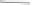Chemistry Reference
In-Depth Information
Norbornadiene would then bind to an equatorial position, probably
trans
to the oxygen,
and in the less-hindered
endo
-mode (Scheme 5.50).
CO
CO
H
H
Co
Co(CO)
3
Co
Co(CO)
3
H
H
H
H
O
N
N
R
R
R
O
O
Ph
N
O
O
Ph
Ph
O
O
Endo
Scheme 5.50
5.3.5 Chiral 2-Alkynoates
In an extensive study of the intermolecular PKRs of 2-alkynoic esters derived from chi-
ral alcohols (including
trans
-2-phenylcyclohexanol, menthol, 8-phenylmenthol and the
camphor-derived alcohols
4a
,
4b
and
9a
, among others), Moyano, Pericas, and co-
workers found that under standard thermal conditions (toluene, 40-70
◦
C), the expected
exo
-cycloadducts were obtained with good yields and with excellent regioselectivity, but
with low diastereoselectivity (Schemes 5.51 and 5.52).
51
O
Co
2
(CO)
8,
rt;
O
R
R
OR*
*RO
R = Me, Ph
O
Toluene,
40-70 °C,
1-38 h
60-97%, up to 3.7:1 dr
Scheme 5.51
O
O
O
Co
2
(CO)
8,
rt;
Me
Me
H
Me
Ph
Ph
O
Me
O
Me
Me
26%, 1.3:1 dr
To l u e n e ,
50 °C,
15 h
Scheme 5.52
Much better results were subsequently obtained with chiral
N
-(2-alkynoyl)
oxazolidinones.
24, 52
These compounds, that are readily prepared in a one-pot procedure