Growing old holds many pleasures, but for someone with Alzheimer’s disease (AD), it can be a confusing and frightening experience. The image of an absentminded elderly man or woman has been with us for a long time. People today are in the habit of thinking that this is the natural consequence of growing old, but gerontologists have taught us to be cautious of this stereotype. Old people may be slower at certain tasks, but they are not necessarily senile or any more absentminded than a 20-year-old. Aging makes us more susceptible to certain diseases, but those diseases are not an inevitable consequence of growing old. Several other age-related diseases are described in this topic, but there are none so devastating as Alzheimer’s disease.
Alzheimer’s disease
Alzheimer’s disease (AD) is a neurological disorder affecting the central nervous system (CNS) that leads to a progressive loss of memory, language, and the ability to recognize friends and family. The average course of the disease, from early symptoms to complete loss of cognitive ability, is 10 years. Alois Alzheimer, a German neurologist, first described AD in 1907, and it has since become the fifth-leading cause of death among the elderly. The incidence of this disease increases with age and is twice as common in women as in men. The reason for this difference is unclear, but may be due to the sharp decline in the amount of estrogen that occurs during menopause. In 2009 more than 5 million men and women were living with AD in the United States alone, and this number is expected to increase to 16 million by 2050. Worldwide, there are more than 20 million recorded cases, but because poor medical facilities and diagnostic procedures in many parts of the world result in underreporting of the disease, the real number is likely to be much higher. In the United States, the annual cost of treating AD and other dementias is 148 billion dollars.
Understanding AD, and finding ways to treat it, has proved to be extremely challenging. It affects the brain, the most complex organ ever to evolve. Indeed, for most of the past 100 years scientists have thought that this disease would prove be too difficult to resolve. The brain, after all, consists of 100 billion neurons linked into a three-dimensional network consisting of 100 trillion connections. Nevertheless, over the past 10 years scientists have gained a much better understanding of AD and are now using their discoveries to develop therapies for this terrible disease. These discoveries are the subject of this topic.
The Central Nervous System, which is affected by AD, consists of the brain and the spinal cord. The main part of the brain is called the cerebrum, which is the home of human intellect and the source of individual personality. It also processes and analyzes information from all the sensory nerves of the body. The cerebrum consists of two morphologically identical cerebral hemispheres, connected by a thick bundle of nerves called the corpus callosum. All of the nerve cell bodies are located in the outer layer of the cerebrum called the cerebral cortex. A special area of the cerebrum called the hippocampus is important for processing memories for long-term storage in other parts of the brain. The cerebellum regulates fine motor control over our muscles, making it possible for a person to learn how to play the piano, knit a sweater, and perform other activities that require intricate coordination. The brain stem is in control of our automatic functions, such as the rate at which the heart beats, the contraction of muscles of the digestive tract, and respiratory rate. It also controls our ability to sleep and to stay awake.
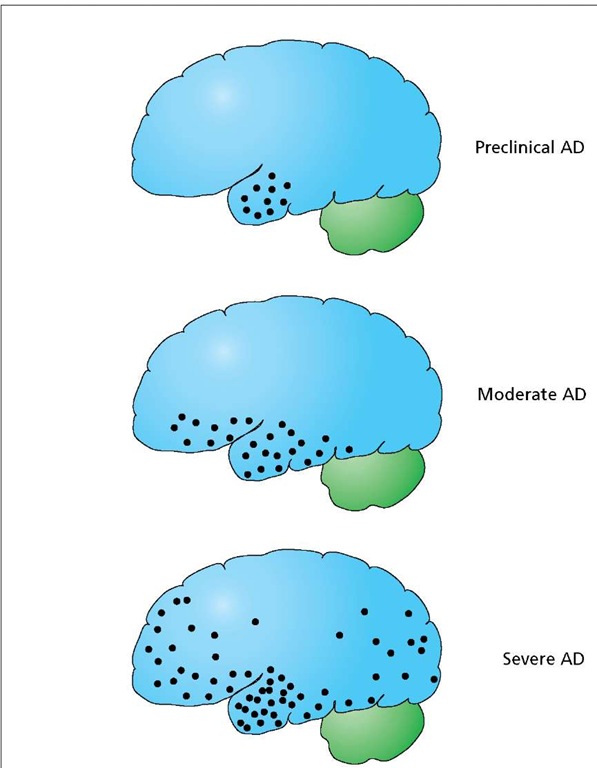
AD begins in the basal cerebral cortex, quickly spreading to the hippocampus. During the early stages, known as preclinical AD, some damage occurs to the brain, but not enough to produce outward signs of the disease. Over a period of years, AD spreads to many areas of the cerebrum, but it does not affect the cerebellum or the brain stem.
The CNS is constructed of neurons, remarkable cells that are designed for communication. These cells have special structures, known as dendrites and axons, that receive and transmit signals. A signal in the form of an electrochemical jolt enters a neuron at its dendrites and is passed along to another neuron through the axon, a process that takes less than a microsecond. Neural circuits are constructed when axons make contact with the dendrites of other neurons. The connection between an axon and a dendrite is called a synapse. Circuits in the human brain consist of billions of neurons, each forming thousands of synaptic junctions with other neurons. These circuits give us our intellect, emotions, senses, and the ability to recognize our friends and loved ones.
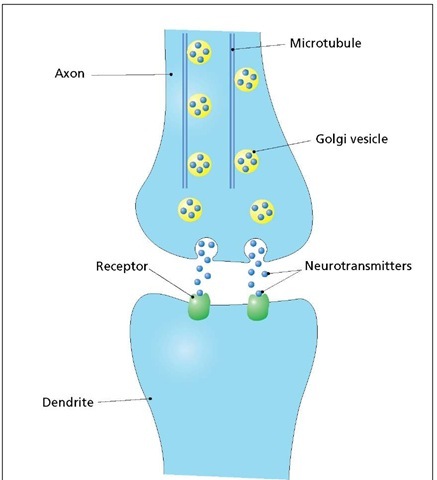
Although neurons communicate through the synapse, they do not actually touch one another. Close inspection of a synapse shows a small gap separating the axon from the dendrite. A signal is transmitted across the gap by the release of small proteins called neurotransmitters, which are stored at the axon terminus in Golgi vesicles.
Progression of AD. Alzheimer’s disease (black circles) begins in the hippocampus, spreading over a period of years to affect several regions of the cerebrum.
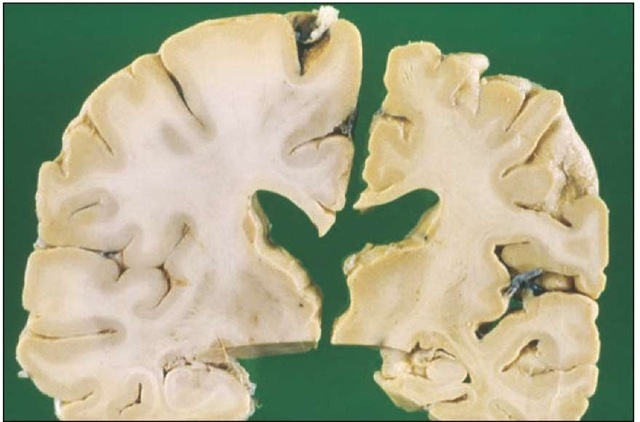
Alzheimer’s disease. Sliced sections from two brains. On the left is a normal brain of a 70-year-old. On the right is the brain of a 70-year-old with Alzheimer’s disease. The right brain is atrophied with a loss of cortex and white matter. Alzheimer’s disease is not a normal part of aging. It is a dementing disorder that leads to the loss of mental and physical functions. The chance of developing this disease increases with age.
The vesicles travel to the axon terminus on a "railroad" constructed of microtubules. When a neuron receives a signal, the Golgi vesicles at the terminus are released from the microtubules and fuse with the axonal membrane, dumping their cargo into the synaptic gap. The neurotransmitters quickly diffuse across the gap and bind to receptors on the dendrite membrane, triggering an electrochemical impulse in the target neuron, thus completing transmission of the signal. This may seem like an awkward way for neurons to signal one another, but the synaptic gap and the use of neurotransmitters are crucial for maintaining the strength of the signal over a network that consists of billions of cells.
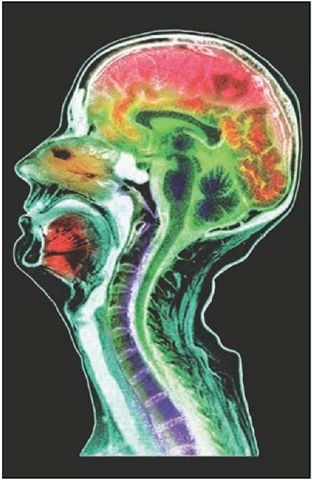
Colored magnetic resonance imaging (MRI) scan of a sagittal section through the brain of a 51-year-old male, showing cerebral atrophy. Atrophy of parts of the cerebrum of the brain occurs in various disorders, including stroke, Alzheimer’s disease, and AIDS dementia. Here the area of the upper cerebrum affected by atrophy is colored dark red. Atrophy is shrinkage and wasting away of tissue. In stroke, brain cells die due to deprived blood supply to the brain; in Alzheimer’s disease, the brain shrinks leading to senile dementia.
Four genes have been identified as associated with the onset of AD. The first of these is Tau, which codes for a protein (Tau) needed for the construction of microtubules. The second gene, APP, codes for amyloid precursor protein, APP, a glycoprotein that is embedded in the cell membrane and may function as a cell-signaling receptor. The third gene, Sen (senilin, also known as presenilin and secretase), codes for a protease (an enzyme that can cut a protein into two or more pieces) that is involved in processing APP. The fourth gene, ApoE, codes for a protein called apolipoprotein E. Defects in any of these genes can lead to the death of neurons that is characteristic of AD.
The Tau gene and its product have a crucial role in the maintenance of neuronal signal transmission. The Tau protein is an important component of the microtubule railroad the Golgi vesicles use to reach the axon terminus.
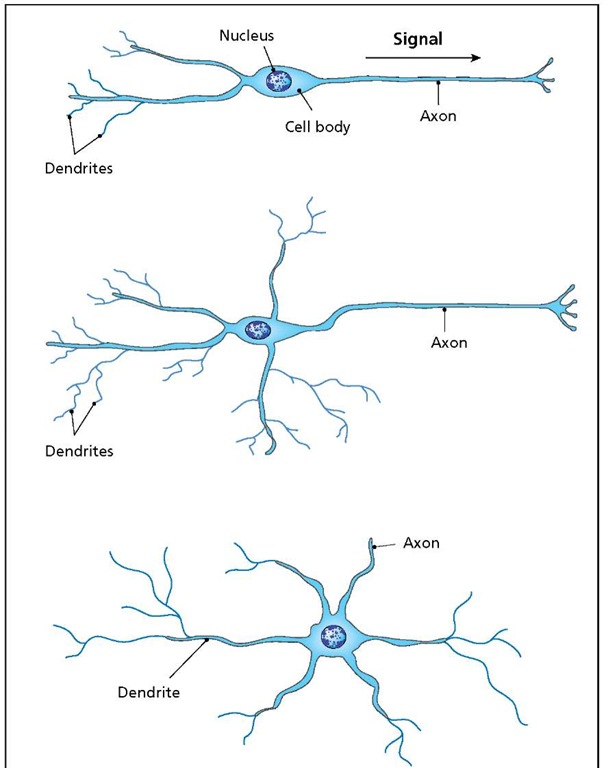
A neuron receives signals at its dendrites and passes them on to other neurons through its axon. Simple bipolar neurons (top) have the dendrites and the axon at opposite ends of the cell. Multipolar neurons (middle and bottom) have a complex dendritic structure that often surrounds the cell body (bottom). In such cases, the identity of the axon is not always obvious.
Neural circuits. These circuits are constructed with axon terminals making connections with the dendrites of other neurons. The connection between an axon and a dendrite is called a synapse. Circuits in the brain consist of billions of neurons, each forming thousands of synaptic junctions with other neurons. These circuits give humans intellect, emotions, ability to see the world, and much more.
Synaptic junction. Axons and dendrites do not touch each other but are separated by a small gap called the synapse or synaptic junction. A signal is transmitted by the release of small molecules called neurotransmitters that are stored at the axon terminus in Golgi vesicles. Binding of the neurotransmitter to the receptor on the dendrite membrane completes the transmission. The Golgi vesicles travel to the axon terminus on a transportation network constructed from microtubules.
A mutation in this gene produces a defective protein, leading to the breakdown of microtubules and a virtual collapse of the cell’s ability to pass on incoming signals. The abnormal Tau and the disintegrating microtubules collect within the cell as neurofibrillary tangles (NFTs). When a neuron loses its ability to communicate, it is as though it loses its will to live. This phenomenon has been observed in patients suffering from a damaged or severed spinal cord. Peripheral nerves starved for signals from the CNS degenerate and die. Similarly, neurons in the brain of an AD patient degenerate and die when signals stop coming in. In this case, however, the loss is more than the movement of an arm or a leg; it results in the destruction of the persona, the core of a person’s being.
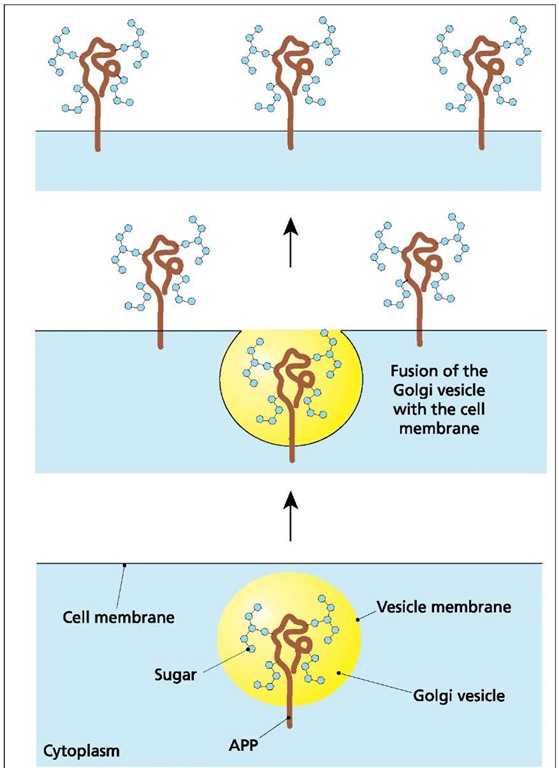
A second route to the development of AD involves the APP and Sen genes. Neurons, like all cells, are covered in a molecular forest called the glycocalyx. This forest consists of a wide variety of glycoproteins, resembling trees, that have many functions: Some are hormone or glucose receptors; others are involved in processing the electrochemical signals generated by neurotransmitters. An important member of a CNS neuron’s glycocalyx is the APP protein, which is believed to be involved in hormonal signal transduction. APP is processed through the Golgi complex and planted on the cell surface by fusion of the Golgi vesicles with the cell membrane.
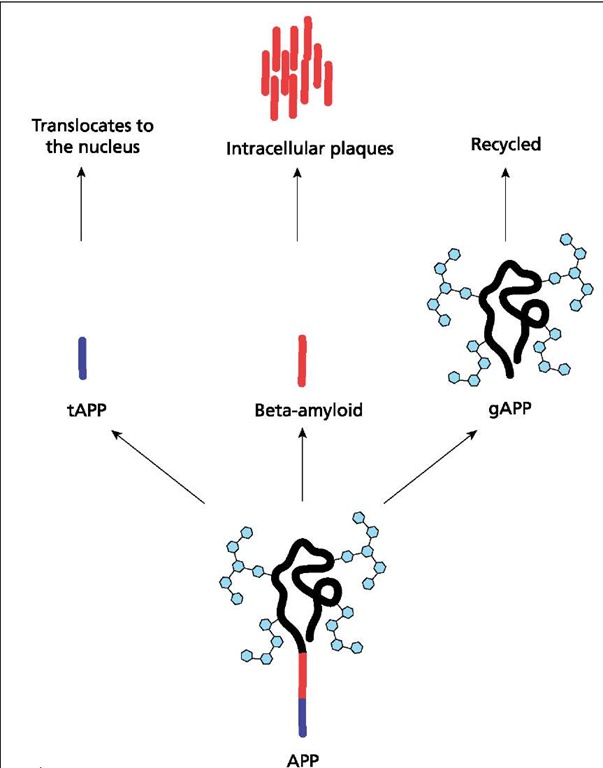
Neurons suffering from AD fail to process App properly. Scientists believe that senilin is activated as part of a normal signal trans-duction pathway. Activation of the pathway begins when a signaling molecule (as yet unidentified) binds to APP, which in turn activates senilin in order to produce a secondary messenger, truncated App (tAPP), as well as two other fragments: beta-amyloid and the glyco-sylated portion of the protein. According to this hypothesis, tAPP translocates into the nucleus, where it activates the appropriate gene or genes. AD develops when a mutation in Sen results in the production of a permanently activated senilin with a subsequent buildup of tAPP and beta-amyloid.
Planting an APP forest. Amyloid precursor protein (APP) is a glycoprotein with a treelike structure that is an important member of the cell’s glycocalyx. The "trunk" (brown) is protein, and the "leaves" are sugar molecules (blue). Vesicles from the cell’s Golgi apparatus carry APP to the cell surface. Fusion of the vesicle membrane with the cell membrane automatically plants APP in the cell membrane.
Scientists have long assumed that the accumulation of beta-amyloid plaques was toxic to neurons and was directly responsible for the extensive neuronal death that is typical of AD. In addition, the fourth AD gene, ApoE, was thought to code for a product that helped clear beta-amyloid from the brain. Loss of this function was the result of mutation being then responsible for excessive plaque formation. Some research suggests that neural damage is caused by an excess of tAPP, which somehow orchestrates the hyperphosphorylation of the Tau protein, leading to its disintegration and the destruction of the cell’s railway.
The chronic and inappropriate destruction of APP by a mutated senilin poses an additional threat to the health of affected neurons. APP is a major component of a neuron’s surface "forest." A normal glycocalyx is crucial for a cell’s survival in more ways than one. The immune system uses the exact composition of the glycocalyx to distinguish self from nonself. Those cells that are a normal part of the body can be branded nonself, or invaders, if the glycocalyx is abnormal. If this happens, the immune system can order the affected cells to commit suicide, in a process known as apoptosis. Thus, whether the onset of AD is through a defective Tau or Sen gene, the final outcome—extensive neuronal death—is the same.
At present, there is no way to cure AD, although treatments are being developed to inhibit senilin and to reduce the accumulation of the beta-amyloid, which could be responsible for some of the neural damage. Other treatments being planned involve a combination of gene therapy and stem cell transplants to correct the mutated Tau and Sen genes and to replace the damaged or dying neuronal population. Experiments show that stem cells injected into damaged rat brains do differentiate into appropriate neurons; whether they make the correct connections, however, is yet to be determined. Given the delicacy of the central nervous system and the complexity of its circuits, it is likely that such therapies will be extremely difficult to develop.
Production of beta-amyloid. In Alzheimer’s disease, APP is cut in two places, producing three fragments by a protease called presenilin or secretase: truncated APP (tAPP), beta-amyloid, and glycosylated APP (gAPP). The fate of tAPP is unclear, although it may translocate to the nucleus, where it acts as a transcription factor. Beta-amyloid collects in the intracellular space, where it forms the plaques that are characteristic of AD. The gAPP is recycled and does not contribute to the clinical symptoms of the disease.
Researchers have long been frustrated by the lack of an effective diagnostic procedure validating new AD therapies. Drs. William Klunk and Chester Mathis, at the University of Pittsburgh, were among the first to develop an effective AD diagnostic procedure.
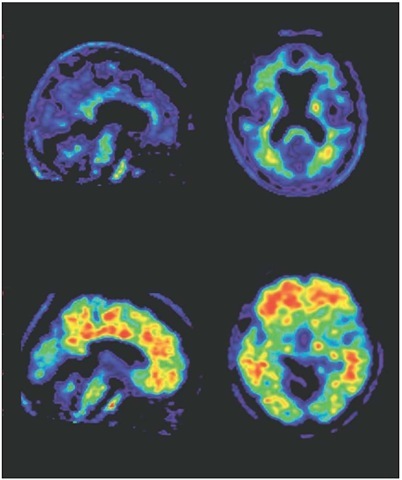
Imaging beta-amyloid. A PET scan of the brain of a healthy volunteer (top pair) and a patient with Alzheimer’s disease (bottom pair). Amyloid plaques show up as red, indicating high uptake of florbetapir.
They began by testing various forms of a biological dye called thioflavin, which has a special affinity for beta-amyloid. They then found a way to attach the radioisotope carbon-11 to the molecule without reducing the dye’s affinity for beta-amyloid while at the same time ensuring that the labeled dye could enter the brain. This labeled dye is now known as Pittsburgh compound B (PiB). After injecting PiB into a patient, the location of the dye is determined with positron emission tomography (PET). The intensity of the signal is proportional to the amount of beta-amyloid present. While accurate, this procedure is difficult to implement because of the very short half-life of carbon-11, which is only 20 minutes. Thus, PiB can only be used in a few hospitals that have the equipment to produce the dye onsite. Moreover, in many cases the PET scan can take up to 90 minutes to administer, which is nearly five times the half-life of the label.
Dr. Daniel Skovronsky and his team have improved on this procedure by using a dye labeled with fluorine-18, a radioisotope with a half-life of 110 minutes. This labeled dye, called florbetapir, has an affinity for beta-amyloid that is similar to that of PiB and is more convenient to use. Skovronsky’s team has also gone to great lengths to validate the procedure by comparing the PET scan results with tissue sections obtained from the same patients after they have died. In most cases, the correspondence is good: Tissue sections testing positive for beta-amyloid were shown to have come from patients testing positive with the PET scan. In some cases, patients diagnosed with AD tested negative with the PET scan, and their tissue sections also tested negative, meaning the diagnosis had been wrong.