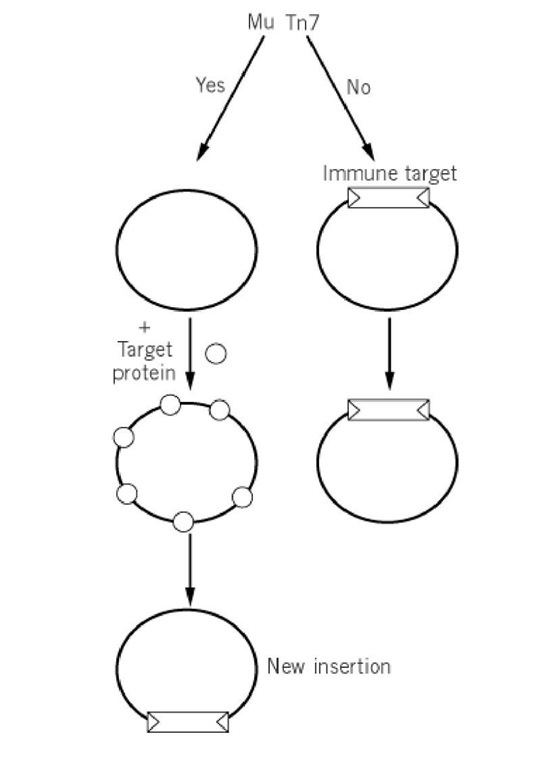
For some transposable elements, the frequency at which they insert into a target DNA that already contains a copy of the element is much reduced from their frequency of insertion into that same DNA lacking a copy of the transposable element (Fig. 1). This inhibition does not represent a global inhibition of transposition within the cell: The inhibition is specifically cis-acting on the DNA that contains the transposable element. This inhibition is called transposition immunity or target immunity. Although target immunity is usually evaluated in intermolecular transposition reactions, it is likely that a principal function of this phenomenon is to discourage intramolecular transposition— that is, to prevent the insertion of a mobile element into itself or into adjacent donor backbone. Target immunity has the effect of discouraging insertions locally and promoting insertion at a distance from the donor site of the mobile element .
Figure 1. Target immunity. For some elements such as Mu and Tn7, the frequency with which they insert into a target DNA already containing a copy of the element is much reduced. A target dNa already containing a copy of the transposon is a poor target because the target binding protein (small circle) is not effectively bound to the immune target. Binding of the target protein is discouraged by transposition proteins that bind specifically to the transposon ends in the immune target DNA.
Target immunity has been observed for a number of bacterial elements. The phenomenon was first observed with Tn3-like transposons and subsequently with bacteriophage Mu (1), and Tn7 (2, 3), a drug-resistance element. Phage Mu uses multiple rounds of transposition to set up replication forks that promote phage replication during lytic growth. Through transposition immunity, Mu ensures that new insertions made during the replication process will not be into extant copies of Mu, destroying these copies; thus transposition immunity facilitates Mu replication.
The mechanism of immunity has been dissected in vitro with Mu (4, 5), and Tn7 (6, 7). Common features of these elements are that the activity of the transposase is controlled by an ATP-dependent regulatory protein that interacts with the transposase and the target DNA via an ATP-dependent DNA-binding activity. Transposition occurs when the transposase interacts with this regulatory protein in an ATP-bound state on the target DNA. The ATP state of the regulatory protein is key to its activity, promoting transposition when it exists in an ATP-bound state on the target DNA and being inactive in a non-ATP state removed from the target DNA. As described below, the state of the ATP regulatory protein appears to be the key to immunity. A reasonable model is that the establishment of immunity involves discouraging the interaction of the ATP-dependent regulatory protein with the target DNA (Fig. 1).
In all systems examined, target immunity is imposed on a potential target DNA by the DNA sequence ends of the transposon—that is, the cis-acting recombination sequences at the ends of the transposon that are the transposase recognition sites. An attractive model for immunity is that when the transposase interacts with transposon end sequences on the potential target DNA, it exists on the target DNA in high local concentration and actively discourages the interaction of the ATP-dependent regulatory protein with the target DNA, thus effectively clearing the target DNA of targeting protein and discouraging transposition into that DNA. The ends of the transposon with bound transposase appear to interact with the target protein by simple protein-protein interactions facilitated by DNA looping (5, 7).
It seems likely that different forms of the transposase will interact with the target protein to execute transposition and transposition immunity. In Tn7, for example, immunity can be imposed by one of the protein components of the transposase, while actual transposition requires collaboration between both protein subunits of the transposase (7). In other cases, such as Mu, different forms of transposase, for example in different oligomeric states, may signal these different reactions.
Retroviruses also show a form of immunity in that intramolecular insertion is discouraged, although they show no inhibition for insertion into DNAs containing retroviral ends (8). The ability to block intramolecular insertion has been correlated with a requirement for a particular human protein (9), but the role of this protein and the mechanism of retroviral immunity remain to be established.