Eye
The frequency of ocular manifestations for sarcoidosis varies depending on race. In Japan, >70% of sarcoidosis patients develop ocular disease, while in the United States only 30% have eye disease, with problems more common in blacks than whites. Although the most common manifestation is an anterior uveitis, over a quarter of patients will have inflammation at the posterior of the eye, including retinitis and pars planitis. While symptoms such as photophobia, blurred vision, and increased tearing can occur, some asymptomatic patients still have active inflammation. Initially asymptomatic patients with ocular sarcoidosis can eventually develop blindness. Therefore, it is recommended that all patients with sarcoidosis receive a dedicated ophthalmologic examination. Sicca is seen in over half of the chronic sarcoidosis patients. Dry eyes appear to be a reflection of prior lacrimal gland disease. Although the patient may no longer have active inflammation, the dry eyes may require natural tears or other lubricants.
Liver
Using biopsies to detect granulomatous disease, liver involvement can be identified in over half of sarcoidosis patients. However, using liver function studies, only 20-30% of patients will have evidence of liver involvement. The most common abnormality of liver function is an elevation of the alkaline phosphatase level, consistent with an obstructive pattern. In addition, elevated transaminase levels can occur. An elevated bilirubin level is a marker for more advanced liver disease. Overall, only 5% of sarcoidosis patients have sufficient symptoms from their liver disease to require specific ther- 17 apy. Although symptoms can be due to hepatomegaly, more frequently symptoms result from extensive intra-hepatic cholestasis leading to portal hypertension. In this case, ascites and esophageal varices can occur. It is rare that a sarcoidosis patient will require a liver transplant, because even the patient with cirrhosis due to sarcoidosis can respond to systemic therapy. On a cautionary note, patients with both sarcoidosis and hepatitis C should avoid therapy with interferon α because of its association with the development or worsening of granulomatous disease.
Bone Marrow and Spleen
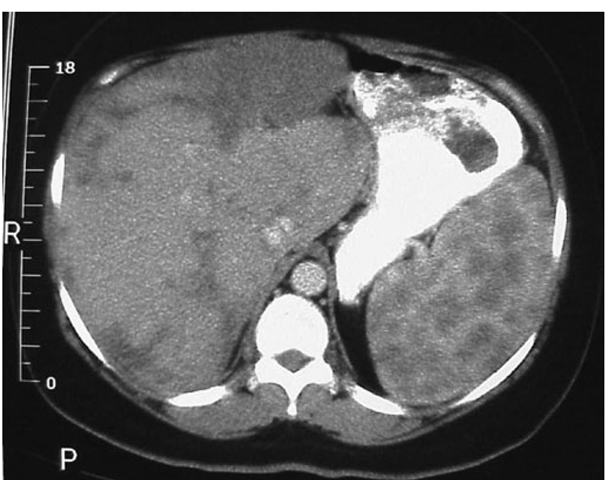
One or more bone marrow manifestations can be identified in many sarcoidosis patients. The most common hematologic problem is lymphopenia, which is a reflection of sequestration of the lymphocytes into the areas of inflammation. Anemia occurs in 20% of patients and leukopenia is less common. Bone marrow examination will reveal granulomas in about a third of patients. Splenomegaly can be detected in 5-10% of patients, but splenic biopsy reveals granulomas in 60% of patients. The CT scan can be relatively specific for sarcoidosis involvement of the spleen (Fig. 13-6). Both bone marrow and spleen involvement are more common in blacks than whites. These manifestations alone are rarely an indication for therapy. On occasion, splenectomy may be indicated for massive symptomatic splenomegaly or profound pancytopenia.
FIGURE 13-6
CT scan of the abdomen after oral and intravenous contrast. The stomach is compressed by the enlarged spleen. Within the spleen, areas of hypo- and hyperdensity are identified.
Calcium Metabolism
Hypercalcemia and/or hypercalciuria occurs in about 10% of sarcoidosis patients. It is more common in whites than blacks and in men. The mechanism of abnormal calcium metabolism is increased production of 1.25-dihydroxyvitamin D by the granuloma itself. The 1.25-dihydroxyvitamin D causes increased intestinal absorption of calcium, leading to hypercalcemia with a suppressed parathyroid hormone (PTH) level. Increased exogenous vitamin D from diet or sunlight exposure may exacerbate this problem. Serum calcium should be determined as part of the initial evaluation of all sarcoidosis patients, and a repeat determination may be useful during the summer months with increased sun exposure. In patients with a history of renal calculi, a 24-h urine calcium measurement should be obtained. If a sarcoidosis patient with a history of renal calculi is to be placed on calcium supplements, a follow-up 24-h urine calcium level should be measured.
Renal Disease
Direct kidney involvement occurs in <5% of sarcoidosis patients. It is associated with granulomas in the kidney itself and can lead to nephritis. However, hypercalcemia is the most likely cause of sarcoidosis-associated renal disease. In 1-2% of sarcoidosis patients, acute renal failure has been encountered as a result of hypercalcemia. Treatment of the hypercalcemia with glucocorticoids and other therapies often improves, but does not totally resolve, the renal dysfunction.
Nervous System
Neurologic disease is reported in 5-10% of sarcoidosis patients and appears to be of equal frequency across all ethnic groups. Any part of the central or peripheral nervous system can be affected.The presence of granulomatous inflammation is often visible on MRI studies. The MRI with gadolinium enhancement may demonstrate space-occupying lesions, but the MRI can be negative due to small lesions or the effect of systemic therapy in reducing the inflammation. The cerebral spinal fluid (CSF) findings include a lymphocytic meningitis with a mild increase in protein. The CSF glucose is usually normal but can be low. Certain areas of the nervous system are more commonly affected in neurosarcoidosis. These include cranial nerve involvement, basilar meningitis, myelopathy, and anterior hypothalamic disease with associated diabetes insipidus. Seizures and cognitive changes also occur. Of the cranial nerves, seventh nerve paralysis can be transient and can be mistaken for Bell’s palsy (idiopathic seventh nerve paralysis). Since this form of neurosarcoidosis often resolves within weeks and does not recur, it may have occurred prior to a definitive diagnosis of sarcoidosis. Optic neuritis is another cranial nerve manifestation of sarcoidosis. This manifestation is more chronic and usually requires long-term systemic therapy. It can be associated with both anterior and posterior uveitis. Differentiating between neurosarcoidosis and multiple sclerosis can be difficult at times. Optic neuritis can occur in both diseases. In some patients with sarcoidosis, multiple enhancing white matter abnormalities may be detected by MRI, suggesting multiple sclerosis. In such cases, the presence of meningeal enhancement or hypothalamic involvement suggests neurosarcoidosis, as does evidence of extraneurologic disease such as pulmonary or skin involvement, which also suggests sarcoidosis. Since the response of neurosarcoidosis to glucocorticoids and cytotoxic therapy is different from that of multiple sclerosis, differentiating between these disease entities is important.
Cardiac
The presence of cardiac involvement is influenced by race. Over a quarter of Japanese sarcoidosis patients develop cardiac disease, whereas only 5% of sarcoidosis patients in the United States and Europe develop cardiac disease. However, there is no apparent difference between whites and blacks. Cardiac disease usually presents as either congestive heart failure or cardiac arrhythmias. Both manifestations result from infiltration of the heart muscle by granulomas. Diffuse granulomatous involvement of the heart muscle can lead to ejection fractions below 10%. Even in this situation, improvement in the ejection fraction can occur with systemic therapy. Arrhythmias can also occur with diffuse infiltration or with more patchy cardiac involvement. If the AV node is infiltrated, heart block can occur. This can be detected by routine electrocardiography. Ventricular arrhythmias and sudden death due to ventricular tachycardia are common causes of death. Arrhythmias are best detected using 24-h ambulatory monitoring. Because ventricular arrhythmias are usually multifocal due to patchy multiple granulomas in the heart, ablation therapy is not useful. Patients with significant ventricular arrhythmias should be considered for an implanted defibrillator, which appears to have reduced the rate of death in cardiac sarcoidosis. While systemic therapy can be useful in treating the arrhythmias, patients may still have malignant arrhythmias up to 6 months after starting successful treatment, and the risk for recurrent arrhythmias occurs whenever medications are tapered.
Musculoskeletal System
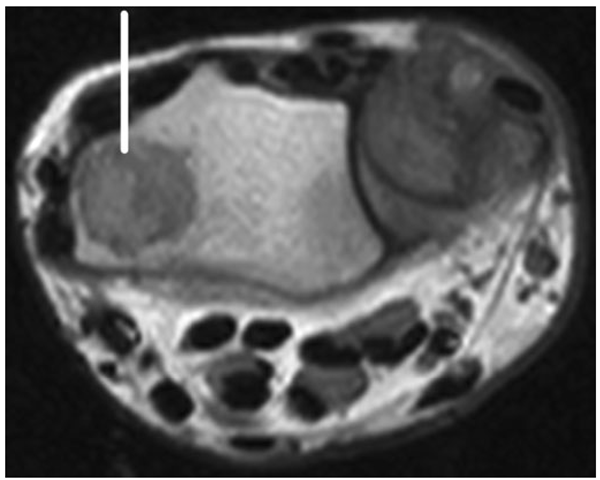
Direct granulomatous bone and muscle involvement as documented by x-ray, MRI (Fig. 13-7), gallium scan, or biopsy can be seen in about 10% of sarcoidosis patients.
FIGURE 13-7
MRI of wrist demonstrating large cyst in a sarcoidosis patient (line).
However, a larger percentage of sarcoidosis patients complain of myalgias and arthralgias. These complaints are similar to those reported by patients with other inflammatory diseases, including chronic infections such as mononucleosis. Fatigue associated with sarcoidosis may be overwhelming for many patients. Recent studies have demonstrated a link between fatigue and small peripheral nerve fiber disease in sarcoidosis.
Other Organ Involvement
Although sarcoidosis can affect any organ of the body, rarely does it involve the breast, testes, ovary, or stomach. Because of the rarity of involvement, a mass in one of these areas requires a biopsy to rule out other diseases including cancer. For example, in a study of breast problems in female sarcoidosis patients, a breast lesion was more likely to be granulomas from sarcoidosis than from breast cancer. However, findings on the physical examination or mammogram cannot reliably differentiate between these lesions. More importantly, as women with sarcoidosis age, breast cancer becomes more common. Therefore, it is recommended that routine screening including mammography be performed along with other imaging studies (ultrasound, MRI) or biopsy as clinically indicated.
Complications
Sarcoidosis is usually a self-limited, non-life-threatening disease. However, organ-threatening disease can occur. These complications can include blindness, paraplegia, or renal failure. Death from sarcoidosis occurs in about 5% of patients seen in sarcoidosis referral clinics. The usual causes of death related to sarcoidosis are from lung, cardiac, neurologic, or liver involvement. In respiratory failure, an elevation of the right atrial pressure is a poor prognostic finding. Lung complications can also include infections such as mycetoma, which can subsequently lead to massive bleeding. In addition, the use of immunosuppressive agents can increase the incidence of serious infections.
Laboratory Findings
The chest roentgenogram remains the most commonly used tool to assess lung involvement in sarcoidosis. As noted above, the chest roentgenogram classifies involvement into four stages, with stages 1 and 2 having hilar and paratracheal adenopathy. The CT scan has been used increasingly in evaluating interstitial lung disease. In sarcoidosis, the presence of adenopathy and a nodular infiltrate is not specific for sarcoidosis. Adenopathy up to 2 cm can be seen in other inflammatory lung diseases such as idiopathic pulmonary fibrosis. However, adenopathy >2 cm in the short axis supports the diagnosis of sarcoidosis over other interstitial lung diseases.
Gallium 67 scanning has been used over the years to detect inflammatory activity in various parts of the body. A negative scan can be easily misinterpreted as the scan will quickly revert to normal during glucocorticoid therapy. In addition, the test requires that the patient return 2-4 days later. More recently, the positron emission tomography (PET) scan, using radiolabeled fluo-rodeoxy glucose as the marker, has provided information similar to that of the gallium scan. While the sensitivity of PET scanning has not been fully evaluated in sarcoidosis, it is important to recognize that a positive PET scan may be due to the granulomas from sarcoidosis and not to disseminated malignancy.
Serum levels of angiotensin-converting enzyme (ACE) can be helpful in the diagnosis of sarcoidosis. However, the test has somewhat low sensitivity and specificity. Elevated levels of ACE are reported in 60% of patients with acute disease and only 20% of patients with chronic disease. Although there are several causes for mild elevation of ACE, including diabetes, elevations of >50% of the upper limit of normal are seen in only a few conditions including sarcoidosis, leprosy, Gaucher’s disease, hyperthyroidism, and disseminated granulomatous infections such as miliary tuberculosis. The ACE level in lymphoma is usually lower than normal, which may provide a useful distinction from sarcoidosis. There is an insertion/deletion (I/D) polymorphism of the ACE gene on what is felt to be in the noncritical part of the gene. There is a phenotypic difference for ACE levels, with II polymorphism having the lowest and DD polymorphism the highest levels of ACE for both sarcoidosis patients and healthy controls. There is no clear-cut association between ACE phenotype and clinical manifestation of disease. Because the ACE level is determined by a biologic assay, the concurrent use of an ACE inhibitor such as lisinopril will lead to a very low ACE level.
Diagnosis
The diagnosis of sarcoidosis requires both compatible clinical features and pathologic findings. Since the cause of sarcoidosis remains elusive, the diagnosis cannot be made with 100% certainty. Nevertheless, the diagnosis can be made with reasonable certainty based on history and physical features along with laboratory and pathologic findings.
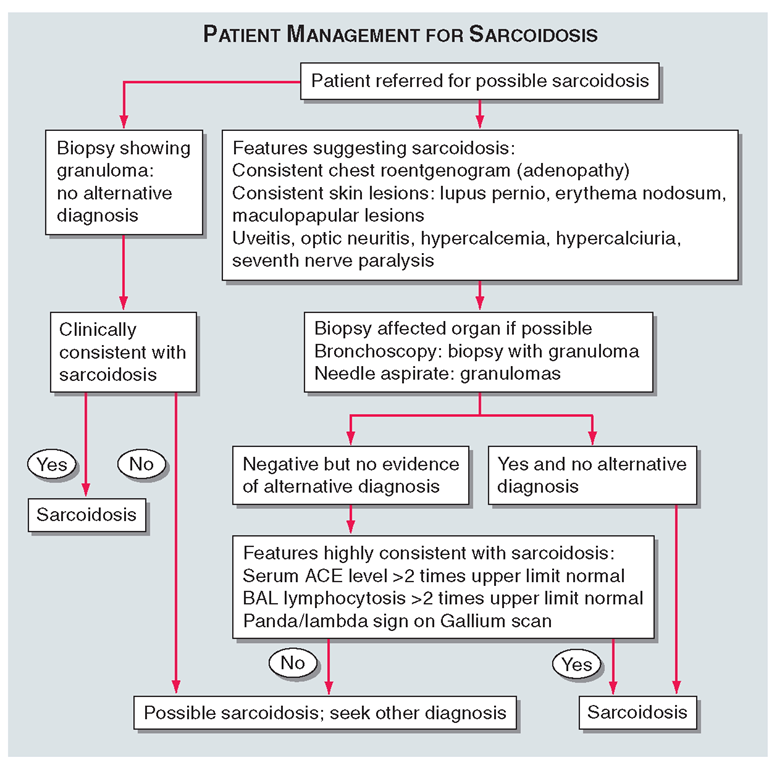
Patients are usually evaluated for possible sarcoidosis based on two scenarios (Fig. 13-8). In the first scenario, a patient may undergo a biopsy revealing a noncaseating granuloma in either a pulmonary or an extrapulmonary organ. If the clinical presentation is consistent with sarcoidosis and there is no alternative cause for the granulomas identified, then the patient is felt to have sarcoidosis.
In the second scenario, signs or symptoms suggesting sarcoidosis such as the presence of bilateral adenopathy may be present in an otherwise asymptomatic patient or a patient with uveitis or a rash consistent with sarcoidosis. At this point, a diagnostic procedure should be performed. For the patient with a compatible skin lesion, a skin biopsy should be considered. Other biopsies to consider could include liver, extrathoracic lymph node, or muscle. In some cases, a biopsy of the affected organ may not be easy to perform (such as a brain or spinal cord lesion). In other cases, such as an endomyocardial biopsy, the likelihood of a positive biopsy is low. Because of the high rate of pulmonary involvement in these cases, the lung may be easier to approach by bronchoscopy. During the bronchoscopy, a transbronchial biopsy, bronchial biopsy, or transbronchial needle aspirate of an enlarged mediastinal lymph node can be performed.
If the biopsy reveals granulomas, an alternative diagnosis such as infection or malignancy must be excluded. Bron-choscopic washings can be sent for cultures for fungi and tuberculosis. For the pathologist, the more tissue that is provided, the more comfortable is the diagnosis of sarcoidosis. A needle aspirate may be adequate in an otherwise classic case of sarcoidosis, but may be insufficient in a patient in whom lymphoma or fungal infection is a likely alternative diagnosis. Since granulomas can be seen on the edge of a lymphoma, the presence of a few granulomas from a needle aspirate may not be sufficient to clarify the diagnosis. Mediastinoscopy remains the procedure of choice to confirm the presence or absence of lymphoma in the mediastinum. Alternatively, for most patients, evidence of extrathoracic disease (e.g., eye involvement) may further support the diagnosis of sarcoidosis.
For patients with negative pathology, positive supportive tests may increase the likelihood of the diagnosis of sarcoidosis. These tests include an elevated ACE level, which can also be elevated in other granulomatous diseases but not in malignancy. A positive gallium scan can support the diagnosis if increased activity is noted in the parotids and lacrimal glands (Panda sign) or in the right paratracheal and left hilar area (lambda sign).
FIGURE 13-8
Proposed approach to management of patient with possible sarcoidosis. Presence of one or more of these features supports the diagnosis of sarcoidosis: uveitis, optic neuritis, hypercalcemia, hypercalciuria, seventh cranial nerve paralysis, diabetes insipidus.
A BAL is often performed during the bronchoscopy. An increase in the percentage of lymphocytes supports the diagnosis of sarcoidosis. The use of the lymphocyte markers CD4 and CD8 can be used to determine the CD4/CD8 ratio of these increased lymphocytes in the BAL fluid.A ratio of >3.5 is strongly supportive of sarcoidosis but is less sensitive than an increase in lymphocytes alone. Although in general, an increase in BAL lymphocytes is supportive of the diagnosis, other conditions must be considered.
These supportive tests when combined with commonly associated clinical features of the disease, which are not diagnostic of sarcoidosis, can enhance the diagnostic probability. These nondiagnostic features include uveitis, renal stones, hypercalcemia, seventh cranial nerve paralysis, or erythema nodosum.
The Kviem-Siltzbach procedure is a specific diagnostic test for sarcoidosis. An intradermal injection of specially prepared tissue derived from the spleen of a known sarcoidosis patient is biopsied 4-6 weeks after injection. If noncaseating granulomas are seen, this is highly specific for the diagnosis of sarcoidosis. Unfortunately, there is no commercially available Kviem-Siltzbach reagent, and some locally prepared batches have lower specificity. Thus, this test is of historic interest and is rarely used in current clinical practice.
Because the diagnosis of sarcoidosis can never be certain, over time other features may arise that lead to an alternative diagnosis. On the other hand, evidence for new organ involvement may eventually confirm the diagnosis of sarcoidosis.