Relationship Between Pathological Change, PrP Accumulation, and Disease
Clinical scrapie has been described in sheep and beige mice in the absence of vacuolar changes (8). Similarly, in the Chediak Higashi strain of mink, transmissible mink encephalopathy infection will cause clinical disease, but does not result in vacuolation (45). For these and other reasons, vacuolation does not appear to correlate with clinical disease (46). Fibrillar PrP, as described in Subheading 3., will damage neuronal tissue and induce gliosis, but not all TSEs result in electron microscopically visible amyloid deposition. However, not all TSEs have amyloid plaques, and the patterns of plaque formation in most of those that do develop plaques is neither sufficiently extensive nor intense to cause neurological deficiency. For example, cerebrovascular amyloid formation in sheep is inconsistently found, and only infrequent plaques are found within individual brains. In BSE, clinical signs include loss of rumi-nal motility and bradycardia (47). These clinical deficits are probably caused by abnormalities of the vagal nucleus, solitary tract, and nucleus ambiguus, sites which, in BSE, are the first and most severely affected nuclei for vacuolar changes and PrP accumulation (12,13). Ultrastructural examination of these neuroanatomical nuclei fails to show any amyloid fibril formation or aggregated forms of PrP, and in only some cases is there gliosis (personal observations). Several studies have shown that apoptotic neuronal loss is a feature of scrapie (48), which may occur early, during the incubation period (49). To determine whether PrP accumulation is directly related to clinical disease we have counted neurons in several neuroanatomical sites of BSE-affected brains (50,51). Although neuronal loss did occur in some sites, such as the vestibular neurons, and may therefore be involved in balance deficiencies, neuronal loss is not a feature of the N. vagus or N. ambiguus, and cannot therefore be implicated in all clinical deficits (51).
In a further model of murine scrapie, we counted dendrites and synapses in the hippocampus of ME7-infected murine scrapie brains, in a model in which there is profound loss of pyramidal neurons within the CA1 sector (52). These studies revealed that loss of synapses and axon terminal degeneration occurred at approx 34-39% of the incubation period (%IP), but neuronal loss did not occur until 72%IP. The nature of the axon terminal degeneration was similar to that seen in excitatory amino acid intoxications. Preamyloid PrP within the extracellular space may perturb synaptic transmission in some sensitive neu-roanatomical regions. These results suggest that neuronal loss may occur following deafferentation of neurons, as a consequence of synaptic loss subsequent to PrP accumulation within the extracellular space. Some studies of PrPsen localization have suggested that PrPsen may be located in the presynaptic membrane (53). These results would be consistent with the above observations, but the ultrastructural localization demonstrated showed only weak and nonspecific labeling, which therefore needs further confirmation. That significant synaptic loss precedes neuronal loss may indicate that synaptic loss is the principal cause of neurological deficits. The axon terminal degeneration described in the ME7 hippocampus is also found at other sites where there is widespread preamyloid PrP accumulation, both at other sites of the ME7-infected brain and in other murine models and in the thalamus of FSE-affected cats. However, we have been unable to find this lesion in BSE-affected brainstems. Although synaptic counting studies have not yet been widely performed in TSEs, it seems unlikely that synaptic loss can account for all the clinical deficits. Therefore, aggregated or fibrillar PrP, neuronal loss, synaptic loss, and possibly vacuolation and gliosis have some significance regarding the development of clinical disease, but that there may be yet other factors that are involved, and possibly may be of major significance in neurological dysfunction.
In the ME7 murine model described above, axon terminal loss is present (34%IP) at up to 80 d before neuronal loss (72%IP) and 140 d before terminal disease. Although PrP accumulation can be detected by immunohistochemis-try in the stratum radiatum at 42%IP, at this stage, the accumulation is slight, and it is by no means clear that the amount of PrP accumulation is sufficient to cause pathological change. PrP res can be detected by Western blotting at about 80 dpi (32%IP) in whole brains of scrapie-infected mice, but disease-specific accumulations of PrP of unknown protease resistance can be detected in both hippocampus and thalamus from 60 dpi (24%IP) by immunocytochemistry (44). Although questions remain about the relative sensitivity of these techniques, these findings open the possibility that PrP is first released from cells as PrPsen, which only later acquires protease resistance. A human recombinant PrP, spanning residues 91-231, has recently been shown to undergo a reversible conformational change between a protease-sensitive soluble a-helical form and a protease-resistant fibrillogenic p-pleated sheet form, depending on the ionic strength of the solution (54).
Whatever the relative sensitivities of Western blotting and immunohis-tochemistry, infectivity levels considerably precede increased PrP accumulation. In ME7-infected CVF1 mice, significant levels of infectivity are present from at least 20 d before the first detection for PrP by immunocytochemistry and the infectivity curves and subjectively assessed levels of PrP accumulation are different, suggesting that infectivity and PrP accumulation may not be directly linked. Similar dissociations between infectivity and PrPres accumulation have been recorded by others working with different rodent models of disease (55,56).
PrPres Processing in Cultured Cells and In Vivo Observations
PrPsen is attached to the exterior of the cell plasma membrane by a glycosyl phosphadidyl inositol (GPI) anchor. Studies of scrapie-infected cell culture lines have shown that PrPres, in contrast to PrPsen, cannot be released from the cell surface on treatment by a bacterial phosphadidyl inositol-specific phos-pholipase that specifically cleaves the GPI anchor (57,58,59). The visualized location of disease-specific PrP accumulations seen in the electron microscope appears to be in conflict with the in vitro studies. If PrPres remains bound to the cell surface, it is difficult to explain the apparent diffusion of disease-specific PrP away from scrapie-infected cells through the extracellular space. It is also difficult to explain why PrPres is released from the surface of infected ependymal cells into the ventricular space to form fibrils, unless, that is, the initial PrP released by scrapie-infected cells is in the form of PrPsen.
The occurrence and distribution of clathrin-coated pits provides a possible discrepancy between in vitro observations of brain and spleen and studies of cell culture systems. Endocytosis of a number of cell surface receptors (e.g., transferrin) takes places via clathrin-coated pits. These receptors are transmem-brane structures with a cytoplasmic domain necessary for the assemblage of clathrin lattices on the cytoplasmic face of the plasma membrane. In cultured scrapie-infected cells, it has been suggested that PrPC is endocytosed via clathrin-coated pits (59,60). However, the PrPC molecule does not appear to have a cytoplasmic component with which to bind clathrin. Our subcellular studies of scrapie-infected cells do not show increased clathrin-coated pit activity in association with early PrP release (?PrPsen) around neurons or follicular dendritic cells. However, markedly increased clathrin-coated pit activity is seen in association with aggregated and fibrillar forms of PrP (PrPres). These findings suggest that aggregated PrPres binds to a transmembrane receptor, which then elicits coated-pit activity.
Features of Amyloidosis
Amyloid is defined as an extracellular glycoprotein complex that is deposited in tissue. Amyloid fibrils are 7.5-10 nm-diameter, rigid, nonbranching, hollow-cored tubules of indeterminate length. When examined by X-ray diffraction, these fibrils have a characteristic P-pleated sheet configuration. This macromolecular helix of 100 nm periodicity, formed from two P-pleated sheet micelles, is responsible for the resistance of amyloid fibrils to solubilization and to proteolytic digestion. Amyloid fibrils are derived from at least 15 different proteins (amyloid precursor proteins), which may be normal or abnormal variant proteins. In most amyloid disease, cell-free production of amyloid fibrils from precursor proteins has been found.
P component is a doughnut glycoprotein present in amyloid. The presence of P component in PrP amyloid is unknown. All classical amyloids contain a matrix proteoglycan. In amyloid-associated lesions of human brain, heparan sulphate co-localized with amyloid (61,62). PrP-associated deposits also contain heparan sulphates (42,63).
Several groups have previously noted that the properties and distribution of PrPres are typical of classical amyloidoses and show features similar to the accumulation of bA4 Protein of Alzheimer’s disease (7,38,64,65). As observed or inferred from our electron microscopy studies, the subcellular localization and onset of protease resistance of PrP is also similar to that observed or predicted for other amyloid proteins. That is, in common with other amyloid proteins, PrP is released as a preamyloid protein, probably as a protease-sensitive molecule, which aggregates, forms P-pleated sheets, binds sulphated proteoglycans, acquires partial protease resistance, and damages tissue, as aggregated isoforms or as amyloid. The N-terminal truncation of PrP does not appear to occur in vivo in the nongenetic forms of TSEs but may do so in human genetic TSEs, suggesting that there may be different amyloid proteins in different diseases. These findings therefore support the suggestion that the transition between PrPsen and PrPres occurs within the extracellular space after release of the normal protein by scrapie-infected cells. However, there is some evidence of strain dependent variation in processing PrP within individual cells and differences in glycoform patterns of PrPres, are found following infections with different strains of agent infection. The different glycoform patterns may be because of infection of different subpopulations of neurons, which express different proportions of the three glycoforms of PrP, or by altered metabolism of PrP in scrapie-infected cells.
If the different glycoforms of PrPres are secondary to infection-induced abnormal accumulation or polymerization of a normal protein, then the nature of the infections of the TSEs remains unexplained. There is also some evidence to suggest that there are some disease models in which PrP accumulation is either not present, or is present at low levels. Many previous studies have searched for and failed to find conventional micro-organisms in tissues of TSE-affected animals and human beings. And, indeed, most previous reports of viral-like organisms have now been shown to be artifacts or other pathological changes associated with TSE infection. There remains, however, one unexplained structure, which may be of significance.
Tubulovesicular Particles
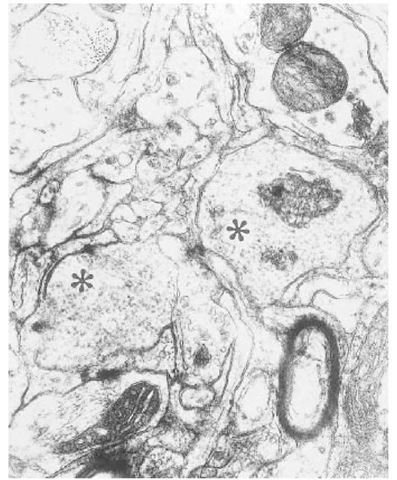
So-called tubulovesicular particles or TVBs, perhaps better named "scrapie-associated particles," because they are not always visualized in tubular form, were first described in the brains of scrapie-infected mice by David-Ferreira et al. (66). TVBs have ever since been regularly, if inconsistently, described in electron microscopic studies of the brains of various experimental and naturally TSEs (67). TVBs have been described in most rodent models of scrapie, in natural sheep scrapie, CJD, GSS, and in BSE (67). We have also seen such particles in feline spongiform encephalopathy (Fig. 6), but these particles have not been reported in spleens or in infected brain cell cultures.
The molecular structure of TVB is unknown, but staining of thin sections by ruthenium red enhances their appearance, suggesting that they contain glyco-syl residues (68). Murine brain tissue, infected with ME7, 87V, or 22CH scrapie isolates, and stained by immunogold methods, were negative for PrP epitopes, using two different anti-PrP sera (17,69). Descriptions of the size and shape of the particles differ, probably because of differences in postmortem delay and different fixation methods. Most reports describe the particles as spherical, with a diameter of between 30 and 35 nm (which is significantly more than the smallest circoviruses, at around 17 nm). The particles are pleomorphic, with some appearing as short rods or ellipsoids. They can be found in axon terminals, but are seen mostly in dendrites.
We have determined the number and density of TVBs present at selected stages of the incubation period in the F1 cross of C57 and VM mice infected with ME7 scrapie, and related their occurrence to the temporal onset of various of pathological changes. (52). As described above, in this model, the earliest changes seen, at about 100 dpi, are a degeneration of axon terminals and synaptic loss.
Fig. 6. The thalamus from a naturally occurring case of feline spongiform encepha-lopathy is shown. Two dendrites (asterisks) contain large numbers of spherical or elliptical structures: so called tubulovesicular bodies. The size of these bodies can be compared to synaptic vesicles in an adjacent axon terminal (star) or cross-sectioned microtubules (arrowheads). Stained with uranyl acetate and lead citrate.
Terminal disease is around 250 dpi. In blind coded trials, the number of tubulovesicular particles were counted and their density was estimated in serial pairs of electron micrographs taken from the stratum radiatum at 84, 100, 126, 154, and 181 dpi, and from four normal brain-inoculated control mice. Tubulovesicular particles were present from 98 dpi, and the density of particles increased with increasing incubation period. The early occurrence of tubulovesicular particles, before the presence of significant pathology, argues that tubulovesicular particles are a part of the primary disease, and are not epiphenomena (70).
The data currently available, therefore, indicate that TVBs are specific to the TSE, and, although they are not composed of PrP, have been found in all forms and animal models of TSE so far examined. They are present at or before the earliest stages of pathology, increase in correlation with infectious titer, and share some of the structural features of viruses. However, they have not yet been found in scrapie-infected cell culture lines or in spleens, which is an absolute requirement, if they are to be considered as a possible infectious cause of the TSEs.
Conclusions
Our studies of several experimental murine disease models of scrapie and naturally occurring animal diseases show that, despite variation in the light microscopic appearance of the forms of PrP accumulation and the neuroana-tomical location of cell type at which it occurs, there is a consistent sequence of events, consisting of plasmalemmal PrP release and extracellular PrP accumulation, followed by aggregation and fibrillization. Some of the pathology of the TSEs, such as gliosis and neuronal apoptosis, is associated with these aggregated forms of PrP, but this is insufficient to explain all the pathology of the TSE or clinical disease. In particular, clinical signs relating to specific neu-roanatomical areas of the BSE and sheep scrapie brain can be identified, in which there is neither fibrillar PrP nor any subcellular features of pathology so far identified. The TEM features associated with abnormal PrP accumulation are closely similar to, or indistinguishable from, amyloid biology. Neither abnormal PrP accumulation nor vacuolation are sufficient to explain all aspects of the pathology of TSE, and factors other than abnormal PrP accumulation may be necessary to explain infectivity. TVB have been found ultrastructurally in many TSEs; they are not directly associated with PrP accumulation or amyloid pathology. The cause and functions of TVB are unknown, but a central role in pathogenesis cannot be discounted.