Introduction
In the past two decades, thoroughly standardized mouse incubation time and brain lesion profile scoring assays have been developed to discriminate between prion strains. However, in these mouse infection experiments, large numbers of animals (about 20 mice/line) from three different highly inbred mouse lines (C57Bl, VM95, RIII), plus their intercrosses, need to be infected, and their brain tissues subsequently examined (1-8). Although results obtained are highly reliable, the effort and time needed for conducting these experiments are considerable. Therefore, alternative criteria and techniques have been developed to characterize transmissible spongiform encephalopathy (TSE) agents. Prion infections are accompanied by the accumulation of an abnormal isoform (designated PrPSc) of normal host-encoded prion protein (PrPC). Both isoforms have the same amino acid sequence and molecular mass, but differ significantly in their three-dimensional structure and biochemical characteristics. The three-dimensional structure of PrPC is characterized by a high a-heli-cal content (9); all or part of it undergoes a posttranslational modification to P-sheet in PrPSc (10-12). Although PrPC (33-35 kDa) is completely hydro-lyzed by protease treatment, PrPSc is partially resistant to proteinase K(PK) as 62 N-terminal amino acids are cleaved, leaving a core fragment of approx 141 amino acids (27-30 kDa) unhydrolyzed (13).
Kascsak et al. described, for the first time, differences in the molecular masses of nonglycosylated PrPSc fragments of scrapie strains after proteinase K digestion, i.e. PrPSc of strain 87 V had a slightly smaller molecular mass than the PrPSc of most other scrapie strains studied (about 19 kDa, compared to 21 kDa) (14,15). A similar difference was later noted for the two known and well-characterized transmissible mink encephalopathy strains, "hyper" and "drowsy" (16,17).
Collinge et al. (18) eventually analyzed PK-treated PrPSc from Creutzfeldt-Jakob disease (CJD) and variant (v)CJD cases, and found three different molecular masses for the nonglycosylated compound: 21, 20, and 19 kDa. PrPSc from all vCJD cases clustered in the 19-kDa type. However, the nonglycosylated 20 kDa PrPSc type has not been observed by others (19,20). This discrepancy may partly result from the presence or absence of ethylendiamine tetraacetic acid (EDTA) during the PK treatment. Wadsworth et al. (21) could show that the addition of this metal ion binding compound leads to a smaller cleavage product of 19 kDa (designated type 1* or 2*, respectively), instead of PrPSc types 1 (21 kDa) and 2 (20 kDa).
Apart from the different molecular masses of nonglycosylated PrPSc, Collinge et al. also noted differences in the ratios of the three glycoforms of PrPSc (18). PrPC precursor protein is glycosylated twice at asparagines at positions 182 and 198 of the protein (22). As a result of this process non-, mono-, and diglycosylated PrPSc fractions are accumulated in infected cells. However, although monoglycosylated PrPSc predominates in all conventional human TSE cases analyzed to date, vCJD PrPSc is characterized by a predominance of diglycosylated PrPSc (18).
By combining the two results, molecular mass determination and glycotyping, it became clear that vCJD represented a novel type of prion disease in humans. Because PrPSc derived from bovine spongiform encephalopa-thy (BSE)-infected mice had shown similar molecular characteristics, such as low molecular mass and heavily stained diglycosylated PrPSc, a link between both diseases has been proposed.
We have recently found that PrPSc from different experimental and field BSE, and scrapie strains/isolates, are characterized by differences in their resistance to long-term exposure to proteinase K. Hence, the determination of the long-term proteinase K (PK) resistance can reveal additional information on the underlying prion strain.
PrPSc Glycotyping, Molecular Mass Determination, and Long-Term Proteinase K Resistance Analysis: Basic Protocol
The determination of molecular masses and relative proportions of non-, mono-, and diglycosylated PrPSc are frequently summarized under the term "glycotyping," although both relate to different molecular characteristics. Therefore, in this topic, both designations are used in their strict sense, and "glycotyping" refers to the determination of the glycoform ratios only.
Sample Preparation
PrPSc containing samples are homogenized. Tissues are cut into small pieces and put into glass homogenizers of appropriate size or syringes with needles of subsequently lower diameters. A number of different lysis buffers have been described in the literature (18-20,23). Good results are obtained using phosphate buffered solution supplemented with 0.5% NP-40 and 0.5% sodium desoxycholate, which is added to yield a 10% (wv) homogenate. Cells are thoroughly disintegrated by 20-30 plunger strokes, and residual tissue clumps further broken by 30-60 s high-power sonification in a closed vessel (for safety reasons). Coarse debris is then removed by centrifugation for 5 min at 325g, and aliquots stored in the freezer at -20°C until further use. PrPSc signals in homogenates, which contain detergents (particularly, sarkosyl or sodium dodecyl sulphate [SDS]), can fade away within weeks, even if homogenates are kept at -20°C, and are not thawed intermediately. This deterioration seems to affect all three PrP bands to the same degree. Therefore, tissue homogenates should be rapidly processed and analyzed. Sonification of repeatedly thawed samples may partially restore the signals. Samples are eventually digested by PK at conditions chosen according to particular needs (see Subheading 3.2.). The digestion is terminated by addition of 5 mM phenylmethylsulfonylfluoride (obtainable from Boehringer, Mannheim, Germany) and/or heat (70°C, 10 min).
Alternatively, scrapie-associated fibrils (SAFs) can be used. SAFs are purified according to previously published protocols (24) which also include a PK digestion step (10 ^g/mL, 1 h, 37°C). PrPSc signals in SAF preparations also tend to fade away over time.
The choice of sample preparation depends mostly on the expected PrPSc content in these tissues. High PrPSc concentrations are found in brain tissues from patients with CJD, vCJD, kuru, and Gerstmann-Straussler Scheincker disease, so that homogenates can readily be used. The same applies for brain tissues of scrapie-infected sheep, hamsters, and mice. In contrast, tissues from patients with familial fatal insomnia and cattle with BSE contain much lower amounts of PrPSc, so that a previous concentration step may be necessary. This is also recommended when brain areas with low PrPSc concentrations are used.
Gel Electrophoresis and Immunoblot
Samples are subsequently boiled in sample buffer (50 mM Tris, 117 mM Saccharose, 2% [w/v] SDS, 1% [w/v] p-mercaptoethanol, 0.01 % (w/v) bro-mphenol blue, pH 6.8) for 5 min. It should be noted that SDS concentrations used in sample buffers vary considerable (0.5-5%) between laboratories. Moreover, different concentrations of reducing substances, such as dithiothreitol or mercaptoethanol are used, with possible impact on the protein separation during electrophoresis.
Samples are run on 12-16% SDS-polyacrylamide gel electrophoresis gels (0.75 mm thickness) at about 10 V/cm, using a commercial minigel apparatus, and proteins are electrotransferred (5 mA/cm2 for 50 min) onto PVDF (available from Immobilon-P, Millipore, Bedford, MA) membranes, using a commercially available semidry blotting apparatus. It may be necessary to check an even protein transfer over the whole electrode area by blotting standard protein mixtures.
Membranes are then blocked by incubation in 5% nonfat dry milk in phosphate-buffered saline containing 0.1% Tween-20 (phosphate-buffered saline [PBS-]Tween). Sheets are incubated for 2 h with antibodies in PBS-Tween supplemented with 5% nonfat dry milk. After three washes in PBS-Tween, the sheets are incubated for another 90 min with horseradish peroxidase- or alkaline phosphatase-conjugated antibodies in PBS-Tween. The addition of 5-10% fetal calf serum to the blocking and/or the antibody solution may reduce nonspecific background reactions.
Horseradish peroxidase- or alkaline phosphatase-conjugated antirabbit IgG or conjugated antimouse immunoglobulin IgG are used as detection antibodies. After another three washes, antibody binding is visualized by chemilumi-nescence substrates, which emit light upon reduction by the enzyme. In earlier studies, we have employed the ECL system (Amersham), but obtained comparable results using other systems. The CDP-Star system (available from Tropix, Bedford, MA) has the advantage that the light emission lasts for a long time which improves the recording of gels by CCD-camera or chemiluminescent phosphor screen. Only clear and discrete banding signals of PrPSc should be recorded. Air bubbles during electrotransfer of the antigens result in uneven signals (holes) and render the results uninterpretable.
GLYCOTYPING
Choice of Samples
Homogenates, as well as SAF preparations, may be used for glycotyping studies, because comparable results were obtained using sheep scrapie samples (M. Groschup and J. Madec, personal observations). However, the choice of tissue samples may have an impact on the glycotyping results, although controversial results are reported in literature: while Somerville (25) saw differences in glycotypes in murine PrPSc derived from different brain areas, such effects were not observed by others (26-28). Substantial source-dependent effects may be seen, however, when PrPSc from peripheral tissues, such as spleen, tonsil, and uterus, are studied (29). It is therefore recommended that only PrPSc from similar tissue samples be compared.
PK Treatment
A number of different PK concentrations have been used, according to the literature, ranging between 10 and 100 ^g/mL (final concentration). In the Tuebingen laboratory, PK concentrations of 50 ^g/mL are generally used. PK is a rigid enzyme that survives repeated freezing thawing cycles, or prolonged storage at room temperature, without substantial loss of activity (30).
Samples are usually digested for 1 h at 37°C, although longer times or higher temperatures have also been reported. It is important to note that complete band shift in immunoblots of diglycosylated PrPSc to a band of molecular mass about 6-7 kDa lower should be achieved and monitored to ensure complete PK digestion. Incomplete cleavage and residual PrPSc shows as an extra band (cap) on top of the immunoblot signal of digested PrPSc. If there is residual uncleaved PrPSc, the cleavage conditions must be improved, to ensure complete cleavage, since a mixture of cleaved and uncleaved glycoforms in the stained PrPSc bands renders any glycotyping analysis impossible.
Immunoblotting
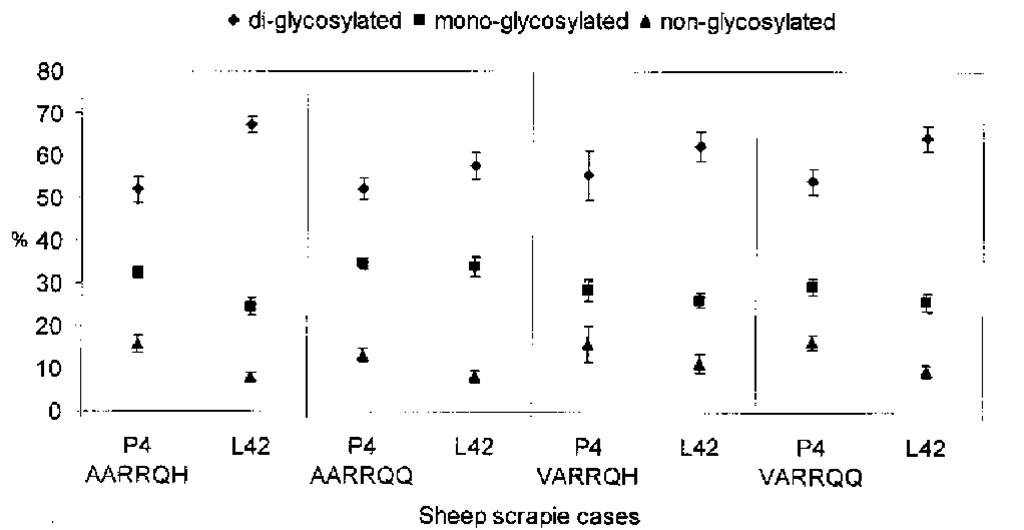
A number of different antibodies diluted in PBS-Tween at appropriate concentrations, can be used for the immunoblot detection of PrPSc. For labeling murine PrPSc we use a polyclonal anti-peptide antibody raised to the N- terminus of mouse PrP (amino acids 95 -110: THNQWNKPSKPKTNMK) (31). To detect hamster or human PrPSc, the monoclonal antibody 3F4, is used (32). Ovine and bovine PrPScs are immunostained by using the monoclonal antibodies mab P4 (amino acids 89-104: GGGGWGQGGSHSQWNK) or L42 (amino acids 145-163: GNDYEDRYYRENMYRYPNQ) (31,33). Mab L42 also reacts with human PrPSc. Apart from these antibodies, a large range of other mono-and polyclonal antibodies have been used in the literature (15,18-20,23). However, the choice of antibody can have a remarkable impact on the results obtained, thereby making comparisons of results between laboratories more difficult. As shown on Figure 1 mAb P4 yields profiles for PrPSc from ovine cases that are distinguishable from those of bovine cases; use of mAb L42 does not allow such a distinction.
Recording and Evaluation of Immunoblot Results
PrPSc protein patterns usually show the three typical PrP bands representing di-, mono-, and nonglycosylated PrP. Initial analysis relied on naked eye examination of blackened autoradiography films, which were exposed to light-emitting immunoblots.
Fig. 1. Proportions of PrPSc glycoforms in four different sheep genotypes after PK treatment. PrPSc was purified from sheep cerebellum homogenates. Fibrils were subsequently exposed to proteinase K (50 ^g/mL) for 1 h at 37°C. Proteins were separated by 16% SDS-PAGE gels. PrPSc bands were revealed by immunoblotting using the monoclonal antibodies, mAb P4 and L42, respectively. mAb P4 is directed to amino acid sequences 89-104 and mAb L42 to residues 145-163 of ovine PrP. Horseradish peroxidase-conjugated affinity-purified goat antimouse IgG served as detection antibody. Membranes were developed using a chemiluminescence enhancement kit, and signal scanned electronically. To glycotype PrPSc from isolates, banding intensities of the di-, mono- and non-glycosylated isoforms were determined and calculated as percentages of the total signal. The percentages of the di-, mono-, and nonglycosylated isoforms are represented as means of at least seven separate gel runs ± SEMs.
However, the limiting blackening capacity of such films may significantly distort the profiles obtained. Therefore, visual evaluation can easily lead to wrong interpretations, if samples are not applied in suitable and comparable amounts. This may also apply for profiles obtained by densitom-eter scanning of films. The suitable range of the detection and visualization systems used can be determined by comparing signal intensities of serially diluted samples. The discrimination power of electronic imaging devices is substantially greater than the ability of the naked eye for detecting strain-specific features. Electronic scanning of glycoprotein patterns should therefore be the technique of choice, because specific patterns can be distinguished over a much wider range.
Molecular Mass Determination
Choice of Tissue Source
As is the case for glycotyping, estimates of the molecular mass determined for PrPSc may vary, depending on the chosen tissue. Accordingly, only PrPSc coming from the same tissue source should be compared.
Enzyme Treatment
It is generally sufficient to digest PrPSc-containing samples such as brain homogenates from humans or sheep with PK (50-100 ^g/mL) for 1 h at 37°C. In some cases, an extension of this digestion time may be useful, in order to obtain a clear-cut band for nonglycosylated PrPSc. It is important to avoid EDTA and other ion-binding compounds during the sample preparation steps because these may affect PK cleavage sites, and lead to altered molecular masses of PK treated PrPSc.
In cases in which nonglycosylated PrPSccompounds are only scarcely detected by antibodies, proteins may be deglycosylated prior to immunoblot analysis. For this purpose samples are digested with 50 U/mL PNGaseF (available from Boehringer) for 6 h at 37°C. The reaction is terminated by heating to 95 °C for 15 min, and samples are stored at -20°C.
Gel Electrophoresis and Immunoblotting
To obtain a better separation, e.g., for molecular mass determinations, long maxigels of similar thickness may be used. The buffer system can also have an impact on the separation. Tris/Tricine gels are frequently used for the separation of peptides in the molecular mass range 0.5-40 kDa. However, in our experiments such gels performed no better than conventional Tris-HCl or Trisborate-buffered systems, for the separation of PrPSc bands. Moreover, because "smile" effects may happen in outer gel lanes, comparisons of molecular masses of bands should preferably only be made for antigens run on inner lanes of the gels.
In polyacrylamide gels, charged proteins migrate as they are pulled forward by the electric field, and their migratory speed generally correlates with their size (molecular mass). Depending on the extent of unfolding achieved by boiling in SDS-containing sample buffer, differences in their shape in the native state should be negligible. However, proteins may not be charged and unfolded to the same degree by the pretreatment. Moreover, sometimes they carry intrinsic charges because of their glycosylation, phosphorylation, glycosylphosphatidylinositol anchor attachment, or other posttranslational modifications, which possibly affect the SDS-PAGE results.
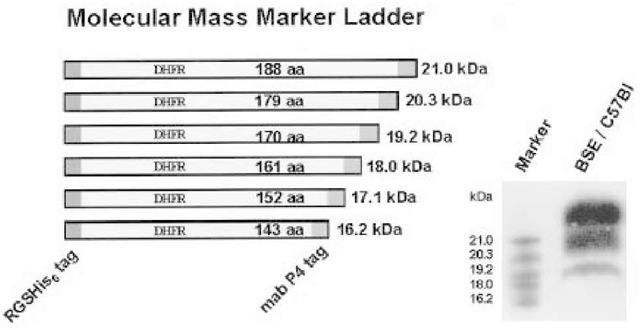
In order to better control the separation performance of the gel used, and to predict the distance in migration location, the authors have constructed a protein marker ladder in the molecular range 16-21 kDa. For this purpose, six deletion mutants of an open reading frame, encoding deletion mutants of dihydrofolate reductase were fused by a polymerase chain reaction with a sequence encoding the mAb P4 epitope (ovine PrP).
Fig. 2. A novel molecular mass marker ladder covering the range of 16-21 kDa was designed, which can be used in immunoblots for analyzing the size differences of proteinase K treated nonglycosylated PrPSc fragments. For this purpose, six open reading frames, encoding dihydrofolate reductase fragments in combination with two epitopes of well-characterized monoclonal antibodies (mAb P4 epitope; His-tag epitope), were expressed in Escherichia coli and purified markers mixed and separated on gels at appropriate concentrations. Markers are stained by conventional immunoblot detection, using either one of the monoclonal antibodies.
Moreover, the open reading frame also encoded a stretch of six histidines. After cloning into a bacterial expression vector, these marker proteins were produced and purified by metal-affinity chromatography. The set of six marker proteins can now be used as an antigen ladder of defined molecular masses: 16.2, 17.1, 18.0, 19.2, 20.3, and 21.0 kDa. Marker antigens are visualized in immunoblots, either by use of mAb P4 or an His-tag specific monoclonal antibody (Fig. 2).
Long-Term PK Resistance Analysis
Using SAF preparation protocols, PrPSc is purified from brain tissue samples from infected individuals. Alternatively, homogenates may be used. PrPSc is subsequently exposed for prolonged times (from 1 to 48 h) to PK (50 ^g/mL), in order to determine their relative proteolytic stabilities (Fig. 3). Nondegraded PrPSc compounds are visualized by immunoblot. The kind of antiserum used makes no difference. Residual antigen amounts are eventually quantified, using enhanced chemiluminescence and electronic imaging technique.
Fig. 3. Analysis of the long-term proteinase K (PK) resistance of mouse and hamster adapted scrapie strains (top) and sheep scrapie isolates (bottom). Strains ME7 and Chandler were propagated in C57BL/6 mice, and strain 263K in Syrian hamsters. Sheep PrPSc was isolated from natural infected sheep by scrapie. PrPSc was purified and fibrils were subsequently exposed for prolonged times (1, 3, 6, 24, 48 h [in the case of the strains]) and for 1, 6, 20 h ([in the case of the sheep isolates]) to PK (50 ^g/mL) at 37°C. After SDS-PAGE and immunoblotting protein bands were detected by using the polyclonal antibody Ra5/7 (mice), mAb 3F4 (Syrian hamster), and mAb P4 (sheep). Horseradish peroxidase-conjugated affinity-purified goat antimouse and goat antirabbit immunoglobulin IgG served as detection antibodies. Membranes were developed using a chemiluminescence enhancement kit, and residual PrPSc compounds recorded electronically. Signal intensities after digestion with PK for 1h were defined as 100% for each strain or isolate. Arithmetric means of at least four runs per sample were calculated. Standard error values are indicated.
Banding signal intensities of PrPSc after digestion with PK for 1 h are defined as 100%, in order to take account of possible interference effects of PK with other co-purified cellular proteins.
The residual PrPSc signal after 6 h of proteinase K exposure gives the most useful information when comparing PrPScs from different strains or origins.
General Comments
The molecular analysis of PrPSc by immunochemical techniques has allowed the characterization of prion strains, without the need for large-scale mouse inoculation experiments. Many prion strains/isolates can readily be discriminated by PrPSc glycotyping and molecular mass analysis. Another technique for the discrimination of prion strains is the analysis of long-term proteinase K resistance of PrPSc. In all these studies, the characteristics of PrPSc are examined by immunoblots to visualize the non-, mono-, and diglycosylated fractions following PK treatment. However, a variety of experimental factors may influence the results obtained in these experiments. The following rules should therefore be followed, if the interpretation of results on the immunochemical characteristics of PrPSc is to be reliable.
• All experiments should be performed under standardized conditions, and even minor changes in the experimental setup should be avoided as much as possible.
• Because the accuracy of an immunoblot detection may be affected by a variety of factors, substantial differences between PrPSc profiles from a given sample may be seen, even if all the above mentioned precautions are applied. Such deviations can only be standardized by repeated testing. In order to obtain representative results, each sample should therefore be electrophoresed and immunoblotted at least 4x and the overall result calculated on the basis of these individual results.
• Glycoform profiles of PrPSc derived from a given host species may only be comparable when they are obtained using the same antibody.
• Glycoform profiles of PrPSc derived from different host species may not be comparable, because detection antibodies can have a higher affinity for particular glycoforms of some species.
Taken together, PrPSc-based typing techniques can provide valuable information for the characterization of TSE cases, and for understanding molecular mechanisms involving different prion strains. Combined with the data on lesion profiles and incubation times in mice (as determined by the Neuropathogenesis Unit in Edinburgh), this information allows a better discrimination and definition of strains.
![Analysis of the long-term proteinase K (PK) resistance of mouse and hamster adapted scrapie strains (top) and sheep scrapie isolates (bottom). Strains ME7 and Chandler were propagated in C57BL/6 mice, and strain 263K in Syrian hamsters. Sheep PrPSc was isolated from natural infected sheep by scrapie. PrPSc was purified and fibrils were subsequently exposed for prolonged times (1, 3, 6, 24, 48 h [in the case of the strains]) and for 1, 6, 20 h ([in the case of the sheep isolates]) to PK (50 ^g/mL) at 37°C. After SDS-PAGE and immunoblotting protein bands were detected by using the polyclonal antibody Ra5/7 (mice), mAb 3F4 (Syrian hamster), and mAb P4 (sheep). Horseradish peroxidase-conjugated affinity-purified goat antimouse and goat antirabbit immunoglobulin IgG served as detection antibodies. Membranes were developed using a chemiluminescence enhancement kit, and residual PrPSc compounds recorded electronically. Signal intensities after digestion with PK for 1h were defined as 100% for each strain or isolate. Arithmetric means of at least four runs per sample were calculated. Standard error values are indicated. Analysis of the long-term proteinase K (PK) resistance of mouse and hamster adapted scrapie strains (top) and sheep scrapie isolates (bottom). Strains ME7 and Chandler were propagated in C57BL/6 mice, and strain 263K in Syrian hamsters. Sheep PrPSc was isolated from natural infected sheep by scrapie. PrPSc was purified and fibrils were subsequently exposed for prolonged times (1, 3, 6, 24, 48 h [in the case of the strains]) and for 1, 6, 20 h ([in the case of the sheep isolates]) to PK (50 ^g/mL) at 37°C. After SDS-PAGE and immunoblotting protein bands were detected by using the polyclonal antibody Ra5/7 (mice), mAb 3F4 (Syrian hamster), and mAb P4 (sheep). Horseradish peroxidase-conjugated affinity-purified goat antimouse and goat antirabbit immunoglobulin IgG served as detection antibodies. Membranes were developed using a chemiluminescence enhancement kit, and residual PrPSc compounds recorded electronically. Signal intensities after digestion with PK for 1h were defined as 100% for each strain or isolate. Arithmetric means of at least four runs per sample were calculated. Standard error values are indicated.](http://what-when-how.com/wp-content/uploads/2011/08/tmpD19_thumb.png)
