Chemistry Reference
In-Depth Information
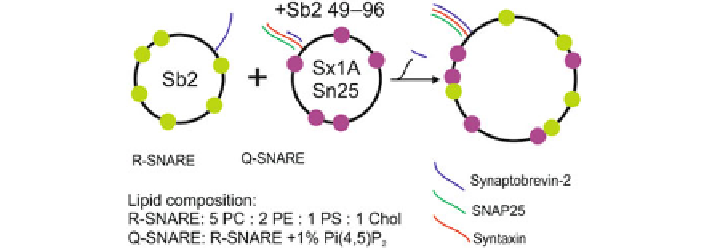
Fig. 3.3
The cartoon sketch above indicates the liposomes conditions used in the experiment.
Large
black circles
indicate the liposomes. Sb2 stands for Synaptobrevin2, Sx1A is Syntaxin 1A
and Sn25 is SNAP25. The
green
and
purple dots
indicate the
green
donor fluorophore and the
red
acceptor fluorophore, of the FRET pair, respectively. A tiny fragment (residues 49-96) of
synaptobrevin-2 is added to the Q-SNAREs to prevent spontaneous fusion. Fusion is then triggered
by cleaving this fragment from the Q-SNAREs by the addition of an appropriate enzyme. The
lipids and their ratio used in the preparation of the liposomes are shown below. PC, PS, and PE are
abbreviations for the lipid head groups.
PC
Phosphatidylcholine,
PS
Phosphatidylserine and
PE
Phosphatidylethanolamine,
Chol
cholesterol
R-SNARE liposomes had a lipid composition of 5:2:1:1 ratio of phosphatidyl-
choline (PC), phosphatidylethanolamine (PE), phosphatidylserine (PS) and choles-
terol (chol) (all lipids from Avanti Polar Lipids) to mimick the composition of
synaptic vesicles. Q-SNARE liposomes mimicking the plasma membrane had the
same composition except that 1mol% PC was replaced with the plasma membrane
lipid
Pi
P
2
. Further, the R-SNARE liposomes were labelled with 1.5mol%
of OG-PE (Invitrogen), the donor fluorophore. 1.5mol% 1,1
-dioctadecyl-3,3,3
,3
-
tetramethylindodicarbocyanine perchlorate (DiD; Invitrogen; emission 670nm) was
chosen as the acceptor fluorophore in the Q-SNARE liposomes.
The synaptobrevin-2
49
−
96
fragment in the Q-SNARE population allows us to
provide a trigger for the fusion process. As we discussed earlier, a coil-coil structure
is formed between the three SNARE proteins which helps the membrane fusion to
occur. However, when only a fragment of the synaptobrevin-2 is added, a full coil
is not formed, thus preventing fusion to occur. To trigger the fusion between the R
and Q-SNARES, an enzyme is added to cleave the synaptobrevin-2
49
−
96
fragment
such that the full synaptobrevin-2 from the R-SNARE can complete the SNARE coil
and fusion proceeds normally. A schematic of this process is shown in Fig.
3.3
.This
schematic is shown in the experimental section together with the data to show the
precise conditions used for that particular experiment.
(
4
,
5
)
3.2.1 Microfluidic Assay for Liposome Fusion
Typically in vitro experiments for fusion studies are performed in a cuvette, with
sample volumes in the range of a few milliliters. While a small sample size is crucial
for experiments involving purified synaptic vesicles, a further disadvantage of the