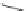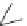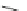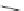Chemistry Reference
In-Depth Information
a
S
S
S
Cu
[6]CPP
S
S
Br
S
S
40
b
n
CHO
PPh
3
Br
[4+2]
Wittig
+
CPPs
n
P
Ph
3
B
r
CHO
41
c
Br
I
Br
macrocycles
+
OMe
I
OMe
42
43
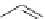
Fig. 20 V¨gtle's attempted syntheses of cycloparaphenylenes [
59
]
It is interesting to note that the nine-membered macrocycle must be the result
of homocoupling of the corresponding nine-membered bisboronate. With these
macrocycles in hand, Jasti developed a reductive aromatization technique that
circumvented the 1,2-phenyl shift observed under acidic or Lewis acidic conditions
for similar systems (Fig.
22
)[
61
].
Treating a solution of the desired macrocycle with lithium naphthalenide at
78
C for 30 min offered the corresponding CPPs in moderate yield [
32
]. The
electron-transfer mechanism of this reduction prevents rearrangement by
circumventing carbocation intermediates, and builds in an amazing amount of strain