Insect immunology is a rapidly maturing discipline, with exciting prospects for novel applications in control of vector-borne diseases and agricultural pests, biotechnology, and medicine. The outcome of the interactions between a parasite or pathogen and its host is determined by a combination of the susceptible vs. refractory status of the host and the virulant/avirulant characteristics of the infecting agent. Such a “pathogen offense/host defense” interface is a coevolutionary “arms race,” the outcome of which determines whether the invader is successful or whether robust immunocompetence of the host thwarts the infection. By manipulation with biotechnology, insect immune responses to parasitoids, pathogens, or symbionts, the vectors of plant and animal diseases can be disarmed, thereby curbing disease transmission. Similarly, the economic impact of agricultural pests can be reduced by enhancing pest susceptibility to pathogenic bacteria, viruses, and fungi, as well as development of biotechnological approaches to manipulating the host range of beneficial parasitoids.
In insects, both cellular and humoral immune responses figure prominently in host defense pathways, with many parasitic and pathogenic infections resulting in the deployment of both cellular and humoral defenses, which act synergistically. Insect hemocytes participate in phagocytosis, nodulation, and encapsulation processes. Humoral defenses include production of antibacterial, antifungal, and antiviral molecules, the melanizing enzyme phenoloxidase and its toxic reactive precursors, agglutinins, serpins, immulectins, and many more humoral mediators of immune defense.
Although much recent progress has been made in deciphering cellular and humoral responses and their regulation by signaling pathways, considerably less is known about the recognition mechanisms responsible for the initial discrimination of “nonself material that sets the stage for a defense response. The biology of insect pattern recognition processes and proteins is garnering increasing attention from
immunologists. This is especially true for the recognition of multicel-lular parasites including parasitoids, which exploit a combination of active and passive strategies to avoid being detected as foreign.
CELLULAR IMMUNE RESPONSES
Insect Hemocytes
Initially, hemocytes were classified on the basis of morphological criteria alone, resulting in the publication of numerous conflicting classification schemes, even for the same species. The different hemocyte types display marked inter- and even intraspecies variability in appearance and behavior, further complicating classification. Additionally, hemocytes that are examined before they have attached to a substrate are difficult to identify in the unspread state, in contrast to spread cells such as plasmatocytes which are readily recognized by their filamentous processes (Fig. 1). Today, tools such as monoclonal antibodies, which bind cell-type-specific epitopes, are used in combination with other biochemical markers to facilitate identification of the different hemocyte classes. Techniques such as fluorescence-activated cell sorting using antibodies or density gradient centrifugation have been employed to purify homogeneous populations of the individual hemocyte morphotypes, facilitating in vitro studies of their biochemistry and behavior.
In the Lepidoptera, the two most abundant hemocyte classes are plasmatocytes and granulocytes, which are the primary phagocytic and encapsulative cells. In the higher Diptera including Drosophila melanogaster, the multifunctional lamellocytes play these roles, and crystal cells carry the melanizing enzyme phenoloxidase. Taxonomic variations are seen in the total hemocyte count, with the Lepidoptera having abundant hemocytes in contrast to dipterans (e.g., mosquitoes), which have many fewer cells per microliter of hemolymph.
The fat body is also an immunoresponsive organ and synthesizes many antimicrobial peptides (defensins, attacins, and others), enzymes including lysozyme, eicosanoids, and other immune mediators. Indeed, the fat body represents the primary source of
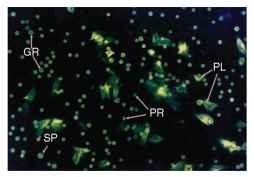
FIGURE 1 Light micrograph showing appearance of different hemocyte classes in hemolymph of a fourth instar nonparasit-ized tobacco hornworm larva. Prohemocytes (PR) are the smallest hemocytes seen in hemolymph preparations and are produced in hemopoietic organs localized in the thoracic region of tobacco horn-worm (Manduca sexta) larvae. These progenitor cells give rise to differentiated plasmatocytes (PL), granulocytes (GR) and spherule cells (SP). Maintenance of hemocyte populations in the larval stages of Lepidoptera occurs by a combination of proliferation of circulating cells and release of hemocytes from hematopoietic organs.
hemolymph-borne macromolecules and is the most metabolically active tissue in the insect. The gut, which is constantly assaulted with both pathogenic and nonpathogenic microbes ingested by the insect, also produces a battery of antimicrobial molecules to prevent the insect from succumbing to active infection. During molting, when the newly synthesized cuticle is most fragile and the cuticular linings of the fore- and hindguts are shed, enhanced production of antimicrobial peptides occur when the animal is most vulnerable to infection.
Hemocyte-Mediated Immune Responses
Phagocytosis is the process by which pathogens such as bacteria and small particles (< 1 |im in diameter) are engulfed byhost hemo-cytes, culminating in death of the invader. The membrane of the cell invaginates, and the pathogen is engulfed in a membrane-bound vesicle into which lytic enzymes are released causing the pathogen’s demise. This process appears to be mediated by prostagland-ins (eicosanoids) produced by the hemocytes. The phagocytic cells include plasmatocytes and granulocytes, although other cell types may also participate in this response to a lesser degree. Large numbers of bacteria-laden hemocytes may clump together to form nodules, which attach to the host’s internal tissues and are removed from circulation. Frequently these nodules are melanized and deposited on lobes of fat body, the Malpighian tubules, or gut tissues.
To counter eukaryotic invaders that are too large to be phagocy-tosed by a single cell, several classes of hemocytes cooperate in the mobilization of the multiphasic encapsulation reaction. Usually the capsule is formed from several hundred to several thousand cells that form tight layers enclosing the parasite. In the initial phases of encapsulation of parasitoid eggs or larvae, or encapsulation of abiotic implants such as Sephadex or latex beads, granulocytes are the first cells to make contact with the target. They attach to the target surface and release their contents in a degranulation reaction, forming a sticky matrix on the surface of the target which induces deposition of multiple layers of plasmatocytes that comprise the bulk of the capsule. The encapsulation reaction is terminated by the adherence of additional granular cells to form a thin envelope around the capsule. Mature capsules frequently undergo melanization; they may adhere to the host’s internal tissues including lobes of fat body, Malpighian tubules, salivary glands, or the gut and are thereby removed from circulation. In rare instances, the encapsulated parasite may actually be extruded from the hemocoel and pass through the epidermis, to be shed with the host’s exuvial cuticle during ecdysis in a molt. This phenomenon is termed “cuticular encystment,” and this process sometimes occurs when a parasitoid develops in a nonpermissive host.
In some insects with encapsulated parasitoids, usually in cases in which the host species or strain is completely refractory to that para-sitoid, the host can undergo successful metamorphosis and survive to the adult stage. In other host-parasitoid combinations, the host dies prematurely following encapsulation of parasitoid eggs or larvae, frequently showing symptoms of developmental disruption due to endocrine imbalance. When the host dies prematurely, the parasitoid also fails to survive and the reproductive fitness of both partners is zero.
Endoparasitoids belonging to the hymenopteran families Braconidae and Ichneumonidae inject symbiotic viruses termed polydnaviruses (poly-disperse DNA viruses or PDVs) that subsequently play a critical role in suppressing the host immune response so that successful development of the parasitoid can proceed. These viruses, which are integrated in the genomic DNA of the wasp and undergo replication only in the female’s ovary, rapidly enter host hemocytes, fat body, and nervous system following parasitization,and viral genes are expressed. Depending on the host-parasitoid combination, the host’s hemocytes either alter their behavior and fail to spread (thereby inhibiting the encapsulation response) or, alternatively, undergo programmed cell death and apoptosis. In Manduca sexta larvae parasitized by Cotesia congregata, massive numbers of dead and dying hemocytes undergo clumping and then are removed from circulation soon after parasitization, resulting in a dramatic drop in the host’s total hemocyte count. The final biochemical events that culminate in death of the parasite remain unclear for the most part. Although melanin and its precursors are toxic, much recent evidence points to the pivotal role of other toxic molecules such as reactive intermediates of nitrogen (nitric oxide) or oxygen (superoxide), released by cells localized in the innermost layers of the capsule, in causing lethality. Although it was formerly presumed that death of the parasite/parasitoid was induced by asphyxiation inside the capsule, biochemically mediated parasite-killing strategies are now recognized as causing the ultimate death of the invader. Phagocytosis is followed by the generation of these cytotoxic intermediates in the phagolysozome, indicating that this pathway is also important to the function of phagocytic cells.
HUMORAL IMMUNE RESPONSES
In addition to hemocyte-mediated immune reactions, insects possess a variety of potent plasma-borne defense molecules that are toxic to parasites and pathogens. Usually these are synthesized by the fat body or the hemocytes and secreted into the plasma, where they act either on the invader directly or via the hemocytes in altering their behavior to enhance the immune response. A battery of antibacterial proteins are produced by many insects, including defensins, drosocin, cecropins, attacins, and diptericins, depending on the species, and, in addition, the ubiquitous lysozyme family of antibacterial proteins. These proteins differ in their specificity for gram-positive vs. gram-negative bacteria, with some acting on both types of bacteria with varying degrees of potency. Often the proteins disrupt the bacterial cell membrane function by inducing pore formation, causing lysis of the cell. Antifungal molecules are also produced by insects, providing a first line of defense against fungal invaders that often infect the insect via penetration of the cuticle. Antiviral responses are emerging as a new subdiscipline in insect immunity (see below).
Many antimicrobial peptides also exhibit effective antiparasite activity against Plasmodium, filarial nematodes, and other harmful eukaryotic species. Microarray analyses have also confirmed that up-regulation of the same or slightly different suites of AMPs by pathogen vs. parasitic organisms provides a window of opportunity to develop strategies to stop transmission of both classes of infectious agents by enhancing AMP production in transgenic mosquitoes by activating the signaling pathways which induce their synthesis.
Dramatic recent progress has been made in deciphering the biochemically complex phenoloxidase pathway leading to melani-zation of nodules and capsules. The pathway begins when tyrosine is converted to DOPA via the action of the monophenoloxidase enzyme (also called tyrosinase). DOPA is then converted to DOPA quinone by diphenoloxidase and thence to melanin via a series of toxic intermediates. Phenoloxidase precursors (prepro- and pro-phenoloxidase) may be associated with hemocytes, but these enzymes can also circulate freely in plasma before they are activated by serine proteases and peptide fragments are removed from the precurser molecules.
In many host-parasitoid systems, including M. sexta larvae parasitized by C. congregata, levels of hemolymph phenoloxidase activity have been found to be suppressed, which benefits the parasitoid by inhibiting melanization of the egg and/or larva. Expression of polydnavi-rus gene products that inhibit translation of phenoloxidase mRNA, thereby suppressing levels of this enzyme in the blood, occurs in several species.
In refractory strains of mosquitoes, melanization of the malaria ookinete occurs in the midgut wall to form Ross’s black spores in the midgut epithelium, which were recognized early on by Ronald Ross before the entire life cycle of the malaria parasite was known. Disease transmission stops because the melanized arasites die trapped in the gut wall, short-circuiting their entry into the hemocoel and subsequent movement to the lumen of the salivary gland. With the advent of improvements in mosquito transformation technology, medical entomologists sought to use phenoloxidase genes to bioen-gineer refractory transgenic mosquitoes using transposable elements to halt malaria transmission by the insect vector. One approach has been to link this gene to highly active promoters, particularly those which are activated when the mosquito takes a blood meal in preparation for production of eggs including the vitellogenin promoter. New strategies and constructs may eventually fulfill the promise offered by biotechnological approaches for control of major scourges like malaria, yellow fever, and dengue. However, there will be a need to overcome regulatory hurdles and public attitudes toward biotechnology in challenging environments such as Africa comprised of many countries with different cultures.
IMMUNITY MODELS
Insects exhibit both innate and inducible immunity to parasites and pathogens. Several insects such as the fruitfly D. melanogaster, the tobacco hornworm, M. sexta, and mosquitoes including the malaria vector Anopheles gambiae, have served as exemplary model systems in insect immunology. The Tribolium beetle is also gaining ground as a model system for immunological research. The wealth of genomic and genetic information available for Drosophila has rapidly accelerated progress in the areas of immune recognition, antimicrobial mechanisms and molecules, and signaling pathways. Many parallels between Drosophila immune mechanisms and mammalian immune pathways link these distantly related phyla and provide strong arguments to support the use of Drosophila as a mammalian immunological model. The concept that some genes and their protein products play dual roles in immunity and development was first discovered by immunologists studying the fruitfly. Conversely, studies of mammalian immune specificity and memory have generated novel insights into how insect immune systems function despite the lack of production of antibodies targeted to specific epitopes. Our extensive knowledge of the physiological factors regulating immune pathways in M. sexta, Bombyx mori, and other lepidopter-ans, together with the relative ease of isolation and sequencing of immune peptides, enzymes, and other biochemical modulators from hemolymph and tissues of large species, complements the enormous genomic and proetomic databank now available for many species.
TECHNOLOGIES FOR MANIPULATING IMMUNITY
Major recent advances in mosquito genomics, transformation, and immunity are facilitating development of new application technologies to achieve enhancement of insect immunity to vector-borne parasites and pathogens with the goal of reducing rates of human disease transmission. Both transgenic and new genetic approaches to manipulation of insect immunity offer tremendous potential advantages as well as challenges in disease vector management. Controlling insect pests via genetic manipulation of symbiotic partners including bacteria also offers exciting prospects for the future.
Recent dramatic advances in the fields of bioinformatics, micro-array analysis, and proteomics are facilitating rapid breakthroughs in both basic insect science and its applications to health and agriculture. New gene silencing strategies that shut down production of immune-related gene products such as RNA interference (RNAi) are now being applied across many insect taxa, yielding fruitful insights into the roles of homologous gene products in diverse phyla.
SIGNALING PATHWAYS
Multiple signaling pathways including the JAK/STAT, immune deficiency (imd), and Toll pathways are now recognized as critical players in mobilizing defenses against pathogens and parasites. These pathways play key roles in activating both humoral and cellular mechanisms of immunity. The pattern recognition molecules and mechanisms giving rise to basic discrimination between nonself and self is a subject at the forefront of current insect immunity research. Increasing attention has focused in the past 5 years on identification of the molecular triggers of effective host defense responses that occur at the initial confrontation between pathogen and host. One proposed idea is to develop strategies to interfere with binding of peptidoglycan recognition proteins to their respective receptors.
NEW VISTAS IN IMMUNOLOGY EVOLUTIONARY IMMUNOLOGY
Evolutionary immunology and the interface between immunity and ecology is a rapidly expanding subdiscipline, posing intriguing questions regarding the physiological “costs” of immunity. The extent to which enhanced levels of immunocompetence are offset by negative impacts on longevity, mating success, reproductive output, and fitness clearly must be considered in developing new technologies to manipulate refractoriness to animal or plant pathogens. Indeed, some recent studies show that there are indeed “metabolic fitness impacts” of launching an immune response on longevity and reproduction. Holistic approaches to analysis of how immunological constraints influence ecological inter- and intraspecies interactions are yielding insights into how physiological “tradeoffs” impact the links between immunity, behavior, and fitness. Framing immunity in an evolutionary context is a concept that has evolved during the past 5 years and is fostering collaborations among physiologists, biochemists, molecular biologists, ethologists, ecologists, and evolutionary biologists who pose questions about the metabolic price of immunity and its impacts on insect life history. Given that enhancing effects of nutrient deprivation and starvation on insect susceptibility to disease has been long recognized, it is not surprising to find that resistance requires metabolic resources that would normally fuel other physiological processes including reproduction.
ANTIVIRAL IMMUNITY
In contrast to the huge databank of information available about antibacterial peptide chemistry and regulation, the mechanisms of insect immunity to viruses have only just begun to be explored. Baculoviruses, many of which attack agriculturally important pest species, must be ingested by the insect and then cross the gut wall to enter the hemocoel and initiate successful infection in target tissues, replicate, and complete their life cycle in the host. As seen in Fig. 2, species-specific genetic factors regulate temporal patterns of virus (i.e., Rachiplusia ou multiple nucleopolyhedrovirus) entry into target tissues including the tracheolar epithelium, hemocytes, fat body, and gut. Genetic factors also influence rates of virus propagation in different hosts. Latent infections can occur with viruses that initially remain cryptic but ultimately erupt to epizootic levels when the
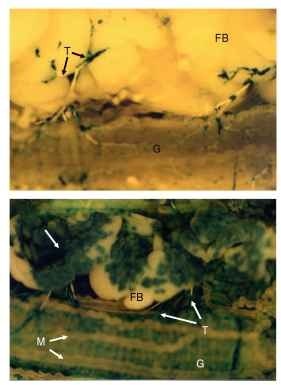
FIGURE 2 Patterns of virion distribution in host tissues following baculovirus (Rachiplusia ou multiple nucleopolyhedrovirus) infection of semi-permissive and permissive lepidopteran host species. Larvae of the European corn borer Ostrinia nubilalis, a semi-permissive host (upper panel), and Heliothis virescens, a permissive host (lower panel) dissected 72 hours after infection with Rachiplusia ou multiple nucleopolyhedrovirus expressing (3-galactosidase under the control of the hsp70 promoter. Infection of H. virescens is significantly more advanced with extensive lacZ blue staining indicative of infection in the tracheoles (T), fat body (FB, arrow), and gut epithelium (G) in permissive H. virescens larva (lower panel). Virus distribution in semi-permissive O. nubilalis is restricted to foci of infection in these tissues (upper panel). The Malpighian tubules (M) are also shown.
host is physiologically stressed. Disruption of “successful” interactions between arboviruses and vector species is clearly promising and likely to be a highly effective strategy to curb disease transmission.
DEVELOPMENTAL AND HORMONAL REGULATION OF IMMUNITY
Isolated reports of hormonal influences on insect immunity have peppered the insect literature for decades, and strong experimental support linking hormones to immune regulatory pathways has now materialized. Endocrine impacts on immunity are well established in the vertebrate realm, with long-standing appreciation of the roles of testosterone, corticosteroids, and other hormones on mammalian immunity. The observation that in many larval-stage insects immunity to parasitoids and pathogens is significantly enhanced in late larval development when the animal approaches pupation has been experimentally documented to be due to the decreasing levels of juvenile hormone (JH) associated with the last instar. The concept that juvenile hormone and 20-hydroxyecdysone (20E) could have antagonistic effects on the induction of antimicrobial peptide gene expression was recently experimentally tested in Drosophila using both i n vivo microarray experiments and i n vitro approaches with cell cultures maintained under different hormone regimens. JH was shown to be a potent hormonal immuno-suppressor, whereas 20E functions as an immune potentiator, at least with respect to antimicrobial peptide synthesis. More research to investigate roles of hormones in modulating insect immunity is obviously needed, as findings may have impacts on potential usages of hormonally-based insect growth regulators to render pests more susceptible to pathogens, or conversely, more refractory to parasites (e.g., Plasmodium).
Surprisingly, even embryonic-stage hosts can mount effective encapsulation responses against parasitoids, indicating that hemocytes are differentiated by hemopoietic tissues very early during development and are fully functional even prior to eclosion of the first instar larva. The encapsulation response seen in egg-stage eucalyptus borers against eggs of an encyrtid wasp parasitoid is illustrated in Fig. 3 . Production of antibacterial proteins by insect embryos and extraembryonic tissues has also been documented. These findings document the ability of insect embryos previously thought to be incapable of mobilizing immune responses to be immunocompetent even while the egg is protected by
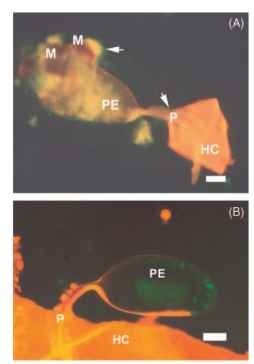
FIGURE 3 Appearance of encapsulated parasitoid egg in non-permissive embryonic stage host eucalyptus longborer Phoracantha recurva egg. Fluorescence micrographs showing encapsulation response mounted by eucalyptus longborer P. recurva host egg against the egg of the encyrtid parasitoid Avetianella longoi (A) vs. appearance of an nonencapsulated parasitoid egg in the permissive host P. semipunctata (B). Arrows indicate areas of host hemocyte attachment in (A). Eggs were stained with the vital dyes ethidium bromide and acridine orange. Note that living cells fluoresced green but changed color (yellow to orange/red) as layers of cells were deposited. The innermost cells of the capsule flattened and released components of the phenoloxidase cascade, melanized and died. The hemocyte-free surface of the parasitoid egg in the permissive host P. semipunctata is seen in (B). HC = host egg chorion, M = melanization, P = parasitoid egg pedicel, and PE = parasitoid egg.
its chorion. Whether hormonal factors can modulate immune responses in developing insect embryos or regulate differentiating pathways during hematopoiesis has yet to be addressed.
IMPACTS ON AGRICULTURE, AND HUMAN AND ANIMAL HEALTH
Another novel area with potential for economically important impacts is the exploitation of the immune-disrupting genes evolved by many insect-specific microbes as well as the para-sitoid-associated symbiotic polydnaviruses to develop hybrid biopes-ticides, with enhanced virulence to more effectively kill insect pests that are particular scourges in agriculture, forestry, and human and plant disease transmission. Polydnavirus genes, including several ichno- and bracovirus genes, are also being exploited in the medical realm as valuable technological tools that enhance levels of expression of baculovirus-promoter-driven pharmaceutical agents, as well as in transgenic plants to suppress insect feeding activity and development. Insect antifungal defensins also offer therapeutic potential for use in treatment of human pathogens. Paratransgenic technology in which genetically modified insect symbionts are induced to express potent antiparasite or antipathogen compounds are being implemented in control of human/animal disease vectors to block disease transmission as well as in economically important agricultural pests and plants.
