Insects are acutely aware of many aspects of their environment, as anyone knows who has tried to catch a fly perched on a slice of pizza. In the chemical realm, and depending on the chemicals and insects involved, insects are often outstandingly sensitive. The most famous and best-studied aspects of chemoreception in insects are mate recognition and finding. Like many, if not most animals, insects produce chemicals called pheromones that allow individuals of one sex in a species to recognize and find individuals of the opposite sex. Usually the female produces a mixture of chemicals to which the male responds. Other important, life-or-death decisions largely based on chemicals include choice of site for egg laying, decisions about what to eat and what to avoid, and communications about immediate danger. How insects taste and smell is therefore of great interest and, given that many insects are serious agricultural pests and vectors of disease, research in this area is both fundamental and practical.
As with most physiological systems, model animals are vitally important for scientists who explore the specific workings of what is always a complex series of interactions. For studies of insect chemoreception, adult moths and caterpillars, flies, cockroaches, and leaf beetles have provided some of the best models. Large moths such as the silkworm, Bombyx mori, and the tobacco hornworm, Manduca sexta, have been essential in studies of pheromones, whereas flies such as the black blowfly, Phormia regina, caterpillars such as the cabbage butterfly, Pieris brassicae, and M. sexta, and beetles such as the Colorado potato beetle, Leptinotarsa decemlineata, have helped unravel the role of chemoreception in food and oviposition-related behavior.
For an insect to sense and respond appropriately to the presence of a chemical, or more often a mixture of chemicals, requires a large number of cuticular, cellular, and molecular processes. Because insects are covered in cuticle, it is appropriate to begin there. The cells involved include the sensory cells themselves and closely associated accessory cells whereas the molecules include a wide array of extracellular, intracellular, and membrane-bound proteins. The processes involved in tasting and smelling include sampling the environment, transport of stimulus molecules to receptors, reception, transduction, coding, and transmission to the higher brain centers. This article looks at both gustation (taste) and olfaction (smell), though in many areas detailed knowledge is more complete for smell than it is for taste.
ROLE OF CUTICLE IN TASTE AND SMELL
Insects, like all arthropods, are covered with a chitin-protein complex called cuticle, which in turn is covered with wax to prevent desiccation. For the creature to taste or smell anything, there must be a pathway from the outside to the sensory cells inside. On various parts of the insect body, but particularly on the antennae, mouthparts, legs, and ovipositor (egg-laying structure), insects possess a variety of cutic-ular elaborations in which are housed chemically sensitive cells. These cuticular structures take the form of hairs (trichoids), pegs, pegs in pits, flat surfaces, and several other shapes. Common to them all is a modified cuticular region that will provide one or more pores through which chemicals can gain entrance. For water conservation, and to keep the important sensory cells functional, these pores cannot allow direct contact of the sensory cell membrane with air. All these pores are small (in the submicrometer range), and there is always a water-protein pathway from the pore to the cell membrane. The cuticular structures plus the associated cells collectively are referred to as sensilla.
Figure 1 represents a reconstruction of a typical mouthpart gustatory sensillum in a caterpillar. All caterpillars so far investigated have this type of sensillum, and it is always important in the food selection processes. The reconstruction is based on careful observations of hundreds of images taken with the electron microscope. The cellular details shown in the drawing cannot be seen with the light microscope. Most of the parts of this drawing below the cuticle could be mistaken for those in an olfactory sensillum. This is because chemo-sensory cells in insect sensilla are modified cilia and the accessory cells are also basically the same in both types. This involvement of cilia is not surprising, because most of the sensory cells of animals, including light, touch, and hearing, as well as chemical sensors, are modified cilia. Only the sensory cells are modified cilia. The accessory cells are more ordinary, although still specialized, epidermal cells, and they have two very different functions. During the development of a sensil-lum (i.e., between molts) these cells are involved in secreting all the
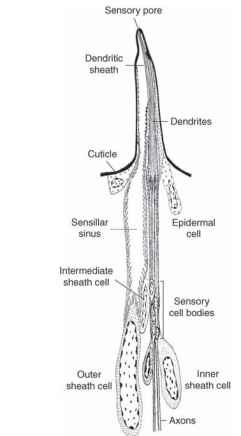
FIGURE 1 Reconstruction of a taste sensillum of the type typically found on the mouthparts of caterpillars. Associated with the maxilla there are four such sensilla, each with four gustatory cells, and it is clear that caterpillars rely heavily on the information provided by the cells to make food choices. The cuticular modification, accessory cells, and sensory cells are all necessary for the sensillum to function properly. In addition to providing the sense of taste, these sensilla are also sensitive to touch.
cuticular elements of the sensillum, including the base, the shaft, the cuticular pore or pores, and the dendritic sheath surrounding the den-drites (above the ciliary rootlets) of the sensory cells. Once the dendritic sheath is in place, the dendrites are physically separated from the rest of the sensillum lumen, though chemicals can pass through. The dendritic sheath is much longer in taste sensilla, as depicted in Fig. 1, running all the way to the single pore in the tip. The dendritic sheath in olfactory sensilla stops nearer the base of the sensillum, and the dendrites are free in the lumen. In both types, the dendritic sheath provides mechanical stabilization for the sensory cells. When development is complete, the accessory cells provide the particular chemical ionic mix that surrounds the dendrites (note the microvilli in the outer sheath cell). The fluid surrounding the dendrites is very different from the general body fluid (hemolymph), and its high cation concentration is critical in allowing the cell to signal its contact with an appropriate chemical stimulus. This signal is in the form of a potential change across the dendritic cell membrane that is eventually turned into normal action potentials near the sensory cell body.
The structural features discussed so far are shared by olfactory and gustatory sensilla. The major differences between the two types have to do with the way chemicals get into the system and the underlying cuticular modifications. Chemicals enter gustatory sen-silla via the single pore in the tip. This pore contains a sugar-protein complex (mucopolysaccharide) that protects the dendrites from desiccation and probably limits the types of chemicals that can pass (though this latter point is in need of further study). Once past this barrier, the chemical enters the solution around the dendrites and potentially can interact with the cell. Olfactory sensilla typically have many pores, and they are different in origin from those in gustatory sensilla. Figure 2 illustrates a section of a typical olfactory sensillum
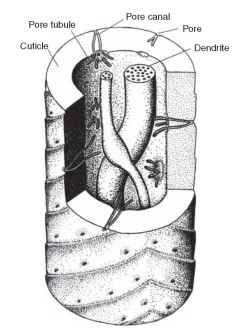
FIGURE 2 Schematic view of a section of a pheromone sensillum in a moth. The features are those revealed in an electron microscopic examination. Olfactory sensilla may have as few as two sensory den-drites, as here, or many more. The arrangement shown is typical of many moth pheromone sensilla.
from the pheromone system of a moth. To understand the nature of the numerous pores on this structure requires knowledge of insect cuticle in general. The surface of insect cuticle is in constant communication with the inside of the animal for the purpose of wax renewal. This communication is provided by numerous pore canals, microscopic and tortuous passages through the cuticle. These canals are filled with a water-wax mix. In olfactory sensilla, the pore canals are taken over for the function of providing access of stimuli to the sensory dendrites. On the inside end of some pore canals are structures called pore tubules; these delicate structures can be seen only in electron micrographs of carefully prepared tissue. It was once thought that pore tubules provided a hydrophobic route for odor molecules to pass from the outside waxy surface of the sensillum to the surface of the dendrite (which is surrounded by water and salts). Discovery of additional molecular components of this system replaced this long-standing and attractive hypothesis.
THE ODOR PATH
Substances animals taste are usually much more water soluble than those that they smell, and the sensory dendrites of both gustatory and olfactory sensilla are in an aqueous medium. Thus, the problem of getting the stimulus to the receptor has received much more attention in olfactory research. In insects, odor molecules first contact the cuticular surface, and because it is waxy, they easily dissolve. From here they move in two dimensions, and some find their way into the opening of a pore canal. Since the pore canal contains wax, passage through it is probably easy, and passage in pore tubules may be similar. Eventually, however, before it arrives at the receptor surface of a dendrite, the hydrophobic odor molecule will encounter water. Recent work, particularly with the antennae of large moths, has uncovered at least two types of protein in the extracellular spaces of sensilla. One type specifically binds chemicals that are part of the moths’ pheromone mixture, and are therefore called pherom-one binding proteins (PBP). The other type binds less specifically a variety of nonpheromone molecules (e.g., food odors) and are called general odorant binding proteins (GOBP).
The odorant binding proteins (OBPs) act as shuttles and carry odor molecules through the aqueous medium to the surface of the dendrite. In the membrane of the sensory cell are receptors for various odors, depending on the specificity of the cell. Cells that respond to only a single pheromone would be expected to have only one type of receptor molecule. More typically, a cell that is sensitive to food odors has a variety of related receptors covering various stimuli. In either case, the OBP, now carrying the odor molecule, comes in contact with a receptor. What happens next is now under investigation, and there are two competing hypotheses. The OBP may simply deliver the stimulus, which itself then interacts with the receptor protein; or, the stimulus-OBP complex may be the actual stimulus. That is, the receptor site may be configured as to recognize only the combined stimulus and OBP; either alone will not fit. The latter hypothesis may also explain how these systems can turn on and off so quickly: namely, because moths can follow a discontinuous (patchy) odor trail, making minute adjustments in flight pattern on a millisecond scale. This precise behavior is corroborated by electrophysi-ological measurements showing that the sensory cells can follow an on-and-off pattern of odor stimulation, also in the millisecond range. It is possible that the OBP-stimulus complex, when first formed, is the effective stimulus for the receptor. During the interaction with the receptor, however, the OBP-stimulus complex changes slightly, becomes inactive, and immediately leaves the receptor. Later, it is broken down by other proteins (enzymes) in the sensillum lumen. Figure 3 summarizes this complex series of events and emphasizes the second hypothesis.
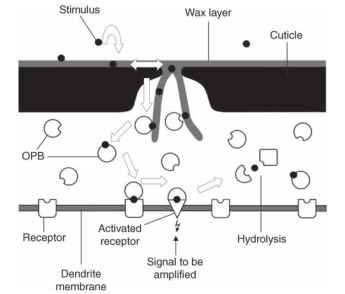
FIGURE 3 Schematic summary of the movement (arrows) of an odor molecule (solid circles) from the surface of a sensillum to the dendritic membrane. Specialized proteins (various shapes) act sequentially as carriers, receptors, and hydrolytic agents to make precise detection of the odorant possible. See text for details.
CHEMICAL-TO-ELECTRICAL TRANSDUCTION
In almost all studies of animal sensory systems, the stimulus being sensed is in a different energetic form than the chemoelectrical transmission used by the nervous system. Thus, in eyes, light (photon) energy needs to be transduced into chemoelectrical energy via photo pigments. Similarly, with a chemical stimulus-receptor complex, once binding between stimulus and receptor has occurred, the event must be communicated to other parts of the sensory cell to ensure that the end result is a message, composed of action potentials, transmitted to the brain. Understanding of chemical transduction in insects is far enough along to permit the statement that the basic elements are probably very much like the arrangement in the vertebrates. There will be differences in detail, but these will continue to be the subjects of active research for some time. Basically, most chemotransduction requires (1) a more or less specific receptor molecule (thus the stimulus-receptor complex can be formed), (2) an amplification step (involving a series of membrane-bound and intracellular molecules) that turns a few stimulus-receptor events into a significant, momentary elevation of some chemical (often calcium) inside the cell, (3) at least one ion channel that senses the rise in calcium and opens, allowing depolarization, and (4) a braking (deactivation) system, composed of more molecular interactions, so the system can be precisely controlled.
Parts of a complete transduction system are beginning to emerge from electrophysiological (patch-clamp) studies of cultured olfactory cells, pharmocological experiments on these cells and on whole-sensillum studies of fly taste sensilla, and from genetic work with Drosophila fruit
flies. The fruit fly work used specific searches of the now complete D. melanogaster genetic database to find some likely candidates for sugar receptor proteins. Carlson has used this information to make specific fluorescent probes, and some of these probes bound only with cells in gustatory sensilla. Combinations of genetic analysis, molecular biology, electrophysiology, and pharmacology will be needed to define all the necessary components.
CHEMOSENSORY CODING
AT THE PERIPHERY
In the real world, animals encounter thousands of chemicals. Most of these are meaningless, in the sense that no behavioral response is required, whereas some are critical. A sensory system thus serves two opposing functions. First, the effective sensory system must act as a filter, allowing the animal to ignore most potential stimuli so that it can concentrate on the important ones. Second, the same system must be sensitive, sometimes exquisitely sensitive, to biologically relevant stimuli and must continuously transmit a “summary” report to the brain or central nervous system. The receptor proteins and associated transduc-tion molecules provide the specificity (only some things are adequate stimuli) and sensitivity (the effectiveness of the amplification step in transduction). The nature of the summary neural message is the problem addressed in studies of peripheral sensory coding. In insect chem-osensory coding, the problem can be as simple as a few highly specific receptor proteins recognizing a three- or four-component blend of pheromone molecules all housed on a pair of cells found in each of many thousand antennal sensilla. At the other extreme, a leaf beetle may be faced with a food choice of two closely related plants, each with many chemicals to which its tens of gustatory cells are capable of responding. When one is comparing these two scenarios, it is not the number of sensory cells that constitutes the relative scale of the coding problem, but the number of chemical compounds that can be sensed by these cells, and the combinations of compounds that are possible.
In the pheromone example, there are two cell types (each sensil-lum has one of each type). They respond differently to, for example, four pheromone molecules and not much else. Also, one or two of the pheromone molecules may be completely nonstimulatory to one of the two cells. In addition, only two of the four compounds in the blend may be sufficient to stimulate a full array of behaviors necessary for the male to find the female. The coding problem, though overly simplified to make the point, could thus be reduced to the following: cell A responds only to compound A, and cell B responds only to compound B. Both cells continuously signal to the antennal lobe the levels of compounds A and B detected in the air. If cell A is firing at twice the rate of cell B and both cells are firing at some rate, then the moth flies upwind. Thus the code is a simple comparison, and the large number of cells involved is a kind of amplifier, reflecting the overwhelming importance of the pheromone system to the animal. The two cells, A and B in this example, can be thought of as labeled lines, each sending unique information about the concentration of compound A or B. The central nervous system uses a simple hardwired rule to compare this paired input, and, accordingly, behavior is or is not released.
The beetle, potentially, has a more difficult coding problem. Many experiments have shown that gustatory cells of plant-feeding insects are affected by numerous single plant compounds. Ubiquitous compounds such as water, salts, amino acids, and sugars are sensed by some cells on the mouthparts of all such insects. Less widely distributed chemicals, such as alkaloids, terpenes, glucosinolates, and other so-called secondary plant compounds, are stimuli for cells that are variously scattered throughout the class Insecta. To exemplify this coding problem, consider a Colorado potato beetle facing the choice of a potato leaf (host plant) or a tomato leaf (marginal host) (Fig. 4A ). The gustatory cells in the beetle’s mouthpart sensilla (on the galea) are all sensitive to different compounds. Both direct stimulation by some molecules and inhibition of one molecule by another are known, as are some injury effects in the presence of too much glycoalkaloid (compounds in potatoes and tomatoes). Not surprisingly, the summary of report such a four-cell system sends to the brain comprises two kinds of message, one for potato and the other for tomato (Fig. 4B).
![(A) Summary of the behaviors exhibited by newly emerged Colorado potato beetle adults when provided with either potato (host plant) or tomato (nonhost plant); numbers of beetles indicated inside heavy arrows. Beetles first examine the leaf and then they squeeze it between their mandibles (macerate) before taking a small bite, which they taste for only a short time. If the plant is acceptable, they very quickly move to sustained feeding. If the plant is less acceptable, few beetles will feed. The decision to not feed is made after considerable time has been spent in examining, macerating, taking small bites, and sometimes repeating one or more of these steps. [Modified from Harrison, G. D., (1987). Host-plant discrimination and evolution of feeding preferences in the Colorado potato beetle, Leptinotarsa decemlineata. Physiol. Entomol. 12, 407-415.] (B) Taste sensilla are important in making the kinds of decisions shown in (A). If potato leaf juice is the stimulus, four cells in nine sensilla on the mouthparts respond by sending a clear, almost labeled-line (cell 1), message to the central nervous system. When tomato leaf juice is the stimulus, a mixed message is provided from the four cells housed in each of the nine sensilla, and this message varies considerably across the available sensilla. The result is a type of across-fiber pattern that signals "do not eat." (A) Summary of the behaviors exhibited by newly emerged Colorado potato beetle adults when provided with either potato (host plant) or tomato (nonhost plant); numbers of beetles indicated inside heavy arrows. Beetles first examine the leaf and then they squeeze it between their mandibles (macerate) before taking a small bite, which they taste for only a short time. If the plant is acceptable, they very quickly move to sustained feeding. If the plant is less acceptable, few beetles will feed. The decision to not feed is made after considerable time has been spent in examining, macerating, taking small bites, and sometimes repeating one or more of these steps. [Modified from Harrison, G. D., (1987). Host-plant discrimination and evolution of feeding preferences in the Colorado potato beetle, Leptinotarsa decemlineata. Physiol. Entomol. 12, 407-415.] (B) Taste sensilla are important in making the kinds of decisions shown in (A). If potato leaf juice is the stimulus, four cells in nine sensilla on the mouthparts respond by sending a clear, almost labeled-line (cell 1), message to the central nervous system. When tomato leaf juice is the stimulus, a mixed message is provided from the four cells housed in each of the nine sensilla, and this message varies considerably across the available sensilla. The result is a type of across-fiber pattern that signals "do not eat."](http://lh3.ggpht.com/_X6JnoL0U4BY/S8FcH2qi9NI/AAAAAAAAXnU/5WHTvNog0OU/tmp4226_thumb_thumb.jpg?imgmax=800)
FIGURE 4 (A) Summary of the behaviors exhibited by newly emerged Colorado potato beetle adults when provided with either potato (host plant) or tomato (nonhost plant); numbers of beetles indicated inside heavy arrows. Beetles first examine the leaf and then they squeeze it between their mandibles (macerate) before taking a small bite, which they taste for only a short time. If the plant is acceptable, they very quickly move to sustained feeding. If the plant is less acceptable, few beetles will feed. The decision to not feed is made after considerable time has been spent in examining, macerating, taking small bites, and sometimes repeating one or more of these steps. [Modified from Harrison, G. D., (1987). Host-plant discrimination and evolution of feeding preferences in the Colorado potato beetle, Leptinotarsa decemlineata. Physiol. Entomol. 12, 407-415.] (B) Taste sensilla are important in making the kinds of decisions shown in (A). If potato leaf juice is the stimulus, four cells in nine sensilla on the mouthparts respond by sending a clear, almost labeled-line (cell 1), message to the central nervous system. When tomato leaf juice is the stimulus, a mixed message is provided from the four cells housed in each of the nine sensilla, and this message varies considerably across the available sensilla. The result is a type of across-fiber pattern that signals “do not eat.”
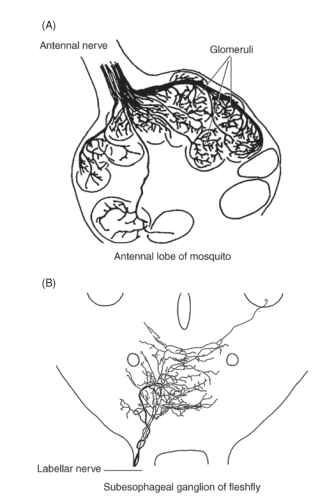
FIGURE 5 In insects, both olfactory and gustatory cells send axons (afferents) directly to the central nervous system. The first synapse (information relay point) is in a particular part of the central nervous system for each sensory modality. (A) Olfactory afferents go to the antennal lobe, where the input is organized in a manner resembling a bunch of grapes—glomerular organization.
Gustatory afferents from mouthpart sensilla go to the subesophageal ganglion, where they project into a discrete space that is not organized into glomeruli. For both (A) and (B), subsequent processing is done by first-level and higher interneurons. [From Edgecomb, R. S., and Murdock, L. L. (1992). Central projections of axons from the taste hairs on the labellum and tarsi of the blowfly, Phormia regina Melgeri. J. Comp. Neurol. 315, 431-444. Reprinted by permission of Wiley-Liss, Inc., a subsidiary of John Wiley & Sons, Inc.]
The complex array of stimuli represented by potato actually stimulates a single cell—the others may well be inhibited. The tomato leaf juice, however, causes several cells to fire in an inconsistent pattern. The first is another example of a labeled-line type of code, while the second is an across-fiber pattern. In the latter type of code, the brain is receiving information from several physiologically distinct cells, and it is the pattern that is important. It is thought that the across-fiber code pattern prevails in many situations involving complex chemical mixtures. Progress in this area is impeded by the inherent variability of the types of recording possible in the across-fiber pattern (see, e.g., Fig. 4B).
CENTRAL PROCESSING OF CHEMOSENSORY INPUT
Over the past 20 years, studies of insect olfactory systems have produced a rich literature on the topic of central processing, particularly for pheromonal systems. Work on gustatory systems is far less advanced. The section on insect pheromones provides more information on olfactory processing. This section simply contrasts the gross morphology of the two systems. Both olfactory and gustatory sensory cells are primary neurons; that is, they connect the periphery (sensillum) directly with the central nervous system. Olfactory cells, on the antennae as well as on the palpi, send their axons directly to the antennal lobe, which is a part of the insect brain. Gustatory cells, for the most part, send their axons to the ganglion for the segment in which the sensory cell occurs. Figure 5A shows a typical innervation pattern for antennal and mouthpart olfactory cells in a mosquito, and Fig. 5B shows innervation from the gustatory cells in the mouthparts of a blowfly. A striking difference in the organization of the two parts of the central nervous systems receiving these imputs is repeated across many animal phyla. Olfactory systems are characterized by a glomerular arrangement (like a bunch of grapes) of the neural centers (neuropil) that receive olfactory afferents (input), but gustatory systems have no such patterned arrangement. The distribution of olfactory inputs into glomeruli suggests a strong association of structure with function, and this is most clearly seen in the macroglomer-uli, which receive only pheromonal afferents in male moths. There is undoubtedly an association of structure with function in the way gustatory inputs are arranged, but the lack of a glomerular substructure makes any such system far less obvious. The two ways of organizing chemosensory input, throughout animals, may also point to important differences in coding and or evolution.
