Geoscience Reference
In-Depth Information
Bornite
10
3
10
-3
20
15
10
2
10
-2
Galena
10
NaHCO
3
CaH(CO
3
)
2
10
1
10
-1
5
Pyrrhotite
NaOH
Na
2
SO
4
MgCl
2
0
10
0
10
0
10
-4
10
-3
10
-2
10
-1
10
0
10
1
10
2
10
3
10
4
10
5
10
6
NaCl
Conductivity (S/m)
10
-1
10
1
20
Magnetite
KCl
Domestic
water
Sea
water
15
10
-2
10
2
10
0
10
1
10
2
10
3
10
4
10
5
Arsenopyrite
10
Salinity (ppm)
5
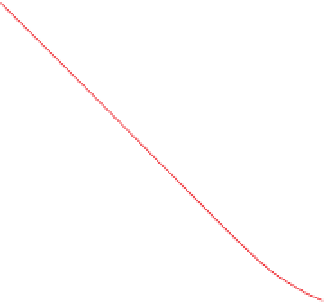
Figure 5.14
Electrical conductivity of selected pure salt solutions as a
function of concentration for solutions at 20 °C. Redrawn, with
permission, from Beblo (
1982
).
Covillite
0
10
-4
10
-3
10
-2
10
-1
10
0
10
1
10
2
10
3
10
4
10
5
10
6
Conductivity (S/m)
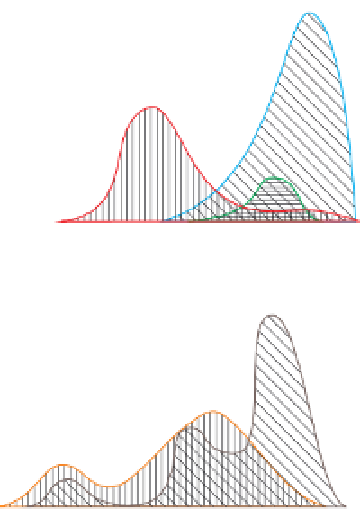
Figure 5.13
Frequency histograms of electrical conductivities of
selected ore mineral species. Redrawn, with permission, from
Parkhomenko (
1967
).
conductivity/resistivity because this group has a wider
range of porosity, which varies from values similar to
highly porous sedimentary rocks to the impervious
crystalline rocks.
required for geophysical surveying may be absent. Mag-
netite is generally one of the more conductive species, and
haematite is normally a very poor conductor, although
impurities can increase its conductivity considerably.
Materials rich in clay minerals, e.g. saprolite and
mudrocks, are amongst the most conductive rock types.
Clay minerals become very conductive when wet, and
their presence in rock pores increase the rock
•
'
s conduct-
•
Of the likely contents of pore space, air and ice are poor
conductors, and water is a comparatively good con-
ductor. Pure water has low conductivity, but naturally
occurring waters are more conductive because of the
impurities they contain, with conductivity increasing as
salinity increases (
Fig. 5.14
). Consequently, the water
table is expected to be associated with a conductivity
contrast, being more resistive in the underlying unsatur-
ated zone, as are deeply weathered water-filled structures
such as faults and shear zones. Although ice is a poor
conductor in the natural environment there is usually
also water present.
ivity considerably.
•
Dry evaporites are amongst the most resistive rock types.
•
Graphite is an important source of enhanced conductiv-
ity. It can extend the conductivity of rocks in which it
occurs, notably shales, towards values comparable to
massive sulphide mineralisation. Graphite is a mineral
whose electrical properties are highly anisotropic, i.e.
they depend on the direction of current flow relative to
the crystallographic structure. Conductivity measured
parallel to cleavage is far greater than that measured
normal to cleavage.
The electrical properties of massive mineralisation tend
towards those of the comparatively conductive constitu-
ent minerals, again because bulk electrical properties
tend to re
ect the most conductive constituents.
•
Electrical resistivity is not diagnostic of rock type. The
ranges in conductivity/resistivity of individual rock types
tend to lie between those of the rock-forming minerals
and groundwater, and are generally closer to the latter,
especially if they have signi
cant porosity. This is
because electrical properties tend to be dominated by
the most conductive components of the rock (see below).
•
Coals have electrical properties similar to other litho-
types with which they are likely to be associated.
•
The electrical properties of rocks can be understood in
the context of two basic forms of electrical conduction:
through the matrix, which requires conductive mineral
species to be interconnected; and ionic conduction through
•
Sedimentary rocks are generally more conductive than
igneous rocks owing to their higher porosity and mois-
ture content. Metamorphic rocks exhibit more variable




































































Search WWH ::

Custom Search