Geoscience Reference
In-Depth Information
Table 18.3 Glyphosate sorption at pH 7 by synthetic aluminum and iron oxides, and pure K
+
-
saturated layer silicates. Reprinted from Borggaard and Gimsing (
2008
). Copyright 2008 with
permission of John Wiley and Sons
Earth minerals
Specific surface area (m
2
/g)
Glyphosate sorption (mmol/kg)
Aluminum and iron oxides
Gibbsite
45
72
Ferrihydrite
343
635
Goethite
85
125
Hematite
33
86
Clay silicates
K
+
-Kaolinite 1
12
3.9
K
+
-Kaolinite 2
22
6.9
K
+
-Illite
43
5.2
K
+
-Montmorillonite
32
6.5
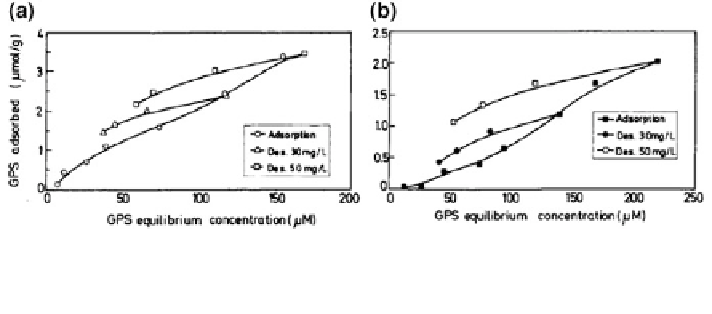
Fig. 18.17 Adsorption-desorption isotherms of glyphosate (GP) on montmorillonite a in the
absence, and b in the presence of Cu. Reprinted with permission from Morillo et al. (
1997
).
Copyright 1997 American Chemical Society
An hysteretic effect was observed during adsorption-desorption studies of GP
with soils and clay minerals, suggesting potential irreversible retention. Morillo
et al. (
1997
) examined GP retention and release on/from montmorillonite in the
presence and absence of Cu
2+
in the desorbing aqueous electrolyte solution
(Fig.
18.17
). The desorption curves showed that about 60 % of GP initially
adsorbed on montmorillonite was retained on the clay surface after three succes-
sive desorption procedures. In this case, some GP initially adsorbed on broken
borders of montmorillonite is replaced by hydroxyl groups or by water during the
desorption process. When Cu
2+
ions are added to the desorption solution, a
reduction is observed in the amount of GP that is retained (Fig.
18.17
b). Here,
some interlayer Cu
2+
desorbs by cation exchange with Na
+
ions from the elec-
trolyte solution, favoring formation of GP complexes with Cu
2+
in solution.
Nonionic pesticides Chlorinated benzenes—aromatic organic compounds with
the chemical formula C
6
H
6-n
Cl
n
—are common hydrophobic solvents widely used
as intermediate materials in the manufacture of various nonionic pesticides. The
adsorption of chlorinated benzenes on natural soil-sediment materials is illustrated