CONCEPT
Of the 92 elements produced in nature, only six are critical to the life of organisms: hydrogen, carbon, nitrogen, oxygen, phosphorus, and sulfur. Though these elements account for 95% of the mass of all living things, their importance extends far beyond the biosphere. Hydrogen and oxygen, chemically bonded in the form of water, are the focal point of the hydrosphere, while oxygen and nitrogen form the bulk of the atmosphere. All six are part of complex biogeochemical cycles in which they pass through the biosphere, atmosphere, hydrosphere, and geosphere. These cycles circulate nutrients through the soil into plants, microbes, and animals, which return the elements to the earth system through chemical processes that range from respiration to decomposition.
HOW IT WORKS
The Elements
An element is a substance composed of a single type of atom, meaning that it cannot be broken down chemically to make a simpler substance. They are listed on the periodic table of elements, a chart that renders them in order of their atomic numbers, or the number of protons in the nucleus of the atom. The elements we want to discuss in the context of biogeochemical cycles are all low in atomic number, starting with hydrogen, which has just one proton in its nucleus. In the following list, the elements are cited by atomic number along with their chemical symbols, or the abbreviation by which they are known to chemists.
Elements Involved in Biogeochemical Cycles
• 1. Hydrogen (B)
• 6. Carbon (C)
• 7. Nitrogen (N)
• 8. Oxygen (O)
• 15. Phosphorus (P)
• 16. Sulfur (S)
Given the fact, as noted earlier, that 92 elements appear in nature, it should come as no surprise that the highest atomic number for any naturally occurring element is 92, for uranium. Beyond uranium there are about two dozen artificially created elements, but they are of little interest outside the realm of certain specialties in chemistry and physics. The naturally occurring elements are the ones that matter to the earth sciences, and of these elements, only a handful play a significant role.
In the essays Minerals and Economic Geology, other elements—most notably silicon—are discussed with regard to their importance in forming minerals, rocks, and ores. Though they are critical to Earth’s systems, elements other than the six discussed here play no role in bio-geochemical cycles. Indeed, it is a fact of the physical sciences that not all elements are created equal: certainly, the universe is not divided evenly 92 ways, with equal amounts of all elements. In fact, hydrogen and helium account for 99% of the mass of the entire universe.
Abundance
On Earth the ratios are quite different, however. Oxygen and silicon constitute the preponderance of the known mass of Earth’s crust, while nitrogen and oxygen form the overwhelming majority of the atmosphere. Hydrogen is proportionally much, much less abundant on Earth than in the universe as a whole, but owing to its role in forming water, a substance essential to the sustenance of life, it is unquestionably of great significance.
The two following lists provide rankings for the abundance of the six elements discussed in this essay. The first table shows their ranking and share in the entire known mass of the planet, including the crust, living matter, the oceans, and atmosphere. The second shows their relative abundance and ranking in the human body.
Abundance of Selected Elements on Earth (Ranking and Percentage)
• 1. Oxygen (49.2%)
• 9. Hydrogen (0.87%)
• 12. Phosphorus (0.11%)
• 14. Carbon (0.08%)
• 15. Sulfur (0.06%)
• 16. Nitrogen (0.03%)
Abundance of Selected Elements in the Human Body (Ranking and Percentage)
• 1. Oxygen (65%)
• 2. Carbon (18%)
• 3. Hydrogen (10%)
• 4. Nitrogen (3%)
• 6. Phosphorus (1%)
• 9. Sulfur (0.26%)
Several things are interesting about these figures. First and most obviously, there is the fact that the ranking of all these elements (with the exception of oxygen) is relatively low in the total known elemental mass of Earth, whereas their ranking is much, much higher within the human body. This is significant, given the fact that these elements are all essential to the lives of organisms.
Furthermore, note that it does not take a great percentage to constitute an “abundant” element: even nitrogen, with its 0.03% share of Earth’s total known mass, still is considered abundant. The presence of the vast majority of elements on Earth is measured in parts per million (ppm) or even parts per billion (ppb).
Chemistry and Geochemistry
In a general sense, chemistry can be defined as an area of the physical sciences concerned with the composition, structure, properties, and changes of substances, including elements, compounds, and mixtures. This definition unites the phases in the history of the development of the discipline,from early modern times—when it arose from alchemy, a set of mystical beliefs based on the idea that ordinary matter can be perfected—to modern times. Our modern understanding of chemistry, however, is quite different from the model of chemistry that prevailed until about 1800, a difference that relates to a key discovery: the atom.
This change, in fact, should be described not in terms of a discovery so much as the development of a model. Long before chemists and physicists comprehended the structure of the atom, they developed an understanding of all chemical substances as composed of atomic units, each representing one and only one element. Until the development of this model— thanks to a number of chemists, most notably, John Dalton (1766-1844) of England, Antoine Lavoisier (1743-1794) of France, and Amedeo Avogadro (1776-1856) of Italy—chemistry was concerned primarily with mixing potions and observing their effects. Thanks to the atomic model, chemists never again would confuse mixtures with chemical compounds.
Chemical reactions
The difference between a mixture and a compound goes to the heart of the distinction between physics and chemistry. A mixture, such as coffee, is the result of a physical process—in this case, the heating of water and coffee beans—and the result does not have a uniform chemical structure. On the other hand, a compound results from chemical reactions between atoms, which form enormously powerful bonds in the process of joining to create a molecule. A molecule is the basic particle of a compound, just as an atom is to an element. It should be noted that some elements, such as nitrogen, typically appear in diatomic form, that is, two atoms bond to form a molecule of nitrogen.
A substance may undergo physical changes without experiencing any alteration in its underlying structure; on the other hand, a chemical reaction makes a fundamental change to the substance. In a chemical reaction, a substance may experience a change of state (i.e., from solid to liquid or gas) without undergoing any physical process of being heated or cooled by an outside source. Chemical reactions involve the breaking of bonds between atoms in a molecule and the formation of new bonds. As a result, an entirely new substance is created—something that could never be achieved through mere physical processes.
Molecular structure. A compound is formed by chemical reactions between atoms, which join together to make molecules.
REAL-LIFE APPLICATIONS
Geochemistry
Just as geochemistry is a branch of the geologic sciences that weds physics and geology,so there is a geologic subdiscipline, geochemistry, in which chemistry and the geologic sciences come together. Geochemistry is concerned with the chemical properties and processes of Earth—in particular, the abundance and interaction of chemical elements and their isotopes. (Isotopes are atoms that have an equal number of protons, and hence are of the same element, but differ in their number of neutrons. Isotopes may be either stable or unstable, in which case they are subject to the emission of high-energy particles. Some elements have numerous stable isotopes, others have only one or two, and some have none.)
Before the mid-twentieth century, geochemistry had a relatively limited scope, confined primarily to the identification of elements in rocks and minerals and the determination of the relative abundance of those elements. Since that time, however, this subdiscipline has come to encompass many other concerns, particularly those discussed in the present context. Geochemistry today focuses on such issues as the recycling of elements between the various sectors of the earth system, especially between living and nonliving things.
Biogeochemical Cycles and Earth Systems
The changes that a particular element undergoes as it passes back and forth through the various earth systems, and particularly between living and nonliving matter, are known as biogeochemical cycles. The four earth systems involved in these cycles are the atmosphere, the biosphere (the sum of all living things as well as formerly living things that have not yet decomposed), the hydrosphere (the entirety of Earth’s water except for vapor in the atmosphere), and the geosphere. The last of these spheres is defined as the upper part of Earth’s continental crust, or that portion of the solid earth on which human beings live and which provides them with most of their food and natural resources.
Carbon, for instance, is present in all living things on Earth. Hence, the phrase carbon-based life-form, a cliche found in many an old science-fiction movie, is actually a redundancy: all life-forms contain carbon. In the context of the physical sciences, organic refers to all substances that contain carbon, with the exception of oxides, such as carbon dioxide and carbon monoxide, and carbonates, which are found in minerals. Still, carbon circulates between the organic world and the inorganic world, as when an animal exhales carbon dioxide.
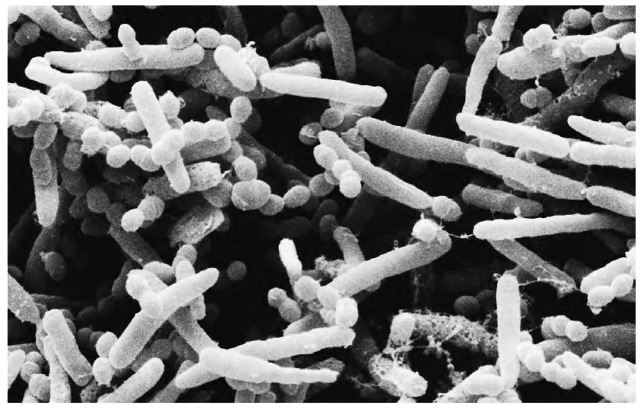
Bacteria begin the process of decomposition of organic waste, breaking down plant matter and converting it into compost.
The carbon cycle is of such importance to the functioning of Earth that it is discussed separately (see Carbon Cycle). So, too, is the nitrogen cycle (see Nitrogen Cycle), whereby nitrogen passes between the soil, air, and biosphere as well as the hydrosphere. The hydrosphere, as noted earlier, is based on a single substance, water, created by the chemical bonding of hydrogen and oxygen, and it is likewise discussed in detail elsewhere (see Hydrologic Cycle).
Despite the emphasis here on carbon in the biosphere, nitrogen in the geosphere, and hydrogen and oxygen in the hydrosphere, it should be noted that biogeochemical cycles involving these four elements take them through all four “spheres.” The same is true of sulfur, whose bio-geochemical cycle is discussed later in this essay. On the other hand, phosphorus, also discussed later, is present in only three of Earth’s systems; it plays little role in the atmosphere.
Decomposers and Detritivores
Most biogeochemical cycles involve a special type of chemical reaction known as decomposition, and for this to take place, agents of decomposition—known as decomposers and detritivores— are essential. Decomposition occurs when a compound is broken down into simpler compounds or into its constituent elements. This is achieved primarily by decomposers, organisms that obtain their energy from the chemical breakdown of dead organisms as well as from animal and plant waste products.
The principal forms of decomposer are bacteria and fungi. These creatures carry enzymes, which they secrete into the materials they consume, breaking them down chemically before taking in the products of this chemical breakdown. They thus take organic matter and render it in inorganic form, such that later it can be taken in again by plants and returned to the biosphere.
Detritivores are much more complex organisms, but their role is similar to that of decomposers. They, too, feed on waste matter, breaking this organic material down into inorganic substances that then can become available to the biosphere in the form of nutrients for plants. Examples of detritivores are earthworms and maggots. As discussed in Energy and Earth, detri-tivores are key players in the food web, the set of nutritional interactions—sometimes called a food chain—between living organisms.
Phosphorus and the Phosphorus Cycle
There are a few elements that were known in ancient or even prehistoric times, examples being gold, iron, lead, and tin. The vast majority, on the other hand, have been discovered since the beginning of the modern era, and the first of them was phosphorus, which is also the first element whose discoverer is known.
In 1674 the German alchemist Hennig Brand (ca. 1630-ca. 1692) was searching for the philosopher’s stone, a mythical substance that allegedly would turn common or base metals into gold. Convinced that he would find this substance in the human body, Brand evaporated water from a urine sample and burned the precipitate (the solid that remained) along with sand. The result was a waxy, whitish substance that glowed in the dark and reacted violently with oxygen. Brand named it phosphorus, a name derived from a Greek term meaning “light-bearer.”
Owing to its high reactivity with oxygen, phosphorus is used in the production of safety matches, smoke bombs, and other incendiary devices. It is also important in various industrial applications and in fertilizers. In fact, ancient humans used phosphorus without knowing it when they fertilized their crops with animal bones.
Phosphates
In the early 1800s, chemists recognized that the critical component in bones was phosphorus, which plants use in photosynthesis—the biological conversion of energy from the Sun into chemical energy (see Energy and Earth). With this discovery came the realization that phosphorus would make an even more effective fertilizer when treated with sulfuric acid, which makes it soluble, or capable of being dissolved, in water. This compound, known as superphosphate, can be produced from phosphates, a type of mineral.
Phosphates represent one of the eight major classes of mineral (see Minerals). All phosphates contain a characteristic formation, PO4, which is bonded to other elements or compounds—for example, with aluminum in aluminum phosphate, or AlPO4. Phosphorus fertilizer is typically calcium phosphate, known as bone ash, the most important industrial mineral (see Economic Geology) produced from phosphorus. Another significant phosphate is sodium phosphate, used in dishwashing detergents. In fact, phosphates once played a much larger role in the detergent industry—with disastrous consequences, as we shall see.
The phosphorus cycle
The majority of phosphorus in the earth system is located in rocks and deposits of sediment, from which it can be removed by one of three processes: weathering, the breakdown of rocks and minerals at or near the surface of Earth as the result of physical, chemical, or biological processes (see Erosion, Sediment and Sedimentation); leaching, the removal of soil materials that are in solution, or dissolved in water; and mining.
Phosphorus is highly reactive, meaning that it is likely to bond with other elements, and for this reason it often is found in compounds. Microorganisms absorb insoluble phosphorus compounds (ones that are incapable of being dissolved) and, through the action of acids within the microorganisms, turn them into soluble phosphates. Algae and other green plants absorb these phosphates and, in turn, are eaten by animals. When they die, the animals release the phosphates back into the soil.
As with all elements, the total amount of phosphorus on Earth stays constant, but the distribution of it does not. Some of the phosphorus passes from the geosphere into the biosphere, but the majority of it winds up in the ocean. It may find its way into sediments in shallow waters, in which case it continues to circulate, or it may be taken to the deep parts of the seas, in which case it is likely to be deposited for the long term.
Fish absorb particles of phosphorus, and thus some of the element returns to dry land through the catching and consumption of seafood. In addition, guano, or dung, from birds that live in an ocean environment (e.g., seagulls) also returns portions of phosphorus to the terrestrial environment. Nonetheless, geochemists believe that phosphorus is being transferred steadily to the ocean, from whence it is not likely to return. It is for this reason that phosphorus-based fertilizers are important, because they feed the soil with nutrients that otherwise would be continually lost.

A stand of fir trees shows the devastating effects of acid rain, which is created when sulfuric acid mixes with moisture in the atmosphere.
EUTROPHICATION
To be sure, phosphorus, in the proper quantities, is good for the environment. But as with medicine or any other beneficial substance, if a little is good, that does not mean that a lot is necessarily better. In the case of phosphorus, an overabundance of the element in the environment can lead to a phenomenon called eutrophication, a state of heightened biological productivity in a body of water. One of the leading causes of eutrophication (from a Greek term meaning “well nourished”) is a high rate of nutrient input, in the form of phosphates or nitrates, a nitrogen-oxygen compound (see Nitrogen Cycle).
As a result of soil erosion, fertilizers make their way into bodies of water, as does detergent runoff in wastewater. Excessive phosphates and nitrates stimulate growth in algae and other green plants, and when these plants die, they drift to the bottom of the lake or other body of water. There, decomposers consume the remains of the plants and, in the process, also use oxygen that otherwise would be available to fish, mollusks, and other forms of life. As a result, those species die off, to be replaced by others that are more tolerant of lowered oxygen levels—for example, worms. Needless to say, the outcome of eutrophication is devastating to the lake’s ecosystem.
During the 1960s, Lake Erie—one of the Great Lakes on the U.S.-Canadian border— became an example of eutrophication gone mad. As a result of high phosphate concentrations, Erie’s waters were choked with plant and algae growth. Fish were unable to live in the water, the beaches reeked with the smell of decaying algae, and Erie became widely known as a “dead” body of water. This situation led to the passage of new environmental standards and pollution controls by both the United States and Canada, whose governments acted to reduce drastically the phosphate content in fertilizers and detergents. Lake Erie proved to be an environmental success story: within a few decades the lake once again teemed with life.
Sulfur and the Sulfur Cycle
If there is any element that can be said to have a bad image—and a falsely bad one at that—it is sulfur. As everyone “knows,” sulfur has a foul smell, and this smell, combined with its combustibility, led to the biblical association of brimstone—the ancient name for the element—with the fires of hell. It may come as a surprise, then, to learn that sulfur has no smell of its own. Only in combination with other elements does it acquire the offensive odor that has led to its unpleasant reputation.
KEY TERMS
Alchemy: A set of mystical beliefs based on the idea that ordinary matter can be perfected. Though it was a pseudo-science, alchemy, which flourished in the late Middle Ages, was a forerunner of scientific chemistry.
Atmosphere: In general, an atmosphere is a blanket of gases surrounding a planet. Unless otherwise identified, however, the term refers to the atmosphere of Earth, which consists of nitrogen (78%), oxygen (21%), argon (0.93%), and other substances that include water vapor, carbon dioxide, ozone, and noble gases such as neon (0.07%).
Atomic number: The number of protons in the nucleus of an atom. Since this number is different for each element, elements are listed on the periodic table in order of atomic number.
Biogeochemical cycles: The changes that particular elements undergo as they pass back and forth through the various earth systems and particularly between living and nonliving matter. The elements involved in biogeochemical cycles are hydrogen, oxygen, carbon, nitrogen, phosphorus, and sulfur.
Biosphere: A combination of all living things on Earth—plants, mammals, birds, amphibians, reptiles, aquatic life, insects, viruses, single-cell organisms, and so on—as well as all formerly living things that have not yet decomposed.
Carnivore: A meat-eating organism.
Chemical bonding: The joining through electromagnetic force of atoms that sometimes, but not always, represent more than one chemical element. The result is the formation of a molecule.
Chemical symbol: A one-letter or two-letter abbreviation for the name of an element.
Compound: A substance made up of atoms of more than one element chemically bonded to one another.
Decomposers: Organisms that obtain their energy from the chemical breakdown of dead organisms as well as from animal and plant waste products. The principal forms of decomposers are bacteria and fungi.
Decomposition reaction: A chemical reaction in which a compound is broken down into simpler compounds or into its constituent elements. In the earth system, this often is achieved through the help of detritivores and decomposers.
Detritivores: Organisms that feed on waste matter, breaking organic material down into inorganic substances that then can become available to the biosphere in the form of nutrients for plants. Their function is similar to that of decomposers; however, unlike decomposers—which tend to be bacteria or fungi—detritivores are relatively complex organisms, such as earthworms or maggots.
Ecosystem: A term referring to a community of interdependent organisms along with the inorganic components of their environment.
Element: A substance made up of only one kind of atom. Unlike compounds, elements cannot be broken chemically into other substances.
Eutrophication: A state of heightened biological productivity in a body of water, which is typically detrimental to the ecosystem in which it takes place. Eutrophication can be caused by an excess of nitrogen or phosphorus in the form of nitrates and phosphates, respectively.
Food web: A term describing the interaction of plants, herbivores, carnivores, omnivores, decomposers, and detri-tivores, each of which consumes nutrients and passes it along to other organisms.
Geochemistry: A branch of the earth sciences, combining aspects of geology and chemistry, that is concerned with the chemical properties and processes of Earth—in particular, the abundance and interaction of chemical elements and their isotopes.
Geosphere: The upper part of Earth’s continental crust, or that portion of the solid earth on which human beings live and which provides them with most of their food and natural resources.
Herbivore: A plant-eating organism.
Hydrosphere: The entirety of Earth’s water, excluding water vapor in the atmosphere but including all oceans, lakes, streams, groundwater, snow, and ice.
Leaching: The removal of soil materials that are in solution, or dissolved in water.
Mineral: A naturally occurring, typically inorganic substance with a specific chemical composition and a crystalline structure.
Mixture: A substance with a variable composition, meaning that it is composed of molecules or atoms of differing types. Compare with compound.
Molecule: A group of atoms, usually but not always representing more than one element, joined in a structure. Compounds typically are made up of molecules.
Omnivore: An organism that eats both plants and other animals.
Organic: At one time chemists used the term organic only in reference to living things. Now the word is applied to most compounds containing carbon, with the exception of carbonates (which are minerals) and oxides, such as carbon dioxide.
Periodic table of elements: A chart that shows the elements arranged in order of atomic number along with their chemical symbols and the average atomic mass for each particular element.
Photosynthesis: The biological conversion of light energy (that is, electromagnetic energy) from the Sun to chemical energy in plants.
Reactivity: A term referring to the ability of one element to bond with others. The higher the reactivity, the greater the tendency to bond.
Sediment: Material deposited at or near Earth’s surface from a number of sources, most notably preexisting rock. There are three types of sediment: rocks, or clastic sediment; mineral deposits, or chemical sediment; and organic sediment, composed primarily of organic material.
Soluble: Capable of being dissolved.
Solution: A homogeneous mixture (i.e., one that is the same throughout) in which one or more substances is dissolved in another substance—for example, sugar dissolved in water.
Weathering: The breakdown of rocks and minerals at or near the surface of Earth due to physical, chemical, or biological processes.
An example of such a compound is hydrogen sulfide, a poisonous substance present in intestinal gas. The May 2001 National Geographic included two stories relating to the presence of natural hydrogen sulfide deposits on opposite sides of the earth, and in both cases the presence of these toxic fumes created unusual ecosystems.
A system of caves known as Villa Luz in southern Mexico contains some 20 underground springs that carry large quantities of hydrogen sulfide. Among the strange creatures that have made Villa Luz their home are species of fish colored bright red; the pigmentation is a result of the fact that they have to produce high quantities of hemoglobin (a component in red blood cells) to survive on the scant oxygen. The waters of the cave also are populated by microorganisms that oxidize the hydrogen sulfide and turn it into sulfuric acid, which dissolves the rock walls and continually enlarges the cave.
Thousands of miles away, in the Black Sea, explorers examining evidence of a great ancient flood like the one depicted in the Bible (see Earth, Science, and Nonscience) found an unexpected ally in the form of hydrogen sulfide. Because the Black Sea lacks the temperature differences that cause water to circulate from the bottom upward, hydrogen sulfide had gathered at the bottom and stayed there, covered by dense layers of saltwater. Oxygen could not reach the bottom of the Black Sea, and thus wood-boring worms could not live in the toxic environment. As a result, a 1,500-year-old shipwreck had been virtually undisturbed.
The sulfur cycle
Sulfur is removed from rock by weathering, at which point it reacts with oxygen in the air to form sulfate, or SO4. This sulfate is taken in by plants and microorganisms, which convert it to organic materials and pass it on to animals in the food web. Later, when these organisms die, decomposers absorb the sulfur from their bodies and return it to the environment. As with phosphorus, however, sulfur is being lost continually to the oceans as it drains through lakes and streams (and through the atmosphere) on its way to the sea.
In the ocean ecosystem, sulfur can take one of three routes. Some of it circulates through food webs, and some drifts to the bottom to bond with iron in the form of ferrous sulfide, or FeS. Ferrous sulfide contributes to the dark color of sediments at the bottom of the ocean. On the other hand, sulfur may be returned to the atmosphere, released by spray from saltwater. In addition, sulfur can pass into the atmosphere as the result of volcanic activity or through the action of bacteria, which release it in the form of hydrogen sulfide, the foul-smelling gas discussed earlier.
As with all biogeochemical cycles, humans play a part in the sulfur cycle, and the role of modern industrial society is generally less than favorable, as is true of most such cycles. A particularly potent example is the production of acid rain. Among the impurities in coal is sulfur, and when coal is burned (as it still is, for instance, in electric power plants), it results in the production of sulfur dioxide and sulfur trioxide—SO2 and SO3, respectively. Sulfur trioxide reacts with water in the air to produce sulfuric acid, or H2SO4. This mixes with moisture in the atmosphere to create acid rain, which is hazardous to both plant and animal life.
