Introduction
Heart transplantation is the ultimate therapy for patients in the end stage of heart failure in order to either restore or promote patients a better functional performance (Bocchi, 2001; Guimaraes, 1999).
The first heart transplant in the world was performed in 1967 and the procedure has been even more present in this area since then (Kaye, 1993). Several clinical efforts and advances were seen in the last decade in this field, with the incorporation of new surgical techniques, new immunosuppressive drugs, new diagnostic methods and approaches in the early and late postoperative. All advances were able to contribute to greater functional status and less impairment or co-morbidities.
Many different centers around in the world perform heart transplantation and, thus, are able to contribute to favorable results in terms of survival and quality of life on heart transplant recipients (Bocchi, 1999).
The practice of regular physical activity has been recommended for rehabilitation after cardiac transplantation in order to reduce and control co-morbidities (such as hypertension, diabetes, vascular disorders, mainly developed due to continuous use of immunosuppressive drugs), as well as to restore physical capacity and daily activities (Guimaraes, 1999).
Nevertheless, crucial attention is needed when prescribing a physical activity program heading for heart transplant recipients, because they remain with some impairment previously to the surgery, and gain some other disabilities due to the transplantation procedure itself and due to the use of required drugs to control organ rejection.
Prevalence
Heart failure (HF) is the final common pathway of most diseases which affect the heart, and it is performed in these patients because of the severity and urgency of their disease (Reemtsma, 1985). Thus, heart transplant, or a cardiac transplantation, is a surgical transplant procedure performed on these patients and it is recognized as the gold standard therapy for patients facing the end stage of heart failure because there is an increased worsening imbalance between the demand for transplants and the supply of the organs in HF (Haywood, 1996).
The surgical procedure takes a working heart from a recently deceased organ donor (an allograft) and implants it into the patient (called recipient). The patient’s own heart may either be removed (orthotopic transplant) or, less commonly, left in to support the donor heart (heterotopic transplant);
The cardiac transplantation contributes to a low operative mortality but an undesirable high postoperative mortality. This elucidates the fact that heart failure and the surgery represent some important current clinical challenges in healthcare and an epidemic in progress (Guimaraes, 2004).
Whenever there is a careful selection for choosing the donor and recipient, there is also a significant increase in survival, exercise capacity, on return to work and quality of life of this group of patients (Banner, 1989).
From the early 80′s, almost 10% of people died on the waiting list for cardiac transplantation, mostly due to sudden cardiac deaths, and the average length of time patients wait before undergoing transplantation is increasing each decade (Haywood, 1996) maybe because of the increase number of patients suffering from HF and the improvement of its palliative treatment (Guimaraes, 2004).
Heart transplantation has evolved and long term survival is expected for most heart transplant recipient because of the improvements in organ preservation, surgery and immunosuppressive drug management, and physical rehabilitation.
Pathophysiology
In general, heart transplant surgery has become an effective therapeutic option in the management of end stage heart disease because it is able to restore physical function because the new heart doesn’t have the limitations the old heart had.
There is an association of better left ventricular function one year after cardiac transplant operation with donor-related factors (donor age and sex, ischemia time), and with recipient-related factors (recipient age and sex, frequency of acute rejection), type of immunosuppressant drug, and frequency of hypertension (Reid, 1988).
However, the procedure itself promotes complete organ denervation by the postganglionic neural axons section. During heart transplant surgery, the atria of both, the giver and the receiver are sutured together (el-Gamel, 1998). The original sinoatrial node remains innervated, the vagus nerve is severed during the operation, but the electrical impulses generated by it cannot cross over the suture line.
Because the sinoatrial node of the transplanted heart, which determines heart rate (HR) is totally denervated, this neural section may be responsible for either a delay or an unsatisfactory autonomic response (Guimaraes, 1999), which will depend mostly on circulating catecholamines to stimulate the heart (Kaye, 1977) unless nerve regrowth occurs.
Therefore, the absence of afferent neural control of heart rate leaves the heart under the influence of hormones in addition to internal control. The lack of neural signals to the sinoatrial node is indicated by the reduced maximal HR, heart rate variability, as well as changes in the HR response during exercise (Di Rienzo, 2001) and during change of posture (Doering, 1996).
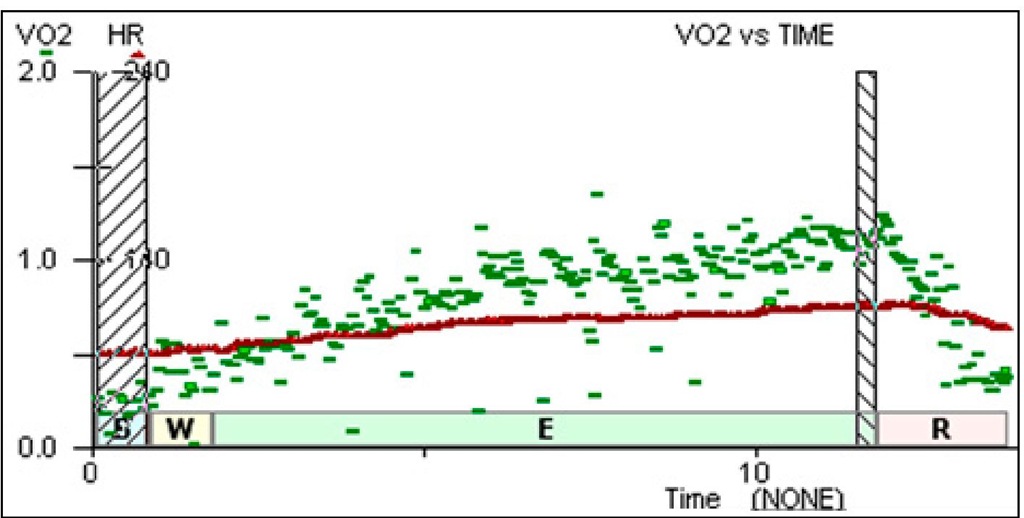
The surgery in these patients promotes inefficient control of heart rate (Kaye, 1977) usually with a high resting HR and a small increase in HR in the first minutes of exercise. Perhaps maximal HR is reached in the recovery period of exercise, and not at peak exercise, taking longer to return to baseline (Guimaraes, 1999), as previous shown in Fig. 1.
However, the vasomotor control (neural and hormonal) is not affected by surgery. The changes in this system may appear due to adaptive mechanisms to drug therapy (Toledo, 2002).
Initially it was thought that the denervation was permanent, and there was only the dependence on circulating catecholamines to achieve maximal heart inotropic and chronotropic stimulation (Kaye, 1977). As time passed by and recent studies have been performed, it has been shown that there is a reinnervation process over time after surgery (Fuentes, 2010), because significant higher VO2, AT (anaerobic threshold), VE (Schwaiblmair, 1999), myocardial function and coronary artery tone are shown after this surgery (Burke, 1995). reinnervation happens in an heterogeneous pattern (Wilson, 1993).
It is known that time after transplantation is recognized as a significant factor correlated to partial autonomic reinnervation among these recipients (Schwaiblmair, 1999). In the majority of recipients with more than one year after cardiac transplantation, cardiac norepinephrine reapers. This support the presence of late heterogeneous sympathetic reinnervation (Wilson, 1993).
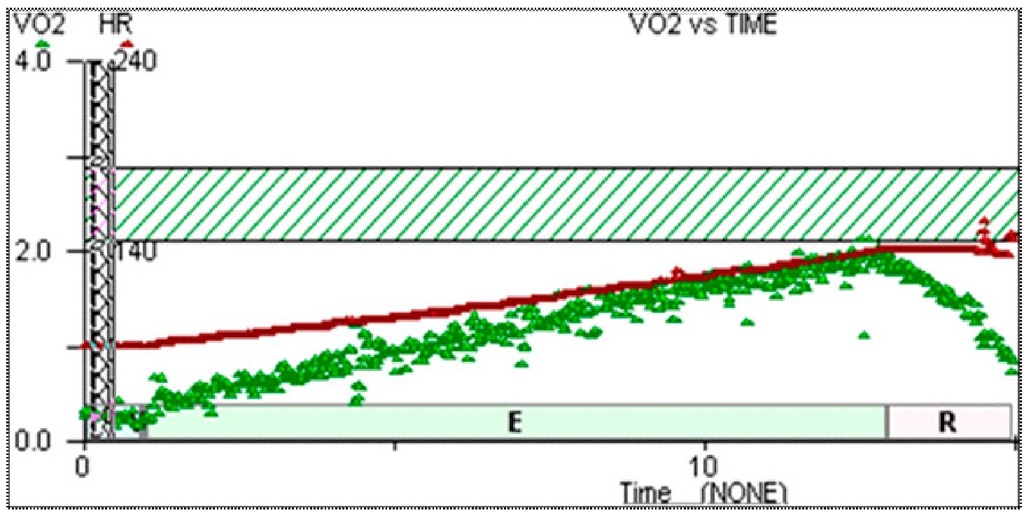
Data from Heart Failure Clinics of Heart Institute compare the O2 consumption increase to the heart rate increase during exercise. It shows that a cardiac transplant recipient at short-term transplantation have the increase of VO2 during cardiopulmonary exercise test not matched to the increase in HR (Figure 1). The same patient at long-term transplantation showed a closer relationship to this increase (Figure 2), so this might corroborate the idea of partial reinnervation described over time.
Fig. 1. Response in recent (5 month) post transplant recipients. HR in red, VO2 in green.
Fig. 2. Response in late (9 year) post transplant recipients. HR in red, VO2 in green.
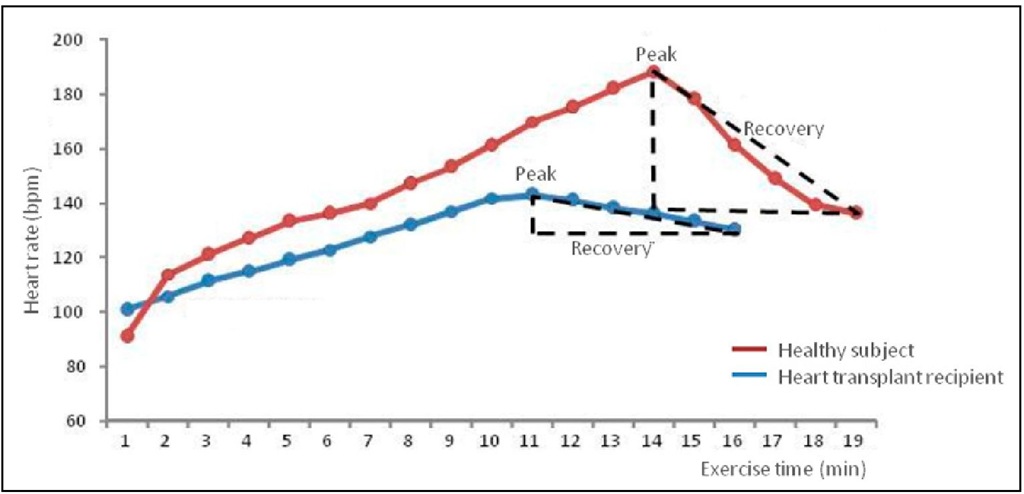
Thus, heart rate control acts different from healthy subjects. It becomes more elevated at rest; nevertheless, during exercise, there is gradual delay of its increase from the exercise start to the peak HR. As a result, by the time heart transplant recipients reach their peak HR, it is, indeed, lower than peak HR in healthy subjects. Then, HR decreases in a slow way during the recovery period (Guimaraes, 1999). This described finding in exercise could be identified in one of the patient referred to Heart Failure Clinics of Heart Institute, in Brazil (Figure. 3).
Fig. 3. Heart rate(HR) representation in a sedentary healthy subject (in red) and in a sedentary heart transplantation recipient (in blue).
Comparing the same data from heart transplant recipients and heart failure patients from Heart Failure Clinics of Heart Institute, both, resting HR and peak HR, after surgery becomes significantly higher compared to the same data from healthy subjects, although there is no change in their HR reserve (Figure 4).
Fig. 4. Resting heart rate, peak heart rate and heart rate reserve in heart failure patients and heart transplant recipients ; HR — heart rate.
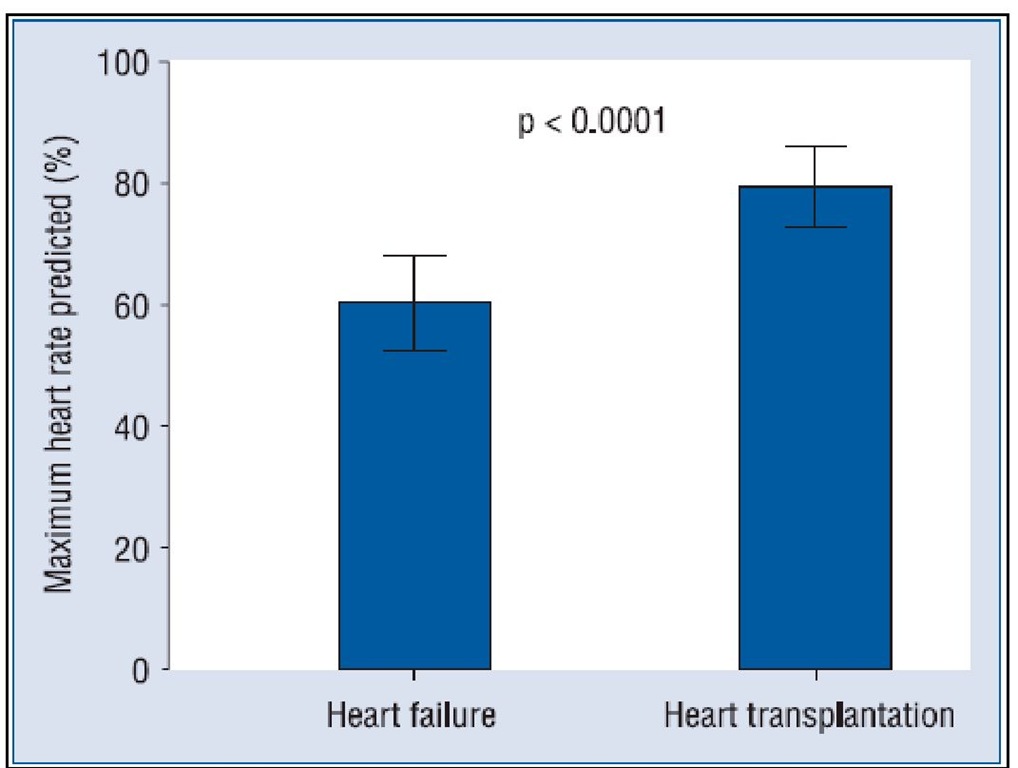
Peak heart rate (percentage of the maximum predicted for age) is significant higher in heart transplant recipients than in heart failure patients (Carvalho, 2009a) because the heart remains partially denervated, as it is shown in Figure 5. Thus, the general use of maximum HR adjusted for age recommended by the Task Force, 2006, known as 220 – age, cannot be applied to transplant recipients in order to identify their maximum effort and to prescribe exercise (Casadei, 1992; Guimaraes, 2010).
Indeed, no patient reached the maximum heart rate predicted for their age during a treadmill cardiopulmonary exercise test. A heart rate increase in heart transplant patients during cardiopulmonary exercise test comes around 80% of the maximum age-adjusted value (Carvalho, 2009a).
Masked mechanisms responsible for the increase of cardiac output in these patients have close correlation to reaching their physical capacity (Casadei, 1992). In fact, heart transplant recipient’s norepinephrine plasma level is lower, at rest, than either healthy or heart failure subjects. But it becomes twice higher than healthy subjects do, after the 6-minute walking test (Guimaraes, 2010).
Increases in heart rate and stroke volume at exercise are first linked to the augmented venous return, and later to the increased plasmatic noradrenaline level. Hence, reduced cardiac output response to exercise results in early anaerobic metabolism, acidosis, hyperventilation and diminished physical capacity.
Peak oxygen consumption is reduced in transplant recipients compared to healthy subjects, which may be related to the existence of remaining systolic and diastolic dysfunction (Patel, 2003), muscular atrophy and hormonal abnormalities due to heart failure persistence after transplantation (Gryglewski, 2001), in addition to the use immunosuppressive drugs that reduce exercise capacity, and sympathetic stimulation (Banner, 1989).
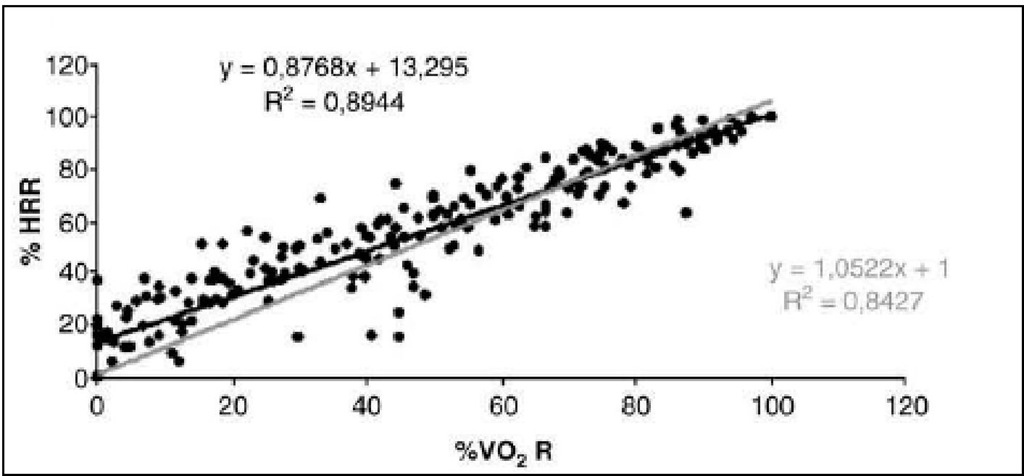
A strong relationship was found between percentage of heart rate reserve (%HRR) and percentage of oxygen consumption reserve (%VO2R) in heart transplant during cardiopulmonary exercise test, as it follows in Figure 6 (r=0.95, p<0.0001). The %HRR-%VO2R linear regression between 0 intercept and 1 intercept between the slope of each patient showed a slope of -0.23, which means that it is far from the perfection expressed by a slope of 1.
Fig. 5. Mean peak heart rate (percentage of the maximum heart rate predicted for age) in heart failure patients and in heart transplantation recipients. Data is presented as the mean ± 95% confidence interval.
Perhaps different terms of heart transplantation and, consequently, different reinnervation status patients could have influenced the imperfect reliability of the %HRR versus %VO2R (Carvalho, 2009b).
Fig. 6. Percentage of heart rate reserve (%HRR) and percentage of oxygen consumption reserve (%VO2R)
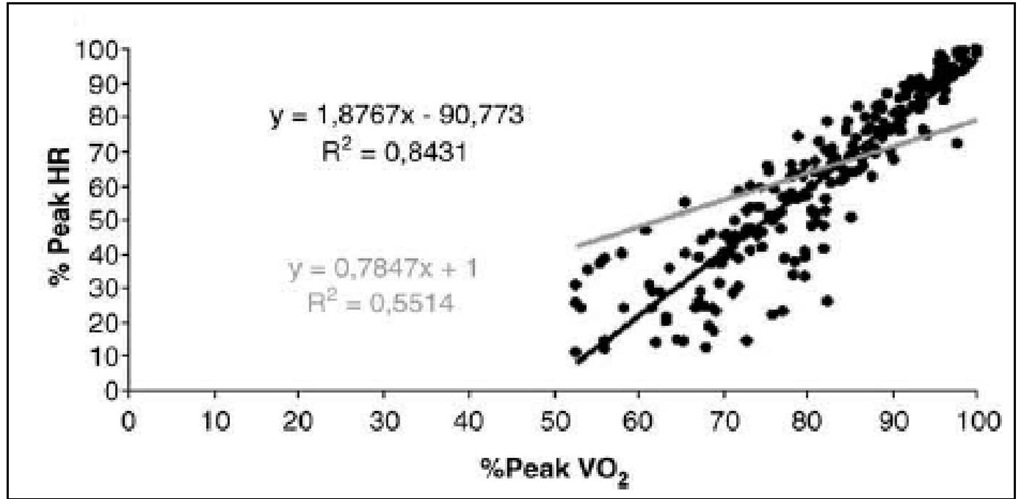
A close relationship was also found between %peak heart rate versus %peakVO2 (r=0.91, p<0.0001) and absolute heart rate versus absolute VO2 (r=0.67, p<0.0001). They are expressed in both, Figure 7 and Figure 8, data extracted from Heart Failure Clinics of Heart Institute (Carvalho, 2009b).
Fig. 7. Heart rate (HR) and peak oxygen consumption (VO2)
Fig. 8. The plot represents stage by stage regression of the cardiopulmonary exercise test. The grey is the identity line and the black line is regression line. Linear regression between 0 intercept and 1 intercept to percentage of heart rate reserve (%HRR) and percentage of oxygen consumption reserve (%VO2R) of each patient.
The increased of muscle work and/or activation of the neurohormonal system, in order to maintain, reduced or increase blood pressure and heart rate as an answer to exercise is mainly responsible for the decrease in physical capacity among heart transplant recipients. This decrease in physical capacity after transplantation leads to insufficient production of nitric oxide (Fischer, 2005) and prostacyclin (Guimaraes, 2007), as well as hyperemia dysfunctional, and activation of other compensatory mechanisms. This occurs mainly when there is increased muscle work and/or activation of the neurohormonal system, in order to perform maintenance, reduced or increased blood pressure and heart rate in response to exercise intensity (Poston, 1999; Carvalho, 2009c).
It has also been reported that physical work capacity in heart transplant recipients decrease typically 40% of age-predicted normal levels (Goodman, 2007).
Finally, the reduction in arterial compliance observed after transplant may be due to changes on endothelial dysfunction or vascular mechanisms. Moreover, greater sympathetic nerve activity can lead to increased smooth muscle tone of arteries and, consequently, increase of the vessels stiffness (Taylor, 2003; Degertekin, 2002).