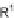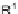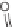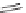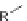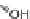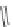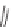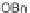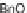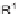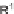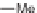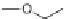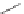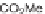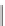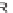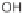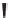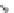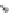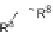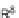Biomedical Engineering Reference
In-Depth Information
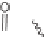
SCHEME 1.2
Diversity-oriented synthesis of 242 compounds based around 18 discrete
molecular frameworks by Thomas et al. Conditions: (a) LiBr, 1,8-diazabicyclo[5.4.0]undec-7-
ene, R
1
CHO, MeCN; (b); AD-mix, (DHQD)PHAL, THF/H
2
O (1:1); c) (
R
)-QUINAP, AgOAc,
i
-Pr
2
NEt, THF,
25
◦
C; (d) chiral bis(oxazoline), Cu(OTf)
2
,3
˚
AMS,CH
2
Cl
2
,C
5
H
6
;
(e) R
2
COCl, DMAP, pyridine, CH
2
Cl
2
; (f) R
3
CHO, BH
3
.
pyridine, MeOH; (g) SOCl
2
, pyridine,
CH
2
Cl
2
,40
◦
C; (h) R
4
Br, Ag
2
O, CH
2
Cl
2
,40
◦
C; (i) R
5
C(O)R
5
, TsOH, DMF, 65
◦
C; (j) R
6
CHO,
TsOH, DMF, 65
◦
C; (k) NaN
3
, DMF, 100
◦
C then DMAD, PhMe, 65
◦
C; (l)
m
CPBA, CH
2
Cl
2
then MeOH, 65
◦
C; (m) CH
2
=
−
78
◦
C
→
CH
2
; (n) OsO
4
,
NMO, CH
3
C(O)CH
3
/H
2
O (10 : 1); (o) RNH
2
,Me
2
AlCl, PhMe, 120
◦
C, then NaH, R
11
X, DMF,
THF then PhMe, 120
◦
C, Grubbs II, CH
2
=
CH
2
; (p) NaIO
4
, THF/H
2
O (1 : 1) then R
7
NH
2
,
NaBH(OAc)
3
,CH
2
Cl
2
; (q) NaIO
4
, THF/H
2
O (1:1) then R
8
NHR
8
, NaBH(OAc)
3
,CH
2
Cl
2
; (r)
R
9
CHO, DMF, TsOH, 60
◦
C; (s) R
10
C(O)R
10
, DMF, TsOH, 60
◦
C. (From [47], with permission
of John Wiley & Sons; copyright (
C
CHCO
2
Bn, PhMe, 120
◦
C, Grubbs II, CH
2
=
2008 John Wiley & Sons.)