k1 and only approximates the binding of S to E . The turnover number, or kCat, is simply Vmax/[Eo] where Eo is the total enzyme concentration. A description of the transformation of substrate to product generally shows V as a hyperbolic function of S concentration with V increasing asymptotically toward Vmax as the active site becomes saturated with S.
Even in this simple case, the extraction of the magnitude of the four specific rate constants requires numerical analysis, with additional complexity being introduced by the appearance of intermediates or the requirement for a second or third substrate. These complications lead to equations in which the additional rate constants cannot be calculated from steady-state data. However, the analogous terms for KM and kcat can be calculated and hold similar meanings. Perhaps the most useful application of steady-state kinetics at this level is the recognition of diagnostic patterns in the reciprocal replots of the initial velocity data as a function of substrate concentration. Two-substrate reactions fall into two general classes represented by version of a single substrate to product, the kinetic scheme is generally represented by
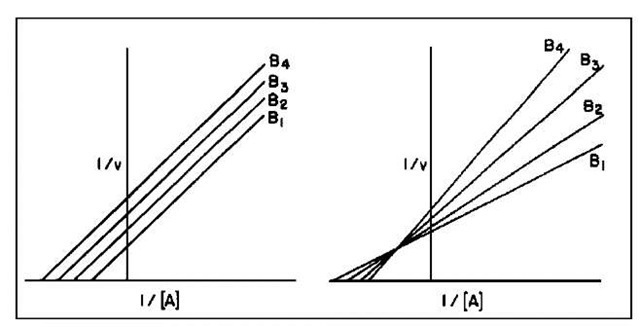
The difference is that in the former process a fragment X of substrate A is transferred covalently to the enzyme and then to the second substrate, whereas in the latter no free enzyme bearing a covalently linked fragment X is formed. Within the second pathway, the addition of A and B, and similarly the release of products C and D, can be ordered (as written) or random. The two pathways give rise to the representative graphs shown in Fig. 1.
FIGURE 1 Plots of the reciprocal initial velocity against the reciprocal concentration of substrate A for a two-substrate reaction at several different concentrations of substrate B. The plot on the left reflects a mechanism in which a free enzyme bearing a covalently linked group is generated, while that on the right shows a sequential one in which two substrates bind and the reaction occurs.
As one might imagine, the kinetic rate laws associated with these mechanisms are generally too complex for dissection of single steps or their evaluation. Moreover, the method provides evidence for only the minimal number of intermediates in a pathway since the form of the equations is unchanged by including multiple species.
Steady-state kinetic parameters such as kcat and KM can vary when they are studied as a function of pH. After one corrects for ionizations of the substrate and controls for possible effects on the native structure of the enzyme, variations in kcat and KM can often be assigned to ionizations of acid/base groups at the active site of the enzyme. The term kcat/KM reflects the proton dissociation constants of the free enzyme, provided that the proton transfers remain fast relative to other steps in the pathway. In the simple one-intermediate kinetic sequence expanded to implicate two ionizations, the term kcat/KM would display pKa and pKb; the term kcat would reflect pK’a and pKb. The pH dependence of the kcat parameter affords information about the substrate-bound state.
Materials that bind to the enzyme either at the active site or at a distal site and slow the turnover of the enzyme but are not themselves transformed act as inhibitors. These compounds may or may not be structurally similar to the substrate; nevertheless, their binding, particularly at the active site, often provides important complexes for structure determination. The most commonly studied type of inhibition is termed competitive, which means that the substrate and the inhibitor compete directly for the active site of the enzyme. The effect of this type of inhibitor on the steady-state kinetic parameters is to alter the graphical evaluation of the Michaelis constant but not the value of Vmax, which can still be attained in the presence of the inhibitor provided that the substrate concentration is high enough. Binding of the inhibitor to regions divorced from that binding the substrate always affects the evaluation of Vmax because no concentration of substrate is sufficient to displace the inhibitor.
The most useful approaches for obtaining information regarding the existence of intermediates and their lifetimes are fast reaction methods that mix enzyme and substrate within milliseconds, which permits the observation of single turnover events by various spectroscopic methods. Alternatively the reaction is rapidly quenched at known time intervals and its progress is analyzed chromatographically. In many cases in which an intermediate accumulates to the level of the enzyme concentration, such methods reveal the presence of "burst kinetic" that feature the rapid buildup of the intermediate in the transient phase followed by its slower rate of formation/decay in the steady state. The simplest kinetic scheme consistent with this phenomenon is given by
![tmp4627_thumb[2][2] tmp4627_thumb[2][2]](http://what-when-how.com/wp-content/uploads/2011/05/tmp4627_thumb22_thumb.jpg)
![tmp4628_thumb[2][2] tmp4628_thumb[2][2]](http://what-when-how.com/wp-content/uploads/2011/05/tmp4628_thumb22_thumb.jpg)
![tmp4630_thumb[2][2] tmp4630_thumb[2][2]](http://what-when-how.com/wp-content/uploads/2011/05/tmp4630_thumb22_thumb.jpg)
