PROCESSING AND MORPHOLOGY DEVELOPMENT
DSC studies show that the onset temperatures of polymerization are much lower for the Epon 862/W with organoclays than those for pure Epon 862/W 65-100°C vs. 122°C). The catalytic effect is likely because of the acidity from the ammonium (RNH3+) group of the pendent group from the organomontmorillonite.[15,18,19,39,40] The mechanism for the catalytic effect that the organoclay has on the epoxy polymerization may be explained as follows: The proton from the ammonium (RNH3+) forms a cation with the glycidyl group, causing the functional group to become more electrophilic, which becomes more easily attacked by the nucleophilic curing agent amine. Thus the pendent group (RNH3+) inside the gallery will catalyze the polymerization of the epoxy resin, resulting in cross-linking of the epoxy resin inside the gallery at relatively lower temperatures than those outside the gallery.
The addition of particles to a polymer generally increases its viscosity. If the organoclay significantly increases the viscosity of the epoxy resin, processing rates for aerospace composites, such as in resin transfer molding, may be difficult. The viscosity-vs.-temperature curves of the Epon 862/W mixture, the 3% SC6/Epon 862/ W mixture, and the 10% I.30E/Epon 862/W mixture, when heated at 2°C/min, is shown in Fig. 8. The complex viscosity of the epoxy resin with organoclays (23 P for 3% SC6/Epon 862/W and 30 P for 10% I.30E/Epon 862/W mixture at 30°C) is slightly higher than that of Epon 862/ W (11 P at 30°C). For the sample with 3% SC6 loading, the viscosity is increased very slightly from 0.6 P at 90°C to 2.7 P at 140°C, increased significantly from 7.6 P at 150°C to 108 P at 160°C, and increased very rapidly from 254 P at 161°C to 90,200 P at 165°C and to 4.6 x 105 P at 170°C. This trend is similar to that seen for the 10% I.30E/ Epon 862/W mixture. The viscosity is increased slightly from 0.7 P at 90°C to 4.2 P at 140°C, increased significantly from 8.4 P at 145°C to 110 P at 152°C, and increased again rapidly from 377 P at 155°C to 1900 P at 160°C and to 6.2 x 105 P at 170°C. For the sample of pure Epon 862/W mixture, the complex viscosity is increased very slightly to 2.1 P at 160°C, increased significantly from 2.1 P at 160°C to 43 P at 167°C, and very rapidly from 430 P at 168°C to 13,192 P at 170°C and to 5.9 x 105 P at 180°C. The rapid increase of the viscosity for the 3% SC6/Epon 862/W and 10% I.30E/ Epon 862/W mixture at relatively low temperatures is related to the catalytic effect for the polymerization from the organoclay. Therefore, the incorporation of the organoclay into the epoxy resin does increase the viscosity not only because of the expected effect of particles, but also through the catalytic effect of the organo-clays. However, the increase of the complex viscosity is limited and the processing window for the 3% SC6/ Epon 862/W mixture and 10% I.30E/Epon 862/W mixture is still quite wide. This is important for subsequent composite processes.
Fig. 8 Complex viscosity of the mixtures of pure Epon 862/W, 3% SC6/Epon 862/W, and 10% I.30E/Epon 862/W heated at 2°C/min.
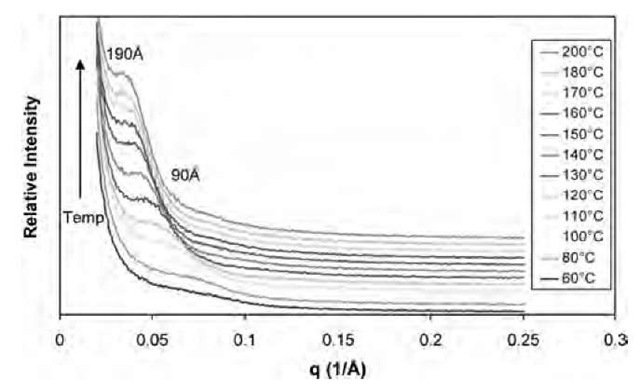
In situ SAXS can be used to monitor the morphology development of the nanocomposites; here, some typical data of a 3% SC6/Epon 862/W mixture heated up at 2°C/ min obtained at the National Synchrotron Light Source at the Brookhaven National Laboratory on Beamline X27C are shown in Fig. 9. SAXS data show that the initial interplanar spacing of the mixture is ~ 85 A compared with the original interplanar spacing of SC6 at 13.0 A. This increase of the interplanar spacing is because of the penetration of epoxy resin into and the expansion of the gallery of the organoclay. The larger expansion of the interplanar spacing for 3% SC6/Epon 862/W mixture (85 A), compared with that of 3% SC12/Epon 862/W mixture (35 A),[40] is due perhaps to the smaller pendent group of SC6, which makes the migration of the epoxy resin inside the gallery relatively easier. SAXS data coupled with thermal analysis can provide insights into the morphology development mechanisms during the curing process of layered-silicate epoxy nanocomposites. When the temperature is elevated from 60°C to 80°C, the peak corresponding to the interplanar spacing between the nano-layers remains fairly constant. During this period, there is almost no polymerization occurring, and the heating role in the expansion of the gallery of the nanoclay is very limited. From 80°C to 100°C, the increase of the interplanar spacing is still fairly limited. This is not unexpected as the onset temperature of curing for this mixture occurs at approximately 85°C based on DSC studies; polymerization is just beginning in this temperature region. From 100°C to 170°C, the peak corresponding to the interplanar spacing is shifted first gradually and then quickly from ~ 97 to ~ 176 A, and the intensity of the peaks increases gradually as well. The quick polymerization in the intragallery because of the catalytic effect from the pendent group in the intragallery results in the consumption of the epoxy monomer inside the gallery and enhances the migration of more epoxy monomers from extragallery to intragallery, thus enhancing the continued expansion of the intragallery. In addition, the exothermal heat of curing provides additional energy to that from the process-imposed heating to help expand the nanoclay sheets. Based on rheological studies (Fig. 8), the viscosities for the 3% SC6/Epon 862/W mixture are 0.8 P at 100°C, 0.9 P at 120°C, 1.1 P at 130°C, 7.6 P at 150°C, 108 P at 160°C, 254 P at 161°C, 1303 P at 162°C, and 90,200 P at 165°C. The low viscosity of the epoxy resin from 100°C to 160°C guarantees that the epoxy resin can easily migrate from extragallery to intragallery. With an increase in temperature, curing of the epoxy resin both intragallery and extragallery results in increasingly high viscosities (4.6 x 105 P at 170°C, and 8.7 x 105 P at 180°C). The epoxy is eventually almost fully cured with a viscosity of 1.0 x 106 P at 190°C, and the material surrounding the nanolayers of the organoclay at this point is rigid. Ultimately, the interplanar spacing between the nanolayers is around 190 A at 200°C.
Fig. 9 The in situ SAXS of the mixture of 3% SC6/Epon 862/W heated at 2°C/min.
Differences in the nanostructural evolution between the above-discussed SC6/Epon 862/W mixture and the previously studied SC12/Epon 862/W can be observed. The nanostructural or morphological evolution for the SC6-based nanocomposite was via a gradual ordered expansion of the galleries. In contrast, the SC12/Epon 862/W mixture morphological evolution occurs in stages, and experiences a transition from ordered to disordered and then back to order structure.1-39-1 For the SC12/Epon 862/W system, the initial interplanar spacing is 35 A after mixing. During initial curing, the silicate layers are distorted as the intragallery epoxy polymerizes and the outside resin infuses into the gallery through the tactoids edges. The SAXS data show an apparent loss of the peak associated with the initial spacing as the number of ordered scatterers is reduced because of resulting curvature of the nano-sheets. As more and more epoxy migrates, the gallery makeup becomes more homogenous, the curvature is reduced, and the ordered (registered) structure is regained. For the SC6/Epon 862/W system, the original interplanar spacing is on the order of ~ 85 A after mixing. In this case, the mixing step brings sufficient epoxy into the galleries as the epoxy is less impeded by the shorter alkyl chains (six-carbon chain in SC6 vs. 12-carbon chain in SC12); this results in an almost homogeneously filled gallery prior to heating. Therefore at the onset of polymerization, no distortion occurs and registry is maintained as the organoclay tactoid expands.
In summary, based on in situ SAXS studies, the exfoliation mechanism is seen to take place during curing. This important phenomenon enables low viscosity during the mixing processes and the ultimate formation of the nanoscale morphology during the curing steps of a process. This is vital to ultimate processability and manu-facturability of nanotailored reinforced composites for aerospace applications.
MORPHOLOGY DEVELOPMENT OF EPOXY NANOCOMPOSITES WITH DIFFERENT CURING AGENTS
The nature of the selected curing agent plays a role in the morphology development of epoxy nanocomposites.
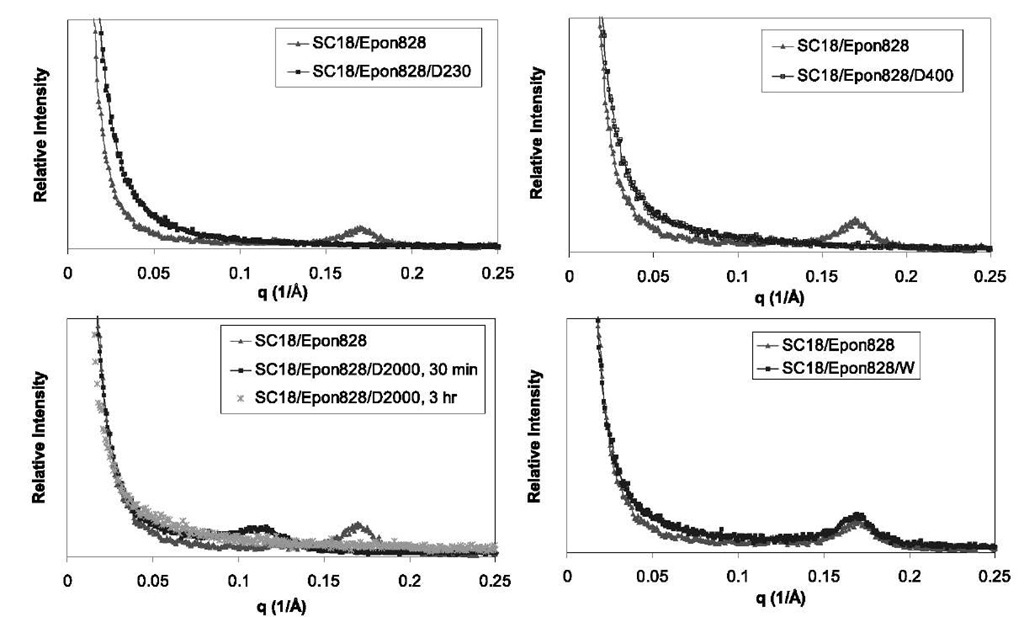
Fig. 10 SAXS of SC18/Epon 828 with SC18/Epon 828/D230, SC18/Epon 828 with SC18/Epon 828/D400, SC18/Epon 828 with SC18/ Epon 828/D2000 after 30 min and 3 hr, and SC18/Epon 828 with SC18/Epon 828/W.
When Epon 828 is mixed with organoclay SC18, Epon 828 penetrates inside the gallery of the organoclay. The SC18/Epon 828 mixture has a very ordered intercalated structure with an interplanar spacing of ~ 35 A compared with the original interplanar spacing of ~ 18 A of the SC18. When the mixture of SC18/Epon 828 was mixed with Jeffamine D230 or Jeffamine D400 curing agents and held for 30 min at room temperature, SAXS showed no peaks (Fig. 10). This indicates that Jeffamine D230 can easily penetrate into the gallery of the organoclay, therefore causing separation of the nanolayers and exfoliation. The SAXS shows the same pattern for fully cured systems. When the SC18/Epon 828 mixture was mixed with Jeffamine D2000 and held for 30 min at room temperature, SAXS showed the ordered nanolaminated structure with interplanar spacings of ~ 55 A (Fig. 10), again because of the penetration of the Jeffamine D2000 into the gallery of the organoclays and the expansion of the gallery. However, the larger molecular weight of Jeffamine D2000 and the size of the molecule reduced its mobility and penetration into the gallery; therefore an intercalated—instead of an exfoliated—structure is obtained. When the SC18/Epon 828/D2000 mixture was held in 60°C for 3 hr, SAXS showed no peaks (Fig. 10). The high temperature increased the mobility of the Jeffamine D2000 molecule, easing penetration of the gallery and causing the separation of the nanolayers. For the mixture of SC18/Epon 828 with curing agent W, SAXS indicates almost no change of the ordered nano-laminated structure and the interplanar spacings remain essentially constant at ~ 35 A (Fig. 10). This suggests that curing agent W is not compatible with the gallery of or-ganoclays, and that there is no or very little penetration of it into the gallery. Exfoliation for the SC18/Epon 828/W system took place during heatup. These observations demonstrate the role that curing agents may play in morphology development. Curing agent W is an aromatic amine containing an aromatic group, whereas the Jeffa-mine is a polyoxypropyleneamine curing agent containing oxygen with a more flexible chain. Simultaneously, the organoclay contains siloxane backbone with many oxygen atoms inside, which perhaps makes it more compatible with Jeffamine. Such properties and nature of different curing agents cause different exfoliation behaviors for epoxy nanocomposites.
EPOXY NANOCOMPOSITE AS PRIMER LAYER FOR AIRCRAFT COATING
Nanocomposites offer significant barrier properties, as observed by many researchers to date. Corrosion protection requirements of modern aircraft coating systems become increasingly demanding, and environmental as well as performance enhancements are sought. The incorporation of nanoclays into epoxy primers offers opportunities to develop environmentally benign, improved anticorrosion protection, which may provide new applications for nanocomposites. The most common primer layer for aircraft coatings is epoxy based on Epon 828 (diglycidylbisphenol-A) with Epi-Cure curing agent 8290-Y-60 [polyethylene polyamine adduct (60) and 2-propoxyethanol (40)-. Epon 828 with Jeffamine D400 curing agent is a model for such primers, and a 2.5% SC18/Epon 828/D400 nanocomposite achieves an essentially exfoliated morphology. This system has very high barrier resistance for anticorrosion protection and can be cured at room temperature within a week, similar to the Epon 828/curing agent 8290-Y-60 system. Corrosion performance was assessed through exposure of AA2024-T3-coated panels to dilute Harrison's solution (a mixture of 0.05% sodium chloride and 0.35% ammonium sulphate) for 24 hr, followed by potentiodynamic polarization scans.[45] The corrosion current was 0.4 pA for pure Epon 828/D400, which is representative of very good barrier properties. The 2.5% SC18/Epon 828/D400 nano-tailored coating exceeded the epoxy with a corrosion current of 0.2 pA. The improvement in the barrier properties of the SC18/Epon 828/D400 coating is attributed to the dispersion of the platy nanolayers in the epoxy matrix and the resulting tortuous path that they provide for fluids. Nanotailored epoxies and nanocomposites offer promise for anticorrosion coatings as well as any polymeric material that requires improved barrier properties or reduced permeability.
CONCLUSION
Both commercially available organoclay (I.30E) and synthetic organoclays (SC6, SC8, SC12, and SC18) are compatible with aerospace epoxy resins such as those studied here. Characterizations of the nanocomposites from WAXD, SAXS, and TEM confirm that their nano-structure can achieve interplanar spacings larger than 100 A—5-10 times the original organoclay spacings. Even loadings as high as 10% (I.30E) can reach relatively large interplanar spacings as confirmed by scattering and TEM studies. Selection of organoclay treatment is critical because it can affect both the math of morphology development during cure processing as well as the final interplanar spacings. Selection of curing agent can also affect nanostructure morphology. Nanomodified epoxies offer properties that are appropriate to many aerospace applications. The modus of the nanocomposite is significantly increased over the baseline epoxy from 15% to 35% in the glass state and to 300% in the rubber state, depending on loading as expected. The increase of the storage modulus is attributed to the high aspect ratio and high stiffness of the silicate nanolayers in the epoxy matrix. The change in the Tg for the nanocomposite compared with the pure epoxy resin is insignificant.
For the materials reported here, the flex strength of the nanocomposites is decreased compared with that of the pristine epoxy resin. This reduction of strength is perhaps attributed to the weak interface between the clay nano-layer and the epoxy resin—an area to be further addressed. Surprisingly, the strength at high temperature (250°F) of some nanocomposites can be greater than that of pure resin, which may allow for more effective utilization of polymer composites in high-temperature environments. The survivability of aerospace epoxy resins under oxygen plasma and perhaps other extreme environments can be improved through nanotailoring by organoclays as the resulting nanocomposite materials can form inorganic barrier layers. Anticorrosion and other permeability-dependent properties may lead to some near-term aerospace applications. For all properties, nanostructure morphology plays a role.
Processability of nanotailored resins is a factor for many current advanced composite fabrication methods. Initial in-depth studies indicate that nanostructural evolution and viscosity development lend themselves to composite processes. The quick polymerization in the intragallery because of the catalytic effect from the pendent group results in the consumption of the epoxy monomer inside the gallery and makes the migration of more epoxy monomers from extragallery to intragallery, thus causing the expansion of the intragallery during the early stages of processing when viscosity is low. In addition, the exothermal heat from curing provides extra energy, in addition to energy from heating, to help expand the nanoclay sheets. Because the development of exfoliated morphology takes place during the cure for epoxy, as opposed to during the mixture processing step as in the case of thermoplastics, the impact of the organoclay on viscosity during the processing of epoxy nanocomposites is minimized. This is a very attractive attribute from the viewpoint of processing.
Ultimate control of properties through fundamental understanding of process morphology and nanoscale morphology properties is key to aerospace applications. Although there are many opportunities that may open up as a result of the insight into nanotailoring of epoxies through organoclays obtained through studies such as those reported and referenced here, many material developments will be required to fully exploit nanocomposites for the aerospace community, such as interface modifications, and validated analytical predictive tools will eventually be required to guide such material development as well as to predict their long-term performance.