Biology Reference
In-Depth Information
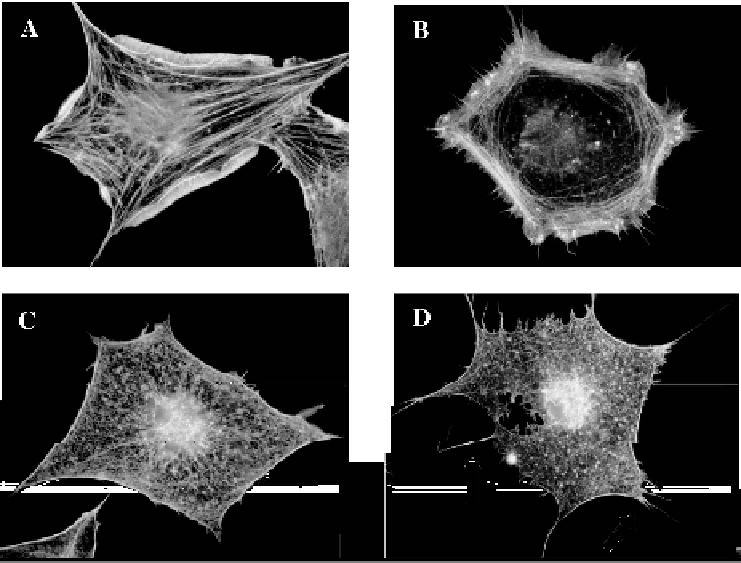
Figure 4.1 Activation of EphB2 and EphA7 triggers actin reorganization in fibroblasts.
Serum-starved subconfluent Swiss 3T3 cells were microinjected with plasmids encoding
EphB2 (A), EphB2+N17Rac (C), EphA7 (B), EphA7+N17Rac (D). After 3 h cells were
treated with 1 mg ephrin-B1-Fc (A, C) or ephrin-A4-Fc (B, D) for 15min before fixation.
Actin filaments (A-D) were visualized with TRITC-phalloidin. Arrows in D indicate
filopodia
filaments (Nobes and Hall, 1995; Kozma et al., 1995; Ridley and Hall, 1992;
Ridley et al., 1992).
To determine whether the lamellipodia induced by stimulated EphB2 and
EphA7 are a result of activation of the small GTPase Rac, we have co-injected
cells with a eukaryotic expression construct encoding myc-tagged dominant
negative Rac(N17Rac). Expression of N17Rac protein, confirmed by
immunofluorescence after fixing cells, does not alter the increase in
phosphotyrosine immunofluorescence observed after addition of ephrins to
Eph receptor expressing cells (data not shown). Expression of N17Rac
completely blocks lamellipodial protrusions stimulated by ephrin-B1 activa-
tion of EphB2, and by ephrin-A4 activation of EphA7 (Figure 4.1C and
4.1D). However, Rac inhibition does not interfere with the assembly of
filopodia in activated EphA7-expressing cells (see arrows in Figure 4.1D).
N17Rac also inhibits the assembly of actin stress fibres after EphB2
and EphA7 activation, suggesting that stress fibres are produced as a result