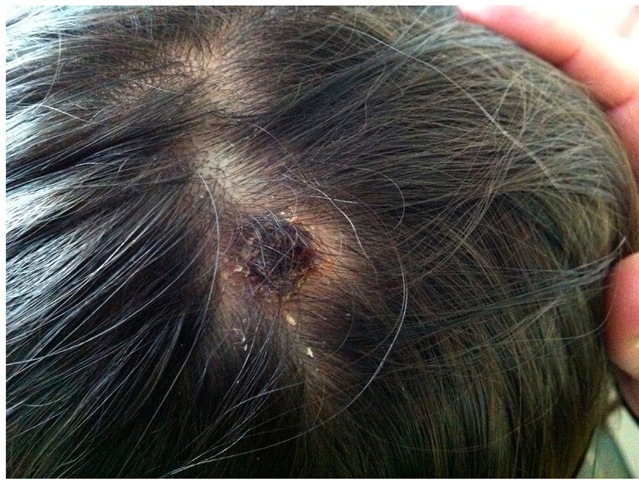
Eschar and lymphadenopathy
This clinical syndrome has been recently reported in Europe, where it is named TIBOLA (TIck-BOrne LimphAdenopaty) or DEBONEL (DErmacentor-BOrne Necrosis-Erythema-Lymphadenopathy). R. slovaca, R. rioja and R. raoultii are the etiological agents, and Dermacentor marginatus is the main vector. This tick species is distributed all over Europe as well as in the North of Africa. Since this rickettsiosis appears in the coldest months of the year, the risk of acquisition for the travellers is lower than for the rickettsioses that are prevalent in spring and summer. In most cases (>90%) the tick-bite is located on the scalp (head) and always in the upper site of the body. After 1-15 days (mean: 4.8 days) of incubation period, the characteristic skin lesion starts as a crusted lesion at the site of the tick-bite (frequently on the scalp). A honey-like discharge from the lesion is observed in some cases. Few days later, a necrotic eschar appears (figure 12). This eschar is usually bigger than the one observed in MSF cases, and it is surrounded by an erythema. When the tick-bite is out of the head, the skin lesion resembles the erythema migrans of Lyme disease. Other typical manifestation, which is always present when the bite is on the head, is the presence of regional and very painful lymphadenopathies.
On the contrary of other rickettsioses, in DEBONEL/TIBOLA there are not systemic clinical signs (or they are rare), such as fever or maculo-papular rash (Oteo et al., 2004b). The clinical course is sub-acute and no severe complications have been described.
Fig. 12. DEBONEL/TIBOLA patient with the typical crusted lesion on the scalp.
Scrub typhus
The etiological agent of scrub typhus is Orientia tsutsugamushi, which is transmitted by chigger bites (trombiculid mite larvae). It is mainly distributed in Afghanistan, India, Pakistan, Sri-Lanka, Kashmir, China, Nepal, Japan, Korea, Vietnam, Indonesia, Laos, Philippines, Papua New Guinea and Australia (Figure 11). Cases are mainly observed in autumn and spring, in temperate zones where the bite of this arthropod, which is on vegetation, is frequent. The incubation period is about 10 or more days and the clinical signs and symptoms are similar to typhus syndrome, including the rash which is transient and easily missed. A difference with typhus syndrome is the presence of eschar that is frequently multiple. The presence of regional lymphadenopathy is also more frequent. The mortality can be high despite the correct antimicrobial treatment. Outbreaks related to military operations have been reported.Most travel acquired cases of scrub typhus occur in patients returning from Southeast Asia (Jensenius et al., 2004, 2006).
Rickettsialpox
Rickettsialpox is a worldwide (North America, Eastern Europe, Korea and southern Africa) rickettsiosis caused by Rickettsia akari and transmitted by the bite of the mouse mite Lyponyssoides sanguineus. We can consider it a remerging infection since several cases have been detected in New York City after September 11 attacks (Paddock et al., 2003). Patients have fever, a prominent eschar -which is the best sign of the disease- and rash that, as occurs in ATBF and Queensland tick typhus, may be vesicular. Palms and soles are not involved. The presence of regional lymphadenopathy is common. Patients recover without treatment in most cases (Raoult, 2010b).
Laboratory diagnostic tools
As occurs for all infectious diseases, the most definitive diagnostic method is the rickettsial isolation in culture. The main problem is that Rickettsia spp. are strictly intracellular bacteria, conventional growth media cannot be used, and a laboratory with P3 safety level (not generally available in clinical microbiology labs) is necessary. Furthermore, culture is not very sensitive and the yield decreases when clinical samples are taken after antibiotic treatment or when samples are not processed within 24 hours. It is a slow technique that is used for research purposes but not for the routine clinical practice. Centrifugation shell-vial technique is a commercially available adaptation of cell cultures that is easier to handle, faster and less hazardous. Isolation attempts on cell cultures may be performed using buffy coat or tissue samples (eschar biopsies when possible). If not processed within 24 h, samples must be frozen at -70°C or in liquid nitrogen.
Detection of rickettsiae by Giménez or Giemsa staining from blood and tissue samples would allow the confirmation of the diagnosis, but these techniques are non-specific and their sensitivity is very low.
In some laboratories molecular biology tools, such as polymerase chain reaction (PCR) and sequencing, are also available. PCR-based assays from anticoagulated blood, biopsies and arthropod tissue samples targeting Rickettsia spp. genes are quite sensitive and useful for a quick diagnosis of these infections. The evaluation of several primer sets for the molecular diagnosis of rickettsioses demonstrated that the performance of three sequential PCRs (nested or semi-nested ones) allowed the detection and identification of Rickettsia species in a high percentage of the samples with previous clinical diagnosis or microbiological confirmation (serological analysis) of rickettsiosis (Santibáñez et al., 2011). Blood and tissue samples should be stored at -20°C or lower if PCR-based diagnosis is delayed for more than 24 hours. The European guidelines for the diagnosis of tick-borne bacterial diseases contain useful information for clinicians and microbiologists (Brouqui et a!., 2004). Indirect diagnostic tests and specifically, immunofluorescence assays (IFA) are considered the standard tests. Besides, since most traveller patients are investigated after returning, IFA are the most available tools for diagnosis. Acute and convalescent sera (collected 4-6 weeks after the onset of the illness) should be taken. In many cases we cannot observe seroconvertion but a high titre of antibodies. Cross-reactions among Rickettsia spp. make very difficult to definitively identify the causative agent by means of IFA. This can only be achieved in reference centres in which different antigens and other serological assays, such as western-blot, are available. Serum samples can be preserved at -20°C or lower for several months without significant degradation of antibodies.
Ticks removed from patients can be used as tools for the diagnosis of tick-borne rickettsioses. The strategy includes the identification of the tick to the species level, and the detection or isolation of rickettsias (Table 3).
|
1. |
Identification of the ticks to the species level |
|
2. |
Detection of bacteria in ticks with the use of staining tests (haemolymph for viable ticks; salivary glands if ticks were frozen), or PCR-based methods (using one-half of the tick, the other half being kept frozen). PCR may also be done using only ticks that stain positive. |
|
3. |
Sequencing of the amplified PCR fragment and comparison with available sequences in sequence databases. |
|
4. |
If there is 100% similarity between the tested sequence and the corresponding sequence of a known organism, the presumptive identification is confirmed |
|
5. |
If the tested sequence appears to be different from all corresponding sequences available, the organism is probably a new strain and should be isolated and characterized from the stored frozen part of the tick |
Table 3. Strategy for detecting and/or isolating rickettsias from ticks
Diagnostic scores with epidemiological, clinical and laboratory tests for some tick-borne rickettsioses (ATBF and MSF) have been proposed (Tables 4 and 5).
|
a. |
Direct evidence of R. africae infection by culture and / or PCR or |
|
b. |
Clinical and epidemiological features highly suggestive of ATBF, such as multiple inoculation eschars and / or regional lymphadenitis and / or a vesicular rash and / or similar symptoms among other members of the same group of travellers coming back from an endemic area (sub-Saharan Africa or French West Indies) and Positive serology against spotted fever group rickettsiae or |
|
c. |
Clinical and epidemiological features consistent with a spotted fever group rickettsiosis such as fever and / or any cutaneous rash and / or single inoculation eschar after travel to sub-Saharan Africa or French West Indies and Serology specific for a recent R. africae infection (seroconversion or presence of IgM φ 1:32), with antibodies to R. africae greater than those to R. conorii by at least two dilutions, and / or a Western blot or cross-absorption showing antibodies specific for R. africae |
Table 4. Diagnostic criteria for African-tick bite fever (ATBF). A patient is considered to have ATBF when criteria A, B or C are met
|
CRITERIA |
SCORE* |
|
Epidemiological criteria |
|
|
Stay in endemic area |
2 |
|
Occurrence in May-October |
2 |
|
Contact (certain or possible) with dog ticks |
2 |
|
Clinical criteria |
|
|
Fever > 39°C |
5 |
|
Eschar |
5 |
|
Maculopapular or purpuric rash |
5 |
|
Two of the above criteria |
3 |
|
All three of the above criteria |
5 |
|
Non-specific laboratory findings |
|
|
Platelets < 150 GL |
1 |
|
SGOT or SGPT > 50 UL |
1 |
|
Bacteriological criteria |
|
|
Blood culture positive for Rickettsia conorii |
25 |
|
Detection of Rickettsia conorii in a skin biopsy |
25 |
|
Serological criteria |
|
|
Single serum and IgG > 1 / 128 |
5 |
|
Single serum and IgG > 1 / 128 and IgM > 1 / 64 |
10 |
|
Four-fold increase in two sera obtained within a 2-week interval |
10 |
SGOT, serum glutamate-oxaloacetate transaminase; SGPT, serum glutamate-pyruvate transaminase. aA positive diagnosis is made when the overall score is > 25.
Table 5. Diagnostic criteria for Mediterranean spotted fever caused by Rickettsia conorii
Prophylaxis
An important topic in the field of rickettsioses is related to prophylaxis. Since the majority of rickettsioses associated to travels are transmitted by ticks, the main preventive measure is to avoid tick-bites. Measures to avoid chiggers’ attacks are the same as the ones used against ticks. Only fleas can be more difficult to avoid when cats and other pets are abundant. If there is risk of getting lice, hygiene measures such as changing clothing (they live in the seams of clothing) may be sufficient.
How can we avoid tick-bites? There are some rules that can be useful to avoid arthropod-bites:
1. You must not wear dark clothes to see the ticks and remove them before attaching. Curiously, dark clothes attract less arthropods than clear ones. But, in our opinion, to look for the arthropods and remove them as soon as possible is more effective.
2. For outdoor activities (grass areas or mountains) you do not have to exposure your body to ticks. Thus, it is very useful to wear clothing that covers the majority of your body. The trousers must be tucked in your shocks with boots. Long sleeves shirt must be tucked into trousers. You must also wear a cap (especially children).
3. Permethrin-based repellents can be used on clothing, although their effect is short in time and the application should be repeated every few hours.
4. A careful inspection of clothing and body looking for ticks after returning from outdoors activities in endemic areas as well as removing them correctly has been effective for the prevention of Lyme disease. The tick needs at least 24-48 hours for the transmission of Borrelia burgdorferi. This measure can be less efficient for Rickettsia spp. because these microorganisms can be transmitted since the first hours. But, anyway, the removal of the tick has to be done. 5. The contact with parasitized pets and wild animals must be avoided. There are two questions that physicians have linked up with tick-bites: How must I remove the tick? Must I take prophylactic drugs after a tick-bite?
The first question is easy to answer. The most useful method to remove an attached tick is using forceps. Smooth forceps (without teeth) must be introduced between the tick’s head and the skin in a 90° angle and then pull (Oteo et al., 1996). Other traditional methods as using oil, burning or freezing must be forgotten.
The other question is the use of prophylactic drugs after arthropod bites. There are no studies to answer this question. The transmission of rickettsias may be very quick, so we cannot extrapolate the recommendations for Lyme disease. Anyway, when people have been bitten by several ticks in an endemic area for a determinate disease (i.e.: Kruger National Park in South Africa and ATBF) and if the patient is anxious, we can offer doxycycline. It has been demonstrated that 3 doses of 100 mg. every 12 hours is safety and sufficient as treatment for the majority of rickettsioses. We must be cautious with the sun to avoid photo-sensibility. Children can take doxycycline for a short period of time. It is only contraindicated for pregnant women and in this case we can use macrolides (i.e. azythromycin). Vaccine approaches for prevention of rickettsial diseases have been developed since the past century, but currently no vaccine is available. Major surface protein antigens (OmpA and OmpB) of R. rickettsii and R. conorii are candidate vaccine antigens. Molecular biology techniques such as selection, cloning and expression of genes encoding R. prowazekii virulence-associated proteins, offer the opportunity to develop new rickettsial vaccines against typhus group rickettsiae. Further research is needed to develop effective vaccines without undesirable toxic reactions (Azad & Radulovic, 2003; Walker, 2009).
Treatment
The treatment of rickettsiosis should be initiated as soon as possible. Antibiotics are very effective and may avoid severe complications and death. In all cases if rickettsiosis is suspected, samples should be sent for laboratory confirmation. In DEBONEL/TIBOLA, in which the clinical signs and symptoms are less severe, recovery without antimicrobials occurs but the use of antibiotics shortens the clinical course and improves the clinical picture (Ibarra et al., 2005).
Doxycycline is the most useful drug in children and adults. Doxycycline can be administered in short course (100 mg. every 12 hours for one day) for the treatment of typhus and scrub typhus. In the case of MSF, 2 doses of 200 mg./12 hous are also very effective (in children, 5 mg./kg./12hours); although most physicians use 100 mg. every 12 hours for 3-7 days after fever disappears. The same can be recommended for ATBF. This antibiotic regimen could probably be followed in other tick-borne rickettsioses but there are not good evidences (clinical assays) to support a recommendation. In RMSF the administration of doxycycline for 7 days is recommended. Other drugs that can be prescribed when not using doxycycline (allergy or pregnancy) are chloramphenicol (50-75 mg./kg./day given in 4 doses for 7-10 days) and azythromycin (500 mg./day for 5 days). Doxycycline for 7 days is the treatment of choice for rickettsialpox. Although there is in vitro susceptibility to quinolones, the use of these drugs has been associated with worse clinical course (Botelho-Nevers et al., 2011).
Conclusion
In conclusion, rickettsioses are a worldwide threat that must be suspected in travellers returning from endemic areas. Most cases are caused by tick-bites, although in some areas of the world old diseases as typhus are present, and the risk exists. Rickettsiosis must be suspected in all patients with fever, exanthema with or without rash. Starting treatment with doxycycline when possible may be essential to rapidly recover and avoid complications. ATBF along with malaria is the leading cause of fever after returning from Sub-Saharan Africa.