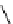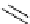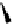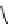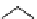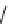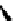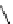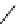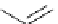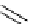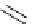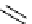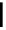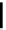Chemistry Reference
In-Depth Information
Table 4.10
Guanidine mediated asymmetric silylation of cyclic secondary alcohols
guanidine
(1 equiv)
+
Silyl-Cl
(1 equiv)
n
n
DCM, rt
OH
OSilyl
(2 equiv)
Run
n
Silyl
Guanidine
Time (d)
Yield (%)
ee (%)
TBDMS
)
1
1
Me
R = H
11
34
37
R = Me
2
1
TBDMS
9
50
39
R = H
3
1
TIPS
6
36
59
N
Ph
H
NN
R
Ph
Ph
Ph
N
TBDMS
)
(
10
H
4
1
78
31
NN
5
1
TIPS
6
79
58
6
2
TIPS
6
15
70
Ph
Ph
Table 4.11
Effect of base in the S
N
Ar reaction of naphthol and fluoronaphthalene
CHO
CHO
HO
CHO
O
CHO
+
base
OMe
F
OMe
Run
Conditions
Yield (%)
NaH, DMSO, 25
C
1
49
K
2
CO
3
, AcNMe
2
, 150
C
2
54
DBU, MeCN, 70
C
3
46
),MeCN, 70
C
4
TMG(
1
59
), MeCN, 70
C
5
BTMG (
2
85