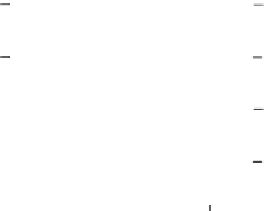Geoscience Reference
In-Depth Information
5.0
plateau
4.48
±
0.03 Ga
7000
4.5
1240
°
1290
°
1340
1380
1450
°
1500
6000
1240
°
1180
°
°
°
1120
°
5000
4.0
1060
°
4000
3.5
(b)
1290
°
3000
1450
°
(a)
950
°
1340
°
2000
1380
°
3.0
1000
890
°
2.5
830
°
0
40
60
80
100
20
5
10
15
0
0
20
39
39
Ar
36
Ar
Ar released (%)
Figure 6.6.
(a) The argon age spectrum (sometimes called an argon-release
diagram) for the Menow meteorite. The ages from the incremental heating as
calculated from Eq. (6.48) are plotted against the percentage of
39
Ar released up to
that step (the width of the lines indicates the error). About 20% of the
39
Ar was
released by the third temperature step which gave an 'age' of 3.3 Ga. However, not
until the temperature reached 1240
◦
C and half the
39
Ar had been released was a
plateau reached. Thereafter the 'age' at
4.5, Ga does not change with increasing
temperature. Such an age spectrum can be most simply visualized as representing
'age' with increasing depth in the mineral. Any metamorphic event may have
caused the mineral to lose argon. The incremental heating essentially samples from
progressively deeper into the mineral. Low temperatures release argon from the
surface of the mineral. These data will have low
40
Ar
∼
39
Ar values (and low 'ages')
due to the previous loss of
40
Ar during metamorphism. Subsequent incremental
steps will yield increasingly higher values of
40
Ar
/
39
Ar (and higher 'ages') as argon
is released from deeper locations that lost less
40
Ar during the metamorphism.
Ultimately, ideally a 'plateau' is reached, which indicates the original time and 'age'
of cooling of the mineral. The high-temperature 'plateau' data points all plot on an
isochron (b) giving an age of 4.48 ± 0.06 Ga. The shape of the age spectrum for this
meteorite implies that it subsequently lost 25% of its argon at about 2.5 Ga. (After
Turner
et al
.(1978).)
/
For chondritic meteorites, the average present-day
147
Sm
/
144
Nd ratio is 0.1967,
and the average present-day
143
Nd
144
Nd ratio is 0.512 638.
11
Therefore, we can
rewrite Eq. (6.50) for such chondrites as
/
143
Nd
144
Nd
143
Nd
144
Nd
147
Sm
144
Nd
(e
λ
t
0
=
now
−
−
1)
now
=
0
.
512 638
−
0
.
1967(e
λ
t
−
1)
(6.51)
where the subscript 'now' represents measurements made now and the subscript
0 refers to values time
t
ago.
11
This isotope ratio is normalized with respect to the reference isotope ratio
146
Nd/
144
Nd
0.7219.
Various laboratories use slightly different values due to varying methods; this is a problem for
isotope geologists using the Sm-Nd method.
=