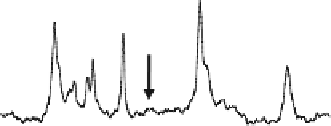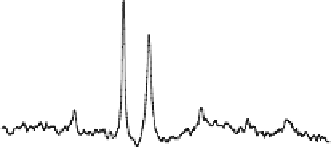Biomedical Engineering Reference
In-Depth Information
PCr
(C)
γ
-ATP
Pi
*
(B)
*
PCr
(A)
γ
-ATP
Pi
8
4
0
-4
-8
-12
-16
-20
Chemical shift (ppm)
Fig. 15.11. In vivo
31
P spectra acquired from a health human occipital lobe in the
absence (
A
) and presence (
B
) of complete γ-ATP saturation, and the difference spec-
trum (
C
) between the two. Only the Pi and PCr resonance peaks show the magnetiza-
tion transfer effect due to the saturation. The intensity reduction of Pi can be used to
determine the forward rate constant and flux for the ATPase reaction, and the intensity
reduction of PCr can be used to determine the forward rate constant and flux for the CK
reaction. Adapted from Lei et al. of Ref.
(20)
.
both Pi and PCr.
Figure 15.11C
shows the difference spectrum
by subtracting the control and
-ATP saturated spectra
(20, 50)
.
The relative Pi signal reduction can be used to calculate the for-
ward rate constant and flux for the ATP
ase
reaction according
to
Eqs. (15.13d)
and
(15.13e)
. Similarly, the relative PCr sig-
nal reduction can be used to calculate the forward rate constant
and flux for the CK reaction according to
Eqs. (15.13c)
and
(15.13e)
.
γ
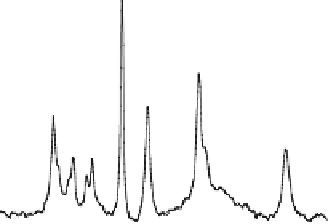
Figure 15.12
illustrates one example of in vivo
31
PMSSMT
measurements in the human occipital lobe
(50)
. A total of four
in vivo
31
P spectra were collected in the absence (
Fig. 15.12A
)
and presence of complete RF saturation on the resonance peak
of Pi (Step 2;
Fig. 15.12B
), PCr (Step 3;
Fig. 15.12C
)
and
4.2.3. In vivo
31
PMSS
MT Measurements for
Determining all ATP
Metabolic Fluxes Involving
PCr
Pi
Exchange in Human
Occipital Lobe
↔
ATP
↔
-ATP (Step 1;
Fig. 15.12D
), respectively. It reveals that
single-site saturation on one phosphate spin can lead to signif-
icant magnetization reductions in the other two coupled phos-
phate spins through the three-spin chemical exchange system
γ