Geoscience Reference
In-Depth Information
Table 2.1 Dissociation reactions and solubility of selected minerals that dissolve congruently in
water (Freeze and Cherry
1979
)
Mineral
Dissociation reaction
Solubility at pH = 7 (mg/L or
g/m
3
)
Al
2
O
3
2H
2
O + H
2
O = 2A1
3
+
+ 6OH
-
Gibbsite
0.001
Quartz
SiO
2
+ 2H
2
O = Si(OH)
4
12
Ca
5
OH(PO
4
)
3
= 5Ca
2+
+ 3PO
4
3-
+ OH
-
Hydroxylapatite
30
Amorphous
silica
SiO
2
+ 2H
2
O = Si(OH)
4
120
CaF
2
= Ca
2+
+ 2F
-
Fluorite
160
CaMg(CO
3
)
2
= Ca
2+
+ Mg
2+
+ 2CO
3
2-
Dolomite
90-480
CaCO
3
= Ca
2+
+ CO
3
2-
Calcite
100-500
CaSO
4
2H
2
O = Ca
2+
+ SO
4
2-
+ 2H
2
O
Gypsum
2100
KCl = K
+
+ Cl
-
Sylvite
264,000
MgSO
4
7H
2
O = Mg
2+
+ SO
4
2-
+ 7H
2
O
Epsomite
267,000
Na
2
SO
4
10H
2
O = 2Na
+
+ SO
4
2-
+ 10H
2
O
Mirabillite
280,000
NaCl = Na
+
+ Cl
-
Halite
360,000
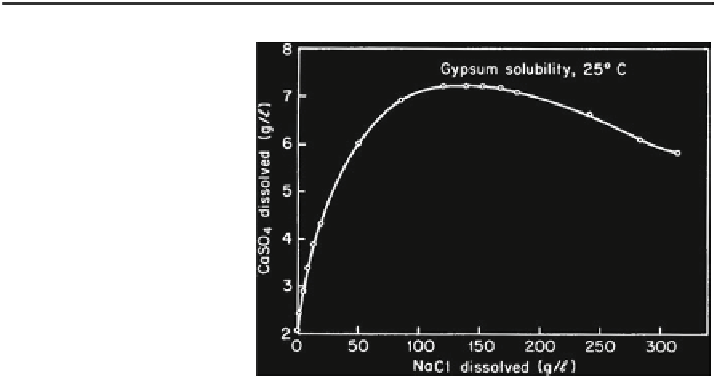
Fig. 2.2 Solubility of
gypsum in aqueous solutions
with an increasing NaCl
content (Shternina
1960
)
transport-controlled kinetics, (b) surface reaction-controlled kinetics where ions or
molecules are detached from the crystal surface, and (c) a combination of transport
and surface reaction-controlled kinetics.
Based on the results of Berner (
1978
,
1983
), Sparks (
1988
) showed that in
transport-controlled kinetics, the dissolution ions are detached very rapidly and
accumulate to form a saturated solution adjacent to the surface. In surface reaction-
controlled kinetics, ion detachments are slow, and ion accumulation at the crystal
surface is equivalent to the surrounding solution concentration. The third type of
rate-limiting mechanism for dissolution of minerals occurs when the surface
detachment is fast, and the surface concentration is greater than the solution
concentration.