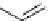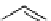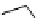Chemistry Reference
In-Depth Information
recently, a more practical method for N-13 carbamate labelling was developed via the
in situ
preparation of chloroformate
analogues from stable precursors [155]. A mixture of
p
-nitrophenol and commercial triphosgene was employed for the prepa-
ration of
p
-nitrophenyl chloroformate in a one-pot process that was found to be more practical. Careful control of the quantities
of triphosgene and appropriate base, usually
i
i-PrNEt, was found to be important to achieve high rCy (Table 4.2).
This strategy was used to prepare a range of urea analogues where isocyanate intermediates were formed from amines and
triphosgene. This method has recently been applied to the synthesis of N-13 labelled carbamazepine [155] and thalidomide
[156] (scheme 4.29).
4.3.5
other
13
n-labelling reactions
4.3.5.1 Preparation and Reaction of [
13
N]nitrogen Dioxide, [
13
N]nitrous Acid, and [
13
N]nitrite
Although much less
popular than
13
NH
3
, the precursors
13
NO
2
,
13
NO
2
-
, and [
13
N]HNO
2
have been used for N-13 labelling reactions. The first
reported preparation of
13
NO
2
was via the oxidation of [
13
N]ammonia using gallium and cobalt oxides (scheme 4.30) [157].
tabLe 4.2
one-pot radiosynthesis of
13
n-labelled urea and carbamate analogues.
1. triphosgene,
i
-Pr
2
NEt (50 μmol)
75°C, 30 min
2.
13
NH
3
, 75°C, 3 min
O
R
H
*
R
NH
2
Entry
rH
Triphosgene (µmol)
radiochemical yields (%)
a
Based on
13
NH
3
NH
2
1
3.3
0-70 (
n
= 5)
2
2.5
78
NH
2
3
2.5
85
N
4
3.3
0-36 (
n
= 5)
5
2.5
52
H
N
6
2.5
34
OH
7
3.3
0-62 (
n
= 5)
8
2.5
84
O
2
N
9
2.5
90
OH
a
radiochemical yield was determined by analytical HPLC. All results are the mean (
n
= 3) with a maximum range of ±10%. radioactive products were
identified using authentic non-radioactive samples.
H
2
*
O
O
N
N
O
NH
*
O
O
[
13
N] thalidomide
[
13
N] carbamazepine
scheme 4.29
Examples of N-13 labelled compounds.