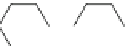Chemistry Reference
In-Depth Information
Ph
Ph
93
Fig. 28
Structure of 14,15-diphenyldibenzo[
f
,
j
]picene (
93
)[
85
]
Ph Ph
O
O
t
-Bu
t
-Bu
t
-Bu
t
-Bu
2
32a
/
n
-BuLi
R
R
94
95
: R = OH, 91%
96
:R=H,83%
Et
3
SiH
CF
3
CO
2
H
Ph
-Bu
refluxing
toluene
KO
t
t
-Bu
t
-Bu
A
E
Ph
97
,47%
Scheme 20
Synthesis of diindeno-fused 1,14-diphenyldibenzo[
c
,
g
]phenanthrene
97
[
76
]
Interestingly, the rotation barrier for the two phenyl substituents is only 12.9 kcal/
mol at 20
C, smaller than the rotation barrier of 18.5 kcal/mol of
85
. Presumably the
added strain at the transition state of rotation could spread over a more extended
framework in
97
. As a result, the molecule could better accommodate the additional
distortion caused by rotation. The AB signals of themethylene hydrogen atoms remain
unaffected at 100
C, indicating a slow rate of racemization on the NMR time scale.
4.3
1,14-Dimethoxydibenzo[
c
,
g
]phenanthrene and Related
Compounds
The photodehydrocyclization reaction of a stilbene-like precursor was found to be
efficient in producing 1,14-dimethoxydibenzo[
c
,
g
]phenanthrene [
86
]. Tandem rad-
ical cyclization reactions have also been used to produce a 1,14-dimethoxy