CONCEPT
The actinides (sometimes called actinoids) occupy the “bottom line” of the periodic table—a row of elements normally separated from the others, placed at the foot of the chart along with the lanthanides. Both of these families exhibit unusual atomic characteristics, properties that set them apart from the normal sequence on the periodic table. But there is more that distinguishes the actinides, a group of 14 elements along with the transition metal actinium. Only four of them occur in nature, while the other 10 have been produced in laboratories. These 10 are classified, along with the nine elements to the right of actinium on Period 7 of the periodic table, as transuranium (beyond uranium) elements. Few of these elements have important applications in daily life; on the other hand, some of the lower-number transuranium elements do have specialized uses. Likewise several of the naturally occurring actinides are used in areas ranging from medical imaging to powering spacecraft. Then there is uranium, “star” of the actinide series: for centuries it seemed virtually useless; then, in a matter of years, it became the most talked-about element on Earth.
HOW IT WORKS
The Transition Metals
Why are actinides and lanthanides set apart from the periodic table? This can best be explained by reference to the transition metals and their characteristics. Actinides and lanthanides are referred to as inner transition metals, because, although they belong to this larger family, they are usually considered separately—rather like grown children who have married and started families of their own.
The qualities that distinguish the transition metals from the representative or main-group elements on the periodic table are explained in depth within the Transition Metals essay. The reader is encouraged to consult that essay, as well as the one on Families of Elements, which further places the transition metals within the larger context of the periodic table. Here these specifics will be discussed only briefly.
Orbital patterns of the transition metals
The transition metals are distinguished by their configuration of valence electrons, or the outer-shell electrons involved in chemical bonding. Together with the core electrons, which are at lower energy levels, valence electrons move in areas of probability referred to as orbitals. The pattern of orbitals is determined by the principal energy level of the atom, which indicates a distance that an electron may move away from the nucleus.
Each principal energy level is divided into sublevels corresponding to the number n of the principal energy level. The actinides, which would be on Period 7 if they were included on the periodic table with the other transition metals, have seven principal energy levels. (Note that period number and principal energy level number are the same.) In the seventh principal energy level, there are seven possible sublevels.
The higher the energy level, the larger the number of possible orbital patterns, and the more complex the patterns. Orbital patterns loosely define the overall shape of the electron cloud, but this does not necessarily define the paths along which the electrons move. Rather, it means that if you could take millions of photographs of the electron during a period of a few seconds, the resulting blur of images would describe more or less the shape of a specified orbital.
The four basic types of orbital patterns are discussed in the Transition Metals essay, and will not be presented in any detail here. It is important only to know that, unlike the representative elements, transition metals fill the sublevel corresponding to the d orbitals. In addition, they are the only elements that have valence electrons on two different principal energy levels.
Lanthanides and actinides
The lanthanides and actinides are further set apart even from the transition metals, due to the fact that these elements also fill the highly complex /orbitals. Thus these two families are listed by themselves. In most versions of the periodic table, lanthanum (57) is followed by hafnium (72) in the transition metals section of the chart; similarly, actinium (89) is followed by rutherfordium (104). The “missing” metals— lanthanides and actinides, respectively—are shown at the bottom of the chart.
The lanthanides can be defined as those metals that fill the 4/orbital. However, because lanthanum (which does not fill the 4/orbital) exhibits similar properties, it is usually included with the lanthanides. Likewise the actinides can be defined as those metals that fill the 5/orbital; but again, because actinium exhibits similar properties, it is usually included with the actinides.
Isotopes
One of the distinguishing factors in the actinide family is its great number of radioactive isotopes. Two atoms may have the same number of protons, and thus be of the same element, yet differ in their number of neutrons—neutrally charged patterns alongside the protons at the nucleus. Such atoms are called isotopes, atoms of the same element having different masses.
Isotopes are represented symbolically in one of several ways. For instance, there is this format: “a”S, where S is the chemical symbol of the element, a is the atomic number (the number of protons in its nucleus), and m the mass num-ber—the sum of protons and neutrons. For the isotope known as uranium-238, for instance, this is shown as 92 U.
Because the atomic number of any element is established, however, isotopes are usually represented simply with the mass number thus: 238U. They may also be designated with a subscript notation indicating the number of neutrons, so that this information can be obtained at a glance without it being necessary to do the arithmetic.
For the uranium isotope shown here, this is written as 2922 U146.
Radioactivity
The term radioactivity describes a phenomenon whereby certain materials are subject to a form of decay brought about by the emission of high-energy particles, or radiation.
Types of particles emitted in radiation include:
• Alpha particles, or helium nuclei;
• Beta particles—either an electron or a subatomic particle called a positron;
• Gamma rays or other very high-energy electromagnetic waves.
Isotopes are either stable or unstable, with the unstable variety, known as radioisotopes, being subject to radioactive decay. In this context, “decay” does not mean “rot”; rather, a radioisotope decays by turning into another isotope. By continuing to emit particles, the isotope of one element may even turn into the isotope of another element.
Eventually the radioisotope becomes a stable isotope, one that is not subject to radioactive decay. This is a process that may take seconds, minutes, hours, days, years—and sometimes millions or even billions of years. The rate of decay is gauged by the half-life of a radioisotope sample: in other words, the amount of time it takes for half the nuclei (plural of nucleus) in the sample to become stable.
Actinides decay by a process that begins with what is known as K-capture, in which an electron of a radioactive atom is captured by the nucleus and taken into it. This is followed by the splitting, or fission, of the atom’s nucleus. This fission produces enormous amounts of energy, as well as the release of two or more neutrons, which may in turn bring about further K-capture. This is called a chain reaction.

Glenn T. Seaborg holds a sample containing artificial elements between 94 and 1D2 on the periodic table .
REAL-LIFE APPLICATIONS
The First Three Naturally Occurring Actinides
In the discussion of the actinides that follows, atomic number and chemical symbol will follow the first mention of an element. Atomic mass figures are available on any periodic table, and these will not be mentioned in most cases. The atomic mass figures for actinide elements are very high, as fits their high atomic number, but for most of these, figures are usually for the most stable isotope, which may exist for only a matter of seconds.
Though it gives its name to the group as a whole, actinium (Ac, 89) is not a particularly significant element. Discovered in 1902 by German chemist Friedrich Otto Giesel (1852-1927), it is found in uranium ores. Actinium is 150 times more radioactive than radium, a highly radioactive alkaline earth metal isolated around the same time by French-Polish physicist and chemist Marie Curie (1867-1934) and her husband Pierre (1859-1906).
Thorium.
More significant than actinium is thorium (Th, 90), first detected in 1815 by the renowned Swedish chemist Jons Berzelius (1779-1848). Berzelius promptly named the element after the Norse god Thor, but eventually concluded that what he had believed to be a new element was actually the compound yttrium phosphate. In 1829, however, he examined another mineral and indeed found the element he believed he had discovered 14 years earlier.
It 1898, Marie Curie and an English chemist named Gerhard Schmidt, working independently, announced that thorium was radioactive. Today it is believed that the enormous amounts of energy released by the radioactive decay of subterranean thorium and uranium plays a significant part in Earth’s high internal temperature. The energy stored in the planet’s thorium reserves may well be greater than all the energy available from conventional fossil and nuclear fuels combined.
Thorium appears on Earth in an abundance of 15 parts per million (ppm), many times greater than the abundance of uranium. With its high energy levels, thorium has enormous potential as a nuclear fuel. When struck by neutrons, thorium-232 converts to uranium-233, one of the few known fissionable isotopes—that is, isotopes that can be split to start nuclear reactions.
It is perhaps ironic that this element, with its potential for use in some of the most high-tech applications imaginable, is widely applied in a very low-tech fashion. In portable gas lanterns for camping and other situations without electric power, the mantle often contains oxides of thorium and cerium, which, when heated, emit a brilliant white light. Thorium is also used in the manufacture of high-quality glass, and as a catalyst in various industrial processes.
Protactinium
Russian chemist Dmitri Ivanovitch Mendeleev (1834-1907),father of the periodic table, used the table’s arrangement of elements as a means of predicting the discovery of new substances: wherever he found a “hole” in the table, Mendeleev could say with assurance that a new element would eventually be found to fill it. In 1871, Mendeleev predicted the discovery of “eka-tantalum,” an element that filled the space below the transition metal tantalum. (At this point in history, just two years after Mendeleev created the periodic table, the lanthanides and actinides had not been separated from the rest of the elements on the chart.)
Forty years after Mendeleev foretold its existence, two German chemists found what they thought might be Element 91. It had a half-life of only 1.175 minutes, and, for this reason, they named it “brevium.” Then in 1918, Austrian physicist Lise Meitner (1878-1968)—who, along with Curie and French physicist Marguerite Perey (1909-1975) was one of several women involved in the discovery of radioactive elements—was working with German chemist Otto Hahn (1879-1968) when the two discovered another isotope of Element 91. This one had a much, much longer half-life: 3.25 • 104 years, or about five times as long as the entire span of human civilization.
Originally named protoactinium, the name of Element 91 was changed to protactinium (Pa), whose longest-lived isotope has an atomic mass of 231. Shiny and malleable, protactinium has a melting point of 2,861.6°F (1,572°C). It is highly toxic, and so rare that no commercial uses have been found for it. Indeed, protactinium could only be produced from the decay products of uranium and radium, and thus it is one of the few elements with an atomic number less than 93 that cannot be said to occur in nature.
Uranium
Uranium’s early history
Chemistry topics, in fact, differ as to the number of naturally occurring elements. Some say 88, which is the most correct figure, because protactinium, along with technetium (43) and two others, cannot be said to appear naturally on Earth. Other topics say 92, a less accurate figure that nonetheless reflects an indisputable fact: above uranium (U, 92) on the periodic table, there are no elements that generally occur in nature. (However, a few do occur as radioactive by-products of uranium.)
But uranium is much more than just the last truly natural element, though for about a century it apparently had no greater importance. When German chemist Martin Heinrich Klaproth (1743-1817) discovered it in 1789, he named it after another recent discovery: the planet Uranus. During the next 107 years, uranium had a very quiet existence, befitting a rather dull-looking material.
Though it is silvery white when freshly cut, uranium soon develops a thin coating of black uranium oxide, which turns it a flat gray. Yet glassmakers did at least manage to find a use for it—as a coating for decorative glass, to which it imparted a hazy, fluorescent yellowish green hue. Little did they know that they were using one of the most potentially dangerous substances on Earth.
The destructive power of uranium
In 1895, German physicist Wil-helm Rontgen (1845-1923) noticed that photographic plates held near a Crookes tube—a device for analyzing electromagnetic radiation—became fogged. He dubbed the rays that had caused this x rays. A year later, in 1896, French physicist Henri Becquerel (1852-1908) left some photographic plates in a drawer with a sample of uranium, and discovered that the uranium likewise caused a fogging of the photographic plates. This meant that uranium was radioactive.
With the development of nuclear fission by Hahn and German chemist Fritz Strassman in 1938, uranium suddenly became all-important because of its ability to undergo nuclear fission, accompanied by the release of huge amounts of energy. During World War II, in what was known as the Manhattan Project, a team of scientists in Los Alamos, New Mexico, developed the atomic
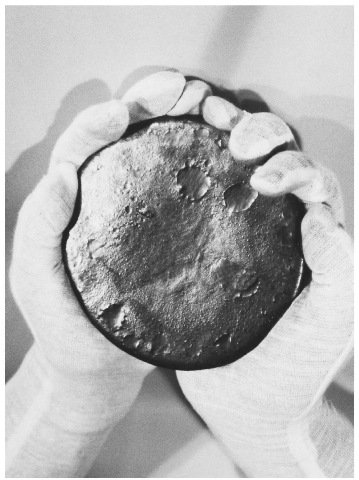
Gloved hands hold a gray lump of uranium. This material has been remolded after having been removed from a Titan II Missile, part of the disarmament after the end of the Cold War.
bomb. The first of the two atomic bombs dropped on Japan in 1945 contained uranium, while the second contained the transuranium element plutonium.
Other uses for uranium
Though nuclear weapons have fortunately not been used against human beings since 1945, uranium has remained an important component of nuclear energy—both in the development of bombs and in the peaceful application of nuclear power. It has other uses as well, due to the fact that it is extremely dense.
Indeed, uranium has a density close to that of gold and platinum, but is much cheaper, because it is more abundant on Earth. In addition, various isotopes of uranium are a by-product of nuclear power, which separates these isotopes from the highly fissionable 235U isotope. Thus, quantities of uranium are available for use in situations where a great deal of mass is required in a small space: in counterweights for aircraft control systems, for instance, or as ballast for missile reentry vehicles.
Because 238U has a very long half-life—4.47 • 109 years, or approximately the age of Earth—it is used to estimate the age of rocks and other geological features. Uranium-238 is the “parent” of a series of “daughter” isotopes that geologists find in uranium ores. Uranium-235 also produces “daughter” isotopes, including isotopes of radium, radon, and other radioactive series. Eventually, uranium isotopes turn into lead, but this can take a very long time: even 235U, which lasts for a much shorter period than 238U, has a half-life of about 700 million years.
The radiation associated with various isotopes of uranium, as well as other radioactive materials, is extremely harmful. It can cause all manner of diseases and birth defects, and is potentially fatal. The tiny amounts of radiation produced by uranium in old pieces of decorative glass is probably not enough to cause any real harm, but the radioactive fallout from Hiroshima resulted in birth defects among the Japanese population during the late 1940s.
Transuranium Elements
Transuranium elements are those elements with atomic numbers higher than that of uranium. None of these occur in nature, except as isotopes that develop in trace amounts in uranium ore. The first such element was neptunium (Np, 93), created in 1940 by American physicist Edwin Mattison McMillan (1907-1991) and American physical chemist Philip Hauge Abelson.
The development of the cyclotron by American physicist Ernest Lawrence (1901-1958) at the University of California at Berkeley in the 1930s made possible the artificial creation of new elements. A cyclotron speeds up protons or ions
(charged atoms) and shoots them at atoms of uranium or other elements with the aim of adding positive charges to the nucleus. In the first two decades after the use of the cyclotron to create neptunium, scientists were able to develop eight more elements, all the way up to mendelevium (Md, 101), named in honor of the man who created the periodic table.
Most of these efforts occurred at Berkeley under the leadership of American nuclear chemist Glenn T. Seaborg (1912-1999) and American physicist Albert Ghiorso. The pace of development in transuranium elements slowed after about 1955, however, primarily due to the need for ever more powerful cyclotrons and ion-accelerating machines. In addition to Berkeley, there are two other centers for studying these high-energy elements: the Joint Institute for Nuclear Research in Dubna, Russia, and the Gesellschaft fur Schwerionen for schung (GSI) in Darmstadt, Germany.
The Transuranium Actinides
Plutonium
Just as neptunium had been named for the next planet beyond Uranus, a second transuranium element, discovered by Seaborg and two colleagues in 1940, was named after Pluto. Among the isotopes of plutonium (Pu, 94) is plutonium-239, one of the few fissionable isotopes other than uranium-233 and uranium-235. For that reason, it was applied in the second bomb dropped on Japan.
In addition to its application in nuclear weapons, plutonium is used in nuclear power reactors, and in thermoelectric generators, which convert the heat energy it releases into electricity. Plutonium is also used as a power source in artificial heart pacemakers. Huge amounts of the element are produced each year as a by-product of nuclear power reactors.
Berkeley discoveries of the 1950s
Because the lanthanide element above it on the periodic table was named europium after Europe, americium (Am, 95) was named after America. Discovered by Seaborg, Ghiorso, and two others in 1944, it was first produced in a nuclear reaction involving plutonium-239. Americium radiation is used in measuring the thickness of glass during production; in addition, the isotope americium-241 is used as an ionization source in smoke detectors, and in portable devices for taking gamma-ray photographs.
Key Terms
Actinides: those elements that fill the 5f orbital. Because actinium—which does not fill the 5f orbital—exhibits characteristics similar to those of the actinides, it is usually considered part of the actinides family. Of the other 14 actinides, usually shown at the bottom of the periodic table, only the first three occur in nature.
Atomic number: The number of protons in the nucleus of an atom. Since this number is different for each element, elements are listed on the periodic table of elements in order of atomic number.
Electron cloud: A term used to describe the pattern formed by orbitals.
Half-life: The length of time it takes a substance to diminish to one-half its initial amount. For a sample of radioisotopes, the half-life is the amount of time it takes for half of the nuclei to become stable isotopes. Half-life can be a few seconds; on the other hand, for uranium-238, it is a matter of several billion years.
Inner transition metals: The lanthanides and actinides, which are unique in that they fill the f orbitals. For this reason, they are usually treated separately.
Isotopes: Atoms that have an equal number of protons, and hence are of the same element, but differ in their number of neutrons. This results in a difference of mass. Isotopes may be either stable or unstable. The unstable type, known as radioisotopes, are radioactive.
Lanthanides: The transition metals that fill the 4f orbital.
Mass number: The sum of protons and neutrons in the atom’s nucleus. The designation 238U, for uranium-238, means that this particular isotope of uranium has a mass number of 238. Since uranium has an atomic number of 92, this means that uranium-238 has 146 neutrons in its nucleus.
Neutron: A subatomic particle that has no electric charge. Neutrons are found at the nucleus of an atom, alongside protons.
Nucleus: The center of an atom, a region where protons and neutrons are located, and around which electrons spin. The plural of “nucleus” is nuclei.
Orbital: A region of probabilities regarding the position of an electron for an atom in a particular energy state. The higher the principal energy level, the more complex the pattern of orbitals.
Principal energy level: A Value indicating the distance that an electron may move away from the nucleus of an atom. This is designated by a whole-number integer, beginning with 1 and moving upward. The higher the principal energy level, the greater the energy in the atom, and the more complex the pattern of orbitals. Elements in the transition metal family have principal energy levels of 4, 5, 6, or 7.
Radiation: In a general sense, radiation can refer to anything that travels in a stream, whether that stream be composed of subatomic particles or electromagnetic waves. In a more specific sense, the term relates to the radiation from radioactive materials, which can be harmful to human beings.
Radioactivity: A term describing a phenomenon whereby certain isotopes, known as radioisotopes, are subject to a form of decay brought about by the emission of high-energy particles. “Decay” does not mean that the isotope “rots”; rather, it decays to form another isotope until eventually (though this may take a long time) it becomes stable.
Radioisotope: An isotope subject to the decay associated with radioactivity. A radioisotope is thus an unstable isotope.
Sublevel: A region within the principal energy level occupied by electrons in an atom. Whatever the number n of the principal energy level, there are n sublevels. Actinides are distinguished by the fact that their valence electrons are in a sublevel corresponding to the 5f orbital.
Transition metals: A group of 40 elements (counting lanthanum and actinium), which are the only elements that fill the d orbital. In addition, the transition metals have their valence electrons on two different principal energy levels. Though the lanthanides and actinides are considered inner transition metals, they are usually considered separately.
Transuranium elements: Elements with an atomic number higher than that of uranium (92). These have all have been produced artificially. The transuranium elements include 11 actinides, as well as 9 transition metals.
Valence electrons: Electrons that occupy the highest principal energy level in an atom. These are the electrons involved in chemical bonding.
Above curium (Cm, 96) on the periodic table is the lanthanide gadolinium, named after Finnish chemist Johan Gadolin (1760-1852). Therefore, the discoverers of Element 96 also decided to name it after a person, Marie Curie. As with some of the other relatively low-number transuranium elements, this one is not entirely artificial: its most stable isotope, some geologists believe, may have been present in rocks many millions of years ago, but these isotopes have long since decayed. Because curium generates great amounts of energy as it decays, it is used for providing compact sources of power in remote locations on Earth and in space vehicles.
When Seaborg, Ghiorso, and others created Element 97, berkelium (Bk), they again took the naming of lanthanides as their cue. Just as terbium, directly above it on the periodic table, had been named for the Swedish town of Ytterby, where so many lanthanides were discovered, they named the new element after the American city where so many transuranium elements had been developed. Berkelium has no known applications outside of research. The Berkeley team likewise named californium (Cf, 98) after the state where Berkeley is located. Researchers today are studying the use of californium radiation for treatment of tumors involved in various forms of cancer.
The remaining transuranium actinides
The remaining transuranium actinides were all named after famous people: einsteinium (Es, 99) for Albert Einstein (1879-1955); fermium (Fm, 100) after Italian-American physicist Enrico Fermi (1901-1954); mendelevium after Mendeleev; nobelium (No,
102) after Swedish inventor and philanthropist Alfred Nobel (1833-1896); and lawrencium (Lr,
103) after Ernest Lawrence.
Both einsteinium and fermium were byproducts of nuclear testing at Bikini Atoll in the south Pacific in 1952. For this reason, their existence was kept a secret for two years. Neither element has a known application. The same is true of mendelevium, produced by Seaborg, Ghiorso, and others with a cyclotron at Berkeley in 1955, as well as the other two transuranium actinides.
Beyond the Transuranium Actinides
As noted earlier, there are nine additional transuranium elements, which properly belong to the transition metals. The first of these is rutherfordium, discovered in 1964 by the Dubna team and named after Ernest Rutherford (18711937), the British physicist who discovered the nucleus. The Dubna team named dubnium, discovered in 1967, after their city, just as berkelium had been named after the Berkeley team’s city. Both groups developed versions of Element 106 in 1974, and both agreed to name it seaborgium after Seaborg, but this resulted in a controversy that was not settled for some time.
The name of bohrium, created at Dubna in 1976, honors Danish physicist Niels Bohr (1885-1962), who developed much of the model of electron energy levels discussed earlier in this essay. Hassium, produced at the GSI in 1984, is named for the German state of Hess. Two years earlier, the GSI team also created the last named element on the periodic table, meitnerium (109), named after Meitner. Beyond meitnerium are three elements, as yet unnamed, created at the GSI in the mid-1990s.
