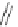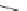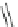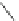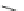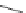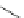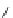Chemistry Reference
In-Depth Information
Alkyl
XR
Alkyl
RX
NiCl
2
(dppp)
Al(Alkyl)
3
,DME
Alkyl
XR
NaXR
31
-
51%
Alkyl
RX
Cl
45
Fe(acac)
3
(Alkyl)MgBr
35
-
68%
Alkyl
48
XR
or CuI, ArOH
1,10-phenanthroline
Cl
X=O,S
R = Alkyl, Aryl
Cl
R
Pd(OAc)
2
,
IPr HCl,
ArZnCl
Cl
R
Ar
46
Cl
Ar
Pd(OAc)
2
IPr HCl
t-
BuO
K
or Pd[P(
t-
Bu)
3
]
2
ArB(OH)
2
,CsF
Ar
or Pd
2
(dba)
3
,IPrHCl
ArB(OH)
2
,Cs
2
CO
3
R
RC
CSnMe
3
R=Ar,SiMe
3
Ar
50
R
49
Ar
i-
Pr
i-
Pr
R
OMe
N
N
O
O
(
i-
Pr)
2
P
Pd
P(
i-
Pr)
2
i-
Pr
i-
Pr
Cl
manisyl
IPr
Eberhard's pincer catalyst
Scheme 13 Synthesis of pentasubstituted corannulene derivatives from pentachlorocorannulene
[
39
,
70
,
83
,
89
-
91
,
93
,
94
]
the reaction of 46 with various sodium alkoxides required high temperature
(180
C) [
83
]. The reaction mechanism should be via a Meisenheimer complex
[
92
] rather than an aryne intermediate. Recently, pentaalkoxycorannulenes [
93
] and
pentaaryloxycorannulenes [
94
] were efficiently achieved by the Cu(I)-catalyzed
Ullmann condensation reaction of 46 with aliphatic alcohols and substituted
phenols, respectively. For the synthesis of pentaalkoxycorannulenes, a large excess
of base (ca. 30 equiv.) was required to complete the substitutions.
Reactions of
sym
-penta(3-butynyl)corannulene 45 (alkyl
3-butynyl) with
azides can generate triazole derivatives under the catalysis of Cu complexes.
These interesting compounds provide potential applications in bioconjugated
chemistry [
95
] and organic materials, which display liquid-crystalline behavior
and organogelation properties [
96
].
Very recently, pentaalkylcorannulene derivatives were used as initiators to
generate corannulene-core polymers by expansion of five pendant groups
(Scheme
14
)[
97
]. The first initiator 45-PrOH was prepared by the Fe-catalyzed
Kumada coupling of 46 with (3-((triisopropylsilyl)oxy)propyl)magnesium bro-
mide, as presented in Scheme
13
, and subsequent desilylation. The second initiator
45-PrE was obtained by the reaction of 45-PrOH with
ʱ
-bromoisobutyryl bromide.
¼