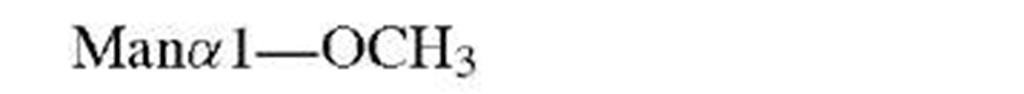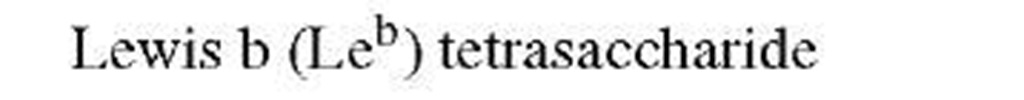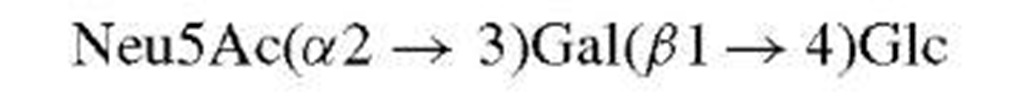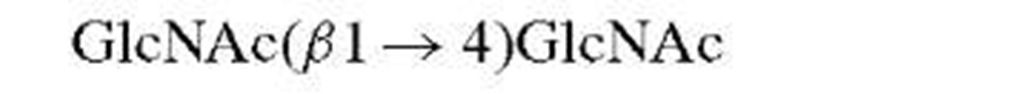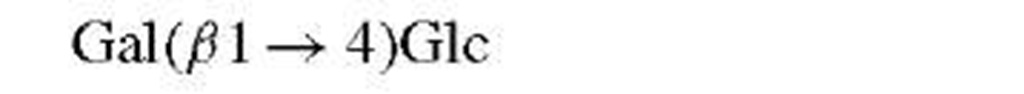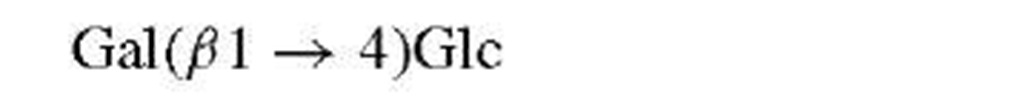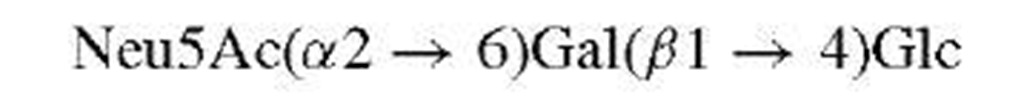Classical methods for the determination of saccharide structures are rarely employed at this time. Thus, methyla-tion and periodate analyses, once the mainstay of carbohydrate work, have been superseded by mass spectrometry and nuclear magnetic resonance (NMR).
Consider the typical problem—that of defining the complete structure of an oligosaccharide (polysaccharide), free or combined. Unlike procedures for analysis of DNA or proteins where automated methods provide rapid and unambiguous sequences, sugar sequences are not readily determined. Needed are linkage positions, configurations, and branching as well as identification of the specific sugars. In some cases, the use of specific enzymes can provide information about the linkage configuration but these do not always work, rarely are quantitative, and often do not discriminate between linkages. The advent of NMR has permitted complete analysis of oligosaccharides as long as sufficient material is available (or spectrometer time). Thus, the linkage configuration for pyranosides is readily apparent from the coupling constant between the protons on carbons-1 and -2 of the sugar in question. The order of sugars and possible branching can be determined directly using mass spectrometric methods. In this case, however, direct identification of a specific hexose may not be possible since all will have the same molecular weight. Therefore, such studies are generally combined with compositional analysis (acid-catalyzed hydrolysis followed by chromatographic separation) and prior knowledge of the type of saccharide involved.
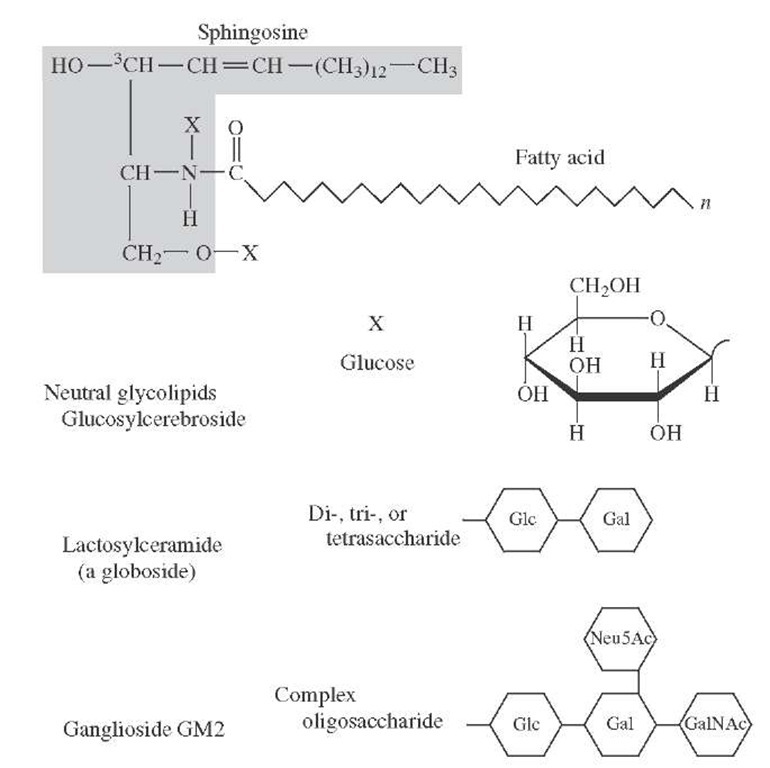
FIGURE 30 Ceramide and glycosphingolipid structures.
TABLE I Representative Lectins and Their Ligands*
Some structural data may also be obtained by the use of specific lectins (Table 1). These are a class of carbohydrate-binding proteins, generally of plant origin, that show high specificity for one or another saccharide. This may include linkage configuration and recognition of oligosaccharide.
Information desired for polymeric structures beyond that of components and linkages will generally include molecular weight and solution conformation. Since polysaccharides (including chains found in proteogly-cans) are polydisperse, the normal definition of molecular weight (as that of a single chemical entity) does not apply. Ideally, one should determine the mole fraction of each species present as a function of degree of polymerization. Currently, there are no methods for achieving this goal. Rather, physical measurements are made that provide number average (osmotic pressure, suitable only for relatively low molecular weights and limited by solubility), weight average (sedimentation or light scattering), and viscosity average (a mixed function that is also a measure of solution conformation—a measurement of mean end-to-end distance). Solution structures are often helical for repeating linear polysaccharides and heavily dependent on solvent (ionic strength) for polyelectrolytes.