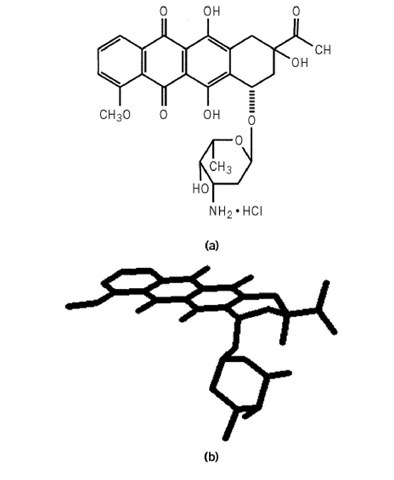
Daunomycin (synonym: daunorubicin) is the prototype of the clinically important anthracycline antibiotics that are widely used in cancer chemotherapy. Daunomycin and its close relative adriamycin are natural products isolated from various Streptomyces species. These two anthracyclines are probably the most widely used antitumor agents worldwide (1, 2). Daunomycin is primarily used in the treatment of acute leukemias. The structure of daunomycin is shown in Figure 1; it is composed of two major parts: an anthraquinone ring system and the pendant daunosamine, an amino sugar.
Figure 1. The chemical structure (a) and molecular model (b) of the anthracycline antibiotic daunomycin.
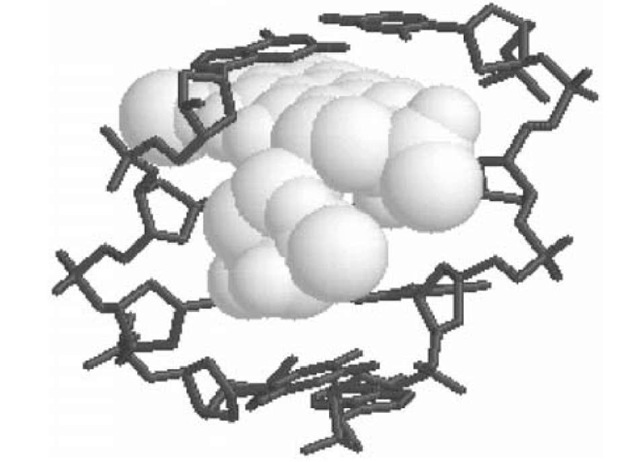
Daunomycin rapidly permeates into cells, passing through the membrane into the cytoplasm, and then accumulates in the nucleus, where it binds avidly to DNA. It binds to DNA by the process of intercalation, in which the anthraquinone ring inserts between adjacent DNA base pairs. The daunosamine moiety provides additional interactions that stabilize the complex by fitting into the minor groove of the DNA double helix, where it participates in van der Waals interactions. These additional interactions in the minor groove distinguish daunomycin from simple intercalators like ethidium bromide, which are stabilized only by the interactions within the intercalation site. Figure 2 shows the structure of a daunomycin-DNA complex determined by X-ray crystallography (3). Intercalation simultaneously lengthens and unwinds the DNA duplex, and it stabilizes DNA against thermal denaturation, increasing its Tm (melting temperature).
Figure 2. Structure of a daunomycin-DNA complex. The anthraquinone portion of daunomycin (shown in a spacefilling representation) is intercalated between the CpG step in a DNA oligonucleotide (shown in a stick representation). The daunosamine portion of daunomycin lies in the DNA minor groove.
Daunomycin, despite its small size (527.5 MW), exhibits several levels of molecular recognition (4). It is structurally specific and strongly prefers to bind to right-handed, B-form DNA over alternate helical forms. Daunomycin will allosterically convert left-handed Z DNA to an intercalated, righted-handed form (5). There is pronounced sequence selectivity in the binding of daunomcyin to DNA, which is revealed by DNase I footprinting experiments. Daunomycin binds preferentially to the triplet sequences 5 (A/T)GC and 5 (A/T)CG, where the notation (A/T) indicates that either A or T may occupy the sequence position (6, 7).
The mechanism by which daunomycin acts to kill cancer cells remains incompletely defined, and it may involve inhibition of several different cellular functions. Daunomycin, like most intercalators, is an effective inhibitor of DNA replication and of transcription, both in vitro and in vivo. The most likely intracellular target for daunomycin, according to recent evidence, is the enzyme topoisomerase II (see DNA Topology). Daunomycin poisons this key enzyme, by trapping an intermediate enzymatic species in which cleaved DNA strands are covalently attached to topoisomerase II, by preventing the normal resealing of the duplex DNA after strand passage (8). A comprehensive study of several anthracycline derivatives, including daunomycin, showed that intercalation is necessary, but not sufficient, for inhibition of topoisomerase II (9).
The anthraquinone moiety of daunomycin readily undergoes a number of oxidation-reduction reactions, with the production of oxygen free radicals. These free radicals can damage cells, and they may represent another important mechanism in the cytotoxicity of daunomycin. A serious, complicating side effect in the clinical use of daunomycin in cancer chemotherapy is its cardiotoxicity (1, 2). Free-radical production has been implicated as the primary cause of such cardiotoxicity.
Daunomycin was one of the first effective cancer chemotherapeutic agents identified, and it remains clinically important after over 30 years of use. It is one of the best characterized and understood intercalators and serves as an important model for understanding how small molecules bind to DNA.