Agriculture Reference
In-Depth Information
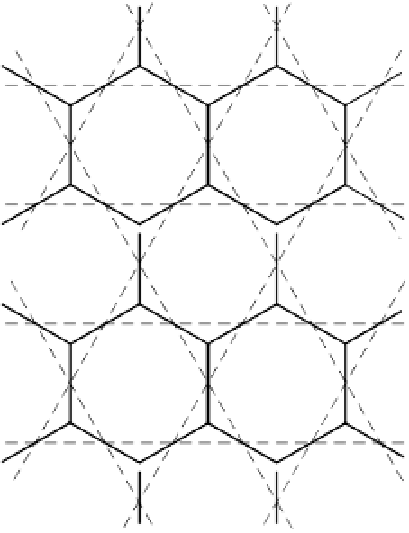
A silica sheet in plan view showing a
pattern of hexagonal holes (White
1997). Reproduced with permission of
Figure 2.5
(Si
4
O
10
)
4-
n
Blackwell Science Ltd.
out of three basal O atoms to form a single extended chain. In the
amphiboles
,
two parallel pyroxene chains are linked by the sharing of an O atom in every al-
ternate tetrahedron. Cations such as Mg
2
, Ca
2
, Al
3
, and Fe
2
are bonded to
the O atoms to neutralize the surplus negative charge. These minerals are collec-
tively called
ferromagnesian
minerals and are found in basic rocks such as basalt,
gabbro, and serpentinite.
A one-dimensional chain extended in two dimensions forms a
silica sheet
, con-
sisting of tetrahedra linked by the sharing of all the basal O atoms (fig. 2.5). The
apical O atoms (superimposed on the Si atoms, as seen in fig. 2.5) form bonds
with metal cations in adjacent sheets by, for example, displacing OH groups from
their positions around a trivalent Al
3
ion, as in the unit Al(OH)
3
. When Al
octahedral units (shown in fig. 2.4b) polymerize by sharing basal OH groups, they
form an
alumina
sheet. Such sheets are the basis of the mineral
gibbsite
[Al
2
OH
6
]
n
,
in which only two-thirds of the available cation positions are occupied by Al atoms.
If Mg
2
is present instead of Al
3
, all the available octahedral positions are filled
and the mineral formed is
brucite
[Mg
3
(OH)
6
]
n
. When two silica sheets sandwich
one alumina sheet, the result is a covalently bonded 2:1 layer-lattice mineral, char-
acteristic of the
micas
(
muscovite
and
biotite
). When mica-type layers sandwich a
brucite layer, the resultant mineral is
chlorite
.
These are examples of
phyllosilicate
minerals, common in rocks and in the
clay fraction of soils (section 2.2.4). Biotite and chlorite are typical minerals of
basic rocks. Some characteristics of phyllosilicate (or layer-lattice) structures are
given in box 2.4.
Three-dimensional Structures
The most important silicate minerals of this type are silica and the feldspars. Sil-
ica minerals consist of polymerized Si tetrahedra of general composition (SiO
2
)
n
.
Silica occurs as the residual primary mineral
quartz
, which is very inert, and as a
2.2.3.3