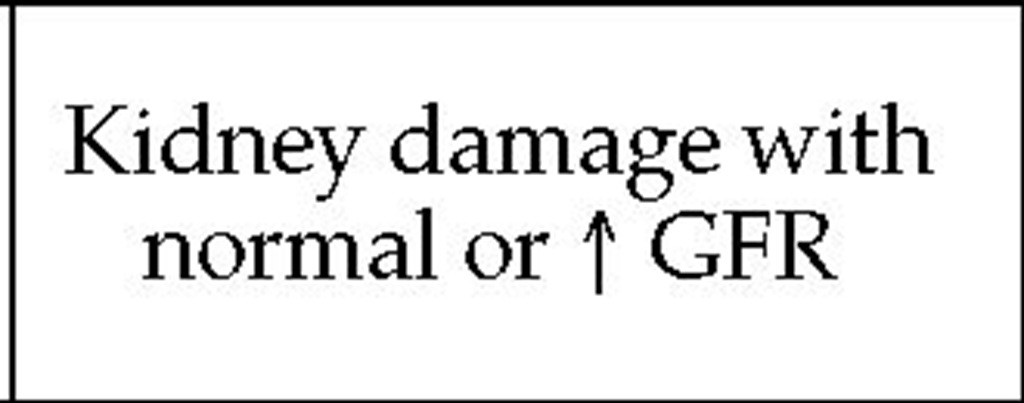End-stage renal disease (ESRD) is an important source of long-term morbidity and mortality. Efforts to reduce or prevent the development of this disorder are of paramount importance. One of the problems that nephrologists often encounter is that patients are referred late in the course of disease, when treatments are largely ineffective. Thus, it is imperative to identify patients early and to treat them aggressively. Recently published clinical practice guidelines are aimed at accomplishing this goal. This topic reviews the management of patients with chronic kidney disease (CKD), giving particular emphasis to the following: (1) methods that can be clinically used to accurately stage and monitor the level of renal function; (2) identification of common comorbidites of CKD; (3) the pathophysiology of CKD; (4) therapies designed to slow the progression of CKD; (5) management of common complications of CKD (e.g., anemia, fluid and electrolyte disturbances, and renal osteodystrophy); and (6) timely referral to a nephrologist so as to ensure a smooth transition to renal replacement therapy when indicated. Information on the causes and manifestations of CKD are covered elsewhere [see 10:X Chronic Renal Failure and Dialysis].
Overview of Clinical Practice Guidelines for Chronic Kidney Disease
It is estimated that 20 million persons in the United States population have chronic kidney disease. The typical course of patients with CKD is progression to kidney failure and development of cardiovascular disease. Increasing evidence suggests that treatment strategies initiated at earlier stages of CKD are effective in slowing the progression toward kidney failure. In addition, addressing cardiovascular risk factors at earlier stages of CKD should be effective in reducing cardiovascular events and mortality, both before and after the onset of kidney failure.
To date, implementation of measures designed to detect and treat CKD at an early stage have been hampered, in part by lack of agreement on a definition and classification of stages in the progression of chronic kidney disease. In addition, there has not been a uniform application of tests for detection and evaluation of such patients. In February 2002, the Kidney Disease Outcomes Quality Initiative (K/DOQI) clinical practice guidelines were published in an attempt to address the growing burden of CKD in the United States.1 These guidelines emphasize the need for prevention, early diagnosis, and aggressive treatment of CKD.
Definition and staging of chronic kidney disease
CKD is defined as either kidney damage or a glomerular filtration rate (GFR) of less than 60 ml/min/1.73 m2 for more than 3 months. Kidney damage is defined as pathologic abnormalities or markers of damage, including abnormal findings in blood or urine tests or imaging studies. In patients with CKD, the stage is defined by the level of the GFR. The level of GFR is considered to be the best measure of overall kidney function in health and disease.
Inulin or iothalamate clearance is regarded as the gold standard for measuring GFR. However, these methods are cumber some and expensive and are not available to many practicing physicians. By contrast, the GFR can be easily estimated by formulas that take into account the patient’s age, gender, ethnicity, and serum creatinine concentration [see Table 1]. The Modification of Diet in Renal Disease (MDRD) and Cockcroft-Gault equations are two such formulas that provide useful estimates of GFR in adults. Of these, the MDRD equation is more accurate and precise for persons with a GFR of less than 90 ml/min/1.73 m2. The equation was based on a large database encompassing persons with various diseases and including both European Americans and African Americans. The K/DOQI clinical practice guidelines state that clinicians should not use serum creatinine concentration as the sole means to assess the level of kidney function. In addition, measurement of the 24-hour creatinine clearance level to assess GFR is no more accurate than estimating GFR from the MDRD equation. Certain clinical conditions, however, may necessitate use of creatinine clearance as a way to better assess the GFR [see Table 2].
Evaluation of all patients with CKD should include testing for proteinuria. Persistent proteinuria is usually a marker of kidney damage, and in the setting of a normal or increased GFR, it signifies the presence of stage 1 CKD. Proteinuria is also a strong and independent predictor of increased risk for cardiovascular morbidity and mortality, particularly in high-risk groups such as diabetics, hypertensives, the elderly, and those with CKD.
The preferred method for measuring proteinuria is the ratio of protein or albumin to creatinine (mg/g) in an untimed urine specimen. Timed urine collections are inconvenient to obtain and may be associated with errors; therefore, they are usually not necessary for measuring proteinuria. In addition to being utilized for the detection of CKD, measurement of the protein- or albumin-to-creatinine ratio is a useful way to monitor proteinuria in patients with known CKD. Changes in the degree of proteinuria are directly associated with the risk of loss of kidney function.
Evaluation and treatment
Once a patient is identified as having CKD, a clinical action plan is implemented on the basis of the patient’s stage of disease [see Table 3]. Although specific therapies vary according to the underlying cause of kidney disease, many aspects of treatment are common to all types of CKD. Treatment of comorbid conditions, interventions to slow the progression of kidney disease, and measures to reduce cardiovascular disease should begin during stages 1 and 2. Particular attention needs to be given in stages 1 and 2 to aggressively control blood pressure. Evaluation and treatment of anemia, malnutrition, bone disease, neuropathy, and decreased quality of life should be undertaken in patients with stage 3 CKD. Preparations for renal replacement therapy should begin as patients enter stage 4. Kidney failure (stage 5) is defined either as a GFR of less than 15 ml/min/1.73 m2 or as complications of decreased GFR severe enough to increase the risk of mortality and morbidity unless initiation of kidney replacement is undertaken. Some patients who have symptoms of uremia may require kidney-replacement therapy although their GFR is greater than 15 ml/min/1.73 m2.
|
Table 1 Equations to Estimate Glomerular Filtration Rate |
|
Abbreviated MDRD (Modification of Diet in Renal Disease Study) equation: GFR (ml/min/1.73 m2) = 186 x (Scr)-1-154 x (age)-0 203 x 0.742 (if female) x 1.210 (if African American) |
|
Cockcroft-Gault equation: Creatinine clearance (ml/min) = [140 - age x weight (kg) / 72 x serum creatinine] x 0.85 (if female) |
|
Table 2 Clinical Conditions That May Require Measurement of GFR Using Clearance Methods |
|
Extremes of age and body size |
|
Severe malnutrition or obesity |
|
Diseases of skeletal muscle |
|
Paraplegia or quadriplegia |
|
Vegetarian diet |
|
Rapidly changing kidney function |
|
Calculation that potentially toxic drugs may be excreted by the kidney |
The K/DOQI guidelines recommend that all routine health encounters include an assessment to determine whether an individual is at increased risk for developing kidney disease. Persons deemed to be at increased risk should undergo testing for proteinuria. Detection of proteinuria in patients at increased risk would allow for early implementation of measures designed to reduce proteinuria, slow the progression of kidney disease, and reduce the risk of cardiovascular mortality and morbidity. Numerous clinical factors are identified with increased risk of developing kidney disease [see Table 4]. Some of these factors are discussed in more detail below.
Comorbidites of Chronic Kidney Disease
In the United States, millions of patients have an increased serum creatinine concentration.2,3 In many of these patients, the renal insufficiency is advanced. Diabetes and hypertension are the underlying causes of renal disease in the majority of these patients. The remaining cases can be accounted for by a variety of primary glomerular diseases, cystic renal diseases, obstruction uropathy, and renal diseases related to infections.
Regardless of the underlying cause, it should be appreciated that CKD carries with it the burden of other comorbidities that directly influence patient outcome. In addition, abnormal renal function confers its own inherent risk. In the Hypertension Detection and Follow-up Program, cardiovascular disease was strongly associated with a serum creatinine level of 1.7 mg/dl or more, and this risk was independent of other factors.4 In the 10,768 participants in this program, the risk of death increased progressively with increasing creatinine concentration. There was an almost fivefold increase in the 8-year mortality between patients in the lowest stratum of creatinine concentration and those in the highest. Similar findings were reported in the Heart Outcomes and Prevention Evaluation.5 In this study, the incidence of cardiovascular death, myocardial infarction, and stroke increased with each quartile of serum creatinine concentration. This association between renal function and mortality is particularly strong in the elderly.6
Stroke
The prospective British Regional Heart Study, comprising 7,690 men 40 to 59 years of age, demonstrated that the risk of stroke was significantly increased when the creatinine concentration was above the 90th percentile—a serum creatinine concentration of 116 mmol/L—whereas all-cause mortality and cardiovascular mortality were significantly increased when the serum creatinine concentration was only above the 97.5 per-centile.7 In elderly persons admitted to an acute care hospital with stroke, the serum creatinine concentration was a highly significant predictor of survival.8
Cardiac disease
The relationship between renal function and myocardial infarction (MI) was studied in 417 patients.9 These individuals did not have diabetes, and their baseline creatinine values ranged from 0.7 to 1.9 mg/dl. Each 0.1 mg/dl increase in baseline creati-nine concentration was associated with a 36% increase in the relative risk of subsequent overall mortality and a 47% increase in the relative risk of subsequent mortality from coronary artery dis-ease. A similar trend between mortality and serum creatinine levels was observed in another study of post-MI patients.10 Nearly 75% of individuals starting dialysis have left ventricular hypertrophy (LVH), and there is an inverse relationship between renal function and the incidence of LVH in patients before undergoing dialysis.11 There is also a high prevalence of congestive heart failure. The incidence of coronary artery disease is approximately 40% in patients receiving dialysis, and there also appears to be an increased incidence of peripheral vascular disease in patients with abnormal renal function.
Table 3 Stages of CKD and Clinical Action Plans
CKD—chronic kidney disease
GFR—glomerular filtration rate
Risk Factors for Progression of Kidney Disease
Most patients with established CKD continue to experience a decrease in renal function over time.3 This section reviews various factors known to contribute to loss of renal function. In addition, therapies that can slow the progression of CKD are discussed.
Diabetes mellitus
Animal models have provided a clear picture of the mechanisms underlying diabetic glomerular destruction. What is not known is why only approximately 40% of patients with type 1 (insulin-dependent) diabetes mellitus are at risk. Obviously, genetics plays an important role, but other potentially manageable factors are also important. Hyperglycemia through nonenzymat-ic glycosylation, production of oxidants, or stimulation of the production of diacylglycerol may activate protein kinase C (PKC). It has been shown that levels of PKC are increased in the diabetic kidney.12,13 PKC causes an increase in production of transforming growth factor-| (TGF-|), which is a potent stimulator of collagen formation and basement membrane thickening. A variety of other alterations, including permeability changes, altered fibrinoly-sis, changes in gene expression, and vascular contractility, may also be stimulated by PKC.14 Interestingly, inhibition of PKC in the kidney and retina reduces diabetes-related abnormalities.15 Micropuncture studies in diabetic animals showed that glomeru-lar intracapillary pressures were increased.16 These changes may lead to structural alterations that are important in the pathogene-sis of diabetes-related complications.
Type 1 Diabetes Mellitus
The most important study regarding the treatment of type 1 diabetes is the Diabetes Control and Complications Trial (DCCT).17 This study examined 1,441 patients with type 1 diabetes. Of these patients, 726 had no retinopathy at baseline; 715 had mild retinopathy. After a mean follow-up period of nearly 6—2 years, intensive insulin therapy reduced the occurrence of microalbu-minuria by 39% and the occurrence of macroalbuminuria (> 300 mg/24 hr) by 54% in the combined groups. Improvement in nephropathy was mirrored by a slowing in the progression of retinopathy and neuropathy. A follow-up to this study indicated that the effects of intensive treatment are long lasting.18
Type 2 Diabetes Mellitus
Although type 1 diabetes has received a great deal of attention, the incidence of type 2 (non-insulin-dependent) diabetes continues to rise at a rapid pace, particularly in members of minority groups, including African Americans and Native Americans. In patients with nephropathy and type 2 diabetes, familial clustering has been observed. Polymorphism of the an-giotensin-converting enzyme (ACE) gene may influence the rate of renal disease progression, and several genes have been linked to renal disease in specific populations.19 Although the renal lesions in type 2 diabetes are similar to those in type 1, a greater proportion of patients with type 2 diabetes have nonspecific lesions, including ischemic changes.20 Control of hyper-glycemia in patients with type 2 diabetes is a controversial issue. Some studies have failed to show an association between good glucose control and the maintenance of functional nephron mass. On the other hand, Hsu and colleagues demonstrated that hemoglobin A1c (HbA1c) levels were independent predictors of a decline in renal function.21
Treatment of Diabetes Mellitus in Patients with CKD
Treatment of type 1 diabetes mellitus Treatment of type 1 diabetes in patients with CKD requires the use of insulin therapy [see 9:VI Diabetes Mellitus]. Management should include a carefully structured diet and exercise. Care must be taken to avoid malnutrition with advancing renal insufficiency because anorexia is part of the uremic syndrome. Continuing podiatric care is an important part of the exercise program. The use of insulin requires special care in patients with renal insufficiency because of the prolonged half-life of insulin. Furthermore, CRF patients with type 1 diabetes frequently have long-standing disease, and there may be few warning signs of hypoglycemia. Gastroparesis with erratic food absorption further increases the risk of hypo-glycemia. Home glucose monitoring is an invaluable element in the care of these patients, as is careful follow-up by providers skilled in diabetes management. Symptomatic hypoglycemia is much more common with tight glucose control, and it is difficult to maintain proper glucose levels within the framework of traditional practice. This is clearly evident in the follow-up of the patients in the DCCT trial.17,18 In the patients assigned to receive tight control of glucose levels, HBA1c increased from 7.2% to 7.9% in the 4-year interval after the conclusion of the original study. Transplantation is gaining increasing favor as a method of treating patients with type 1 diabetes, especially those with advancing renal disease, because of the use of simultaneous kidney and pancreas transplantation.
Treatment of type 2 diabetes mellitus Treatment of patients with type 2 diabetes who have renal insufficiency should also begin with a carefully designed diet and exercise program. Weight loss plays an important role in improving insulin resistance and should be employed as well. It should be kept in mind that there are a number of drugs that are associated with worsening of insulin resistance [see Table 2 and 9:VI Diabetes Mellitus]. One should consider altering the drugs in patients with type 2 diabetes whose glucose levels are difficult to control. Early in the course of disease, when the patient has some pancreatic insulin reserves, hypoglycemic agents can be considered. Their use, however, is complicated in patients with renal disease. Met-formin and other biguanides may cause lactic acidosis and should not be used in patients with even mild degrees of renal dysfunction. Troglitazone may be used in this situation because of its hepatic excretion; however, there is a high risk of acute hepatitis with this agent, necessitating monthly monitoring of liver function. One should avoid the older sulfonylureas, such as chlorpropamide, because of their reduced renal excretion. Glip-izide is inactivated by the liver and may be a better choice. Acar-bose, which reduces carbohydrate absorption, should be avoided. Unfortunately, the majority of patients with type 2 diabetes require insulin therapy. These individuals frequently gain weight with therapy. Other complications include hypoglycemia and hyperinsulinemia. The latter is of concern because of its association with cardiovascular complications. Patients whose glucose levels cannot be controlled within an acceptable range should be referred to specialized clinics. Investigations are needed to understand and reverse the trend toward increasing obesity in the United States, as it is a contributing factor in the development of type 2 diabetes, particularly in susceptible minority populations.
Hypertension
The mechanisms of renal injury in hypertension were presumed to be ischemic in nature, but these mechanisms remained largely unexplained until the work of Brenner and colleagues provided detailed studies of the glomerular alterations in hypertensive rats with reduced renal mass.22,23 These studies demonstrated that afferent arteriolar vasodilatation accompanied by similar but relatively less efferent arteriolar changes increased transcapillary filtration pressures. Although these changes were associated with improving glomerular filtration rates in the short term, they were eventually seen to be detrimental, leading to progressive glomerular sclerosis. In a direct test of this hypothesis in patients, a collaborative study demonstrated that the ACE inhibitor captopril was effective in reducing the development of nephropathy in patients with type 1 diabetes and in retarding the progression of established renal dysfunction over and above its effects in lowering blood pressure.24 A similar benefit was recently shown with use of angiotensin receptor blockers in patients with type 2 diabetes and established nephropathy.
The United Kingdom Prospective Diabetes Study demonstrated that moderate reductions in blood pressure were effective in reducing renal abnormalities; in this study, there were no apparent differences in effectiveness between the beta blocker atenolol and captopril. However, the target blood pressure values may have been too high to substantiate the beneficial effects of the ACE inhibitor.27,28 The Reduction of Endpoints in NIDDM with Angiotensin II Antagonist Losartan (RENAAL) study comprised 1,513 participants who were followed for an average of 3.4 years. It demonstrated that an antihypertensive program containing the drug, compared with placebo, significantly decreased the risk of the doubling of serum creatinine, ESRD, and death.26 A repeat analysis of these data was performed to look specifically at systolic blood pressure and pulse pressure; every 10 mm Hg rise in baseline systolic blood pressure increased the risk for ESRD or death by 6.7%; a similar rise in diastolic blood pressure decreased the risk by 10.9%. Pulse pressure was a significant risk factor that was dramatically improved by losartan.29
Hypertension in Diabetic Patients
Although there is a great deal of focus on negative outcomes in patients with CRF, the Heart Outcomes and Prevention Evaluation (HOPE) trial provides some reasons for optimism. This study followed 3,577 individuals with diabetes for a median of 4.5 years. This group included 1,139 patients with microalbuminuria and 333 patients with renal insufficiency (i.e., serum creatinine level less than 2.3 mg/dl). During the follow-up period, serum creatinine values remained stable in those persons without overt proteinuria, as well as in those persons with renal insufficiency, microalbuminuria, or both. It should be noted that the patients were treated aggressively with lipid-lowering agents and antihypertensives. In addition, the study included only a few smokers.30
Hypertension in Nondiabetic Patients
Blood pressure control is equally important in nondiabetic patients with CKD. In the Seventh Report of the Joint National Committee on Detection, Evaluation, and Treatment of High Blood Pressure (JNC VII), a blood pressure goal of less than 130/85 mm Hg is recommended.31 It should be anticipated that multiple medications will be required to achieve these levels of control in most CKD patients.
Treatment of Hypertension in Patients with CKD
The MDRD study helped define the target levels of blood pressure in persons with renal disease. The results suggest that in patients with less than 1 g of protein in a 24-hour urine collection, the target blood pressure should be 130/80 mm Hg or less.32 For those with findings of more than 1 g of protein, a target blood pressure value of 125/ 75 mm Hg should be sought. A recent consensus group sponsored by the National Kidney Foundation has suggested that the target blood pressure value for adults with hypertension and diabetes should be 130/80 mm Hg.33 In most cases, these lower blood pressure goals can be obtained without significantly increasing side effects [see 1:III High Blood Pressure]. The findings of the African American Study of Kidney Disease and Hypertension (AASK) trial provides evidence that factors other than blood pressure play important roles in the functional decline in CKD. A low blood pressure, (e.g., average of 128/78, compared with 141/85) did not improve protection in the participants. However, an ACE inhibitor proved superior to a dihydropyridine calcium channel blocker in retarding progression of hypertensive kidney disease.34
Proteinuria
In a study of elderly persons, urinary albumin excretion was found to be associated with cardiovascular disease.6 In this study, prognostic factors were male sex, serum creatinine concentration, and hypertension. In a study of 11,343 German patients, the presence of microalbuminuria served to identify hypertensive patients with cardiovascular risks.35 In diabetic patients, microalbuminuria is a well-known risk factor for progression to ESRD. A study from the Royal Infirmary in Edinburgh, United Kingdom, examined the course of 190 patients with type I diabetes who had had the disease for least 30 years’duration; the study followed the participants for at least 5 years. Microalbuminuria was absent in 66% of participants at baseline; 11% of these individuals died during the follow-up period. Individuals with microalbuminuria at baseline were 22% of the total; of these, 26% died during the follow-up period. Of the 8% of patients who had macroalbuminuria at baseline, 44% died during the follow-up period. These data provide strong evidence for the influence of albuminuria on overall mortality.
Interestingly, microalbuminuria is also a predictor of renal disease in patients who do not have diabetes.37 The association between proteinuria and a decline in renal function is particularly strong in patients with a finding of more than 1 g of protein in a 24-hour urine collection. The mechanism behind the association between proteinuria and a decline in renal function appears to be in part related to the fact that high levels of urinary protein stimulate interstitial inflammation and fibrosis.38 For these reasons, drug therapy that minimizes urinary protein loss seems prudent. Because angiotensin II stimulates TGF-fi1 gene expression and type IV collagen production, treatment with agents that block the effects of angiotensin are likely to be effective.
Smoking
Smoking is a well-known risk factor for the development of cardiovascular disease. The World Health Organization estimates that as many as three million deaths a year worldwide are related to tobacco use. This number of deaths will likely increase substantially in the future. Smoking is an independent risk factor for microalbuminuria in both hypertensive and nonhypertensive patients.39 The relationship between smoking and progression of renal disease is not widely appreciated; however, the risk of the progression of diabetic nephropathy has been known for over 20 years. A multicenter case-control European study of 582 patients with either IgA nephropathy or polycystic kidney disease demonstrated an increase in the risk of progression to ESRD with increased levels of smoking, as measured in pack-years; this risk was mitigated by the use of ACE inhibitors.40 Bleyer and colleagues analyzed data from the Cardiovascular Health Study Cohort. In 4,142 nondiabetic patients who were at least 65 years of age, the number of cigarettes smoked per day was found to correlate with an increase in creatinine concentration of at least 0.3 mg/dl. In general, these patients had a relatively low incidence of progressive renal deterioration.41 The mechanisms of smoking-re-lated renal injury are complex. Possible alterations include changes in intrarenal hemodynamics, altered sympathetic activity, direct tubular damage, changes in intrarenal hormones, and oxida-tive stress. Physicians should be aware of the specific data regarding the overall risk of smoking to health. The renal risks of smoking should not be underestimated, and smoking should trigger an aggressive approach by the health care provider [see Clinical Essentials: III Reducing Risk of Injury and Disease].
Protein intake
Protein intake has long been considered a potential risk factor for the progression of renal disease; protein restriction, however, remains a controversial topic in the treatment of chronic kidney disease. Interest in protein restriction in patients with renal disease has been fueled by animal studies conducted over several decades that demonstrate that diets high in protein lead to histo-logic abnormalities in the kidney and to proteinuria and that such diets are associated with high death rates. These effects have been shown to be reduced or eliminated by protein or calorie restriction. The fact that the controversy still exists suggests that the effects of protein restriction are likely to be small and difficult to achieve. A meta-analysis of 890 nondiabetic patients reported in 46 studies suggested that protein restriction was beneficial in reducing ESRD.42 There was a 46% decrease in the number of patients requiring replacement therapy. Another meta-analysis, of 1,413 patients, demonstrated a reduction of ESRD or mortality in both diabetic patients and nondiabetic patients who were assigned to receive a low-protein diet.43 A third meta-analysis, of 1,919 randomized patients and 2,248 nonrandomized patients, found that a reduction in dietary protein had only a weak effect on the development of ESRD, with diabetic patients tending to benefit the most and with more benefit being associated with longer follow-up.
The most ambitious test of the low-protein-diet hypothesis was the MDRD study, in which 585 patients with GFRs of 25 to 55 ml/min were randomized to receive either 1.3 or 0.58 g protein/kg/day. A second group, with GFRs of 13 to 24 ml/min, were randomized to receive either 0.58 or 0.29 g protein/kg/day.45 The diets of patients in the latter group were supplemented with an amino acid-keto acid mixture that equalized the protein intake. Nuclear clearances with iodine-125-iothalamate were employed; the follow-up period was slightly longer than 2 years. During the initial 4 months of the study, renal function worsened more rapidly in the low-protein group; after that initial 4-month period, the rate of decline in GFR was lower. The decline in renal function seen at the end of the study did not differ between the two patient groups. Similarly, there was no clear difference in renal function in the group receiving a very low protein diet, but there was a trend toward better outcomes in the group receiving the lowest amount of protein. A more recent review of the data indicates that individuals in the lowest GFR category whose diets contained the lowest amount of protein may have benefited from the intervention. A multicenter trial in Europe assigned 456 adults with renal disease to either a low-protein diet (0.6 g/kg/day) or a normal-protein diet (1 g/kg/day).46 The end point of the study was a doubling of the baseline creati-nine level or a need for dialysis. The difference between the groups in cumulative renal survival was of borderline significance (P < 0.06). Compliance was good in the group receiving the higher amount of protein and poor in the low-protein group. Protein restriction is of particular concern in children because of the added complication of growth. During a 3-year study, a low-protein diet did not affect renal outcome in children from 2 to 18 years of age.
Several caveats are important when one considers diet therapy in patients with CKD. First, one must ensure an adequate caloric intake of approximately 35 kcal/kg/day. Second, physicians caring for these patients should understand the techniques used to monitor overall nutrition, including anthropo-metric measurements and use of serum transferrin and serum albumin levels. Protein intake can be estimated through the use of 24-hour urine urea nitrogen measurements plus an estimate of nonurea nitrogen losses. Third, physicians should understand that uremic symptoms respond to diets low in protein. These diets normally entail reduced intake of sodium, potassium, phosphate, and acid. Thus, renal osteodystrophy, hypertension, and metabolic acidosis are improved. Fourth, low-protein diets can be used without provoking malnutrition; however, patients should be closely monitored. A low-protein diet of 0.6 to 0.7 g/kg/day can be safely used as adjunctive therapy in individuals with CKD to ameliorate symptoms and to reduce complications of uremia. In certain patients, particularly those with diabetes, such diets may slow the rate of renal disease progression.