Chemistry Reference
In-Depth Information
3.5.2.2 Garnets
The most important iron and silicon containing garnet minerals are the pyrope-
almandine series (Mg,Fe
2+
)
3
Al
2
(SiO
4
)
3
and andradite Ca
3
(Al,Fe
3+
)
2
(SiO
4
)
3
. When
Mg is replaced by Mn the garnet is called spessartine (Mn,Fe
2+
)
3
Al
2
(SiO
4
)
3
. The
garnet structures contain SiO
4
tetrahedra with commonly trivalent cations in
6-coordination and divalent cations in 8-coordination.
Members of the pyrope-almandine series exhibit a doublet at RT with an isomer
shift d
Fe
= 1.2-1.4 mm/s and a very large quadrupole splitting D in the range
3.47-3.70 mm/s [
180
]. An additional doublet of Fe
3+
due to the six-fold coordi-
nated Al site is observed with d
Fe
= 0.35-0.45 mm/s and D = 0.29-0.75 mm/s
when going from almandine towards andradite. These intermediate minerals are
often called grossular. Pure almandine orders magnetically around 10 and at 4 K
an Fe
2+
octet is observed with a hyperfine field of 23-24. 5 T [
181
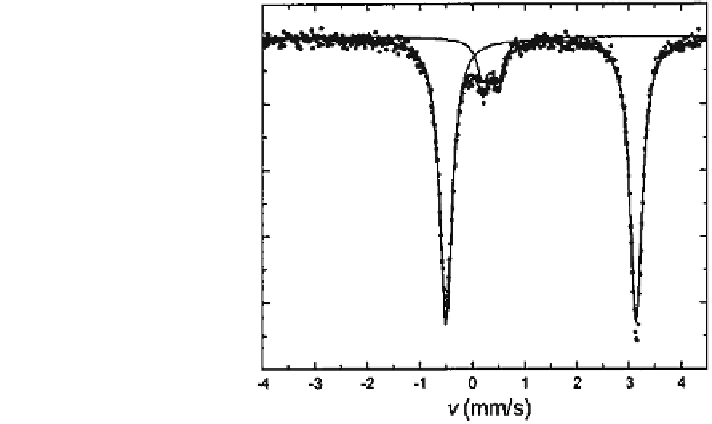
]. The typical
large quadrupole splitting in garnets is shown in the spectrum of spessartine in
Fig.
3.23
.
Andradite possesses at RT the typical Fe
3+
hyperfine parameters, namely
d
Fe
= 0.39-0.41 mm/s and D = 0.5-0.6 mm/s [
180
]. The latter is, however,
strongly dependent on the composition. It orders magnetically below 11 and at 4 K
the hyperfine field amounts to 51.9 T [
182
]. Actually, the spectrum is asymmetric
and can be analyzed with two sextets having different quadrupole shifts 2e = 0.32
and -0.16 mm/s, due to a complex spin structure with different orientations with
respect to the EFG principal axis. In some garnets such as titanium andradite,
Ca
3
(Fe,Ti,Si)
5
O
12
,Fe
3+
also occupies partly the tetrahedral Si sites, exhibiting
very low isomer shifts and rather high quadrupole splittings [
183
].
Fig. 3.23 RT spectrum of
spessartine showing the
predominant Fe
2+
doublet
with the typical large
quadrupole splitting for
garnets. The spectrum shows
an additional small doublet
from octahedral Fe
3+
(after
Eeckhout et al. [
308
])